Estradiol

|

|
| Names
|
| Pronunciation
|
ES
-tr?-
DY
-ohl
[1]
[2]
|
| IUPAC name
Estra-1,3,5(10)-triene-3,17β-diol
|
Systematic IUPAC name
(1
S
,3a
S
,3b
R
,9b
S
,11a
S
)-11a-Methyl-2,3,3a,3b,4,5,9b,10,11,11a-decahydro-1
H
-cyclopenta[
a
]phenanthrene-1,7-diol
|
| Other names
Oestradiol; E2; 17β-Estradiol; 17β-Oestradiol
|
| Identifiers
|
|
|
|
|
|
|
| ChEBI
|
|
| ChEMBL
|
|
| ChemSpider
|
|
| DrugBank
|
|
| ECHA InfoCard
|
100.000.022

|
| EC Number
|
|
| KEGG
|
|
|
|
|
| UNII
|
|
|
|
|
InChI=1S/C18H24O2/c1-18-9-8-14-13-5-3-12(19)10-11(13)2-4-15(14)16(18)6-7-17(18)20/h3,5,10,14-17,19-20H,2,4,6-9H2,1H3/t14-,15-,16+,17+,18+/m1/s1
 Y Y
Key: VOXZDWNPVJITMN-ZBRFXRBCSA-N
 Y Y
|
C[C@]12CC[C@@H]3c4ccc(cc4CC[C@H]3[C@@H]1CC[C@@H]2O)O
|
| Properties
|
|
|
C
18
H
24
O
2
|
| Molar mass
|
272.38 g/mol
|
|
|
-186.6·10
?6
cm
3
/mol
|
| Pharmacology
|
|
|
G03CA03
(
WHO
)
|
| License data
|
|
|
|
Oral
,
sublingual
,
intranasal
,
topical
/
transdermal
,
vaginal
,
intramuscular
or
subcutaneous
(as an
ester
),
subdermal implant
|
| Pharmacokinetics
:
|
|
|
Oral: <5%
[3]
|
|
|
~98%:
[3]
[4]
?
Albumin
: 60%
?
SHBG
: 38%
? Free: 2%
|
|
|
Liver
(via
hydroxylation
,
sulfation
,
glucuronidation
)
|
|
|
Oral: 13?20 hours
[3]
Sublingual: 8?18 hours
[5]
Topical (gel): 36.5 hours
[6]
|
|
|
Urine
: 54%
[3]
Feces
: 6%
[3]
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25 °C [77 °F], 100 kPa).
|
Chemical compound
Estradiol
(
E2
), also spelled
oestradiol
, is an
estrogen
steroid hormone
and the major
female
sex hormone
. It is involved in the regulation of female
reproductive cycles
such as
estrous
and
menstrual cycles
. Estradiol is responsible for the development of female
secondary sexual characteristics
such as the
breasts
,
widening of the hips
and a
female pattern of fat distribution
. It is also important in the development and maintenance of female
reproductive tissues
such as the
mammary glands
,
uterus
and
vagina
during
puberty
,
adulthood
and
pregnancy
.
[7]
It also has important effects in many other
tissues
including
bone
,
fat
,
skin
,
liver
, and the
brain
.
Though estradiol levels in males are much lower than in females, estradiol has important roles in males as well. Apart from humans and other
mammals
, estradiol is also found in most
vertebrates
and
crustaceans
,
insects
,
fish
, and other
animal
species
.
[8]
[9]
Estradiol is produced especially within the
follicles
of the
ovaries
, but also in other tissues including the
testicles
, the
adrenal glands
, fat,
liver
, the breasts, and the brain. Estradiol is
produced in the body
from
cholesterol
through a series of
reactions
and
intermediates
.
[10]
The major
pathway
involves the formation of
androstenedione
, which is then converted by
aromatase
into
estrone
and is subsequently converted into estradiol. Alternatively, androstenedione can be converted into
testosterone
, which can then be converted into estradiol. Upon
menopause
in females, production of estrogens by the ovaries stops and estradiol levels decrease to very low levels.
In addition to its role as a natural hormone, estradiol is used as a
medication
, for instance in
menopausal hormone therapy
and
feminizing hormone therapy
for
transgender women
; for information on estradiol as a medication, see the
estradiol (medication)
article.
Biological function
[
edit
]
Sexual development
[
edit
]
The development of
secondary sex characteristics
in women is driven by estrogens, to be specific, estradiol.
[11]
[12]
These changes are initiated at the time of
puberty
, most are enhanced during the reproductive years, and become less pronounced with declining estradiol support after
menopause
. Thus, estradiol produces
breast development
, and is responsible for changes in the
body shape
, affecting bones, joints, and
fat deposition
.
[11]
[12]
In females, estradiol induces breast development,
widening of the hips
, a
feminine fat distribution
(with fat deposited particularly in the breasts, hips, thighs, and buttocks), and maturation of the
vagina
and
vulva
, whereas it mediates the
pubertal growth spurt
(indirectly via increased
growth hormone
secretion)
[13]
and
epiphyseal closure
(thereby limiting
final height
) in both sexes.
[11]
[12]
Reproduction
[
edit
]
Female reproductive system
[
edit
]
In the female, estradiol acts as a growth hormone for tissue of the reproductive organs, supporting the lining of the
vagina
, the cervical glands, the
endometrium
, and the lining of the fallopian tubes. It enhances growth of the
myometrium
. Estradiol appears necessary to maintain
oocytes
in the
ovary
. During the
menstrual cycle
, estradiol produced by the growing follicles triggers, via a positive feedback system, the hypothalamic-pituitary events that lead to the
luteinizing hormone
surge, inducing ovulation. In the luteal phase, estradiol, in conjunction with
progesterone
, prepares the endometrium for
implantation
. During
pregnancy
, estradiol increases due to
placental
production. The effect of estradiol, together with
estrone
and
estriol
, in
pregnancy
is less clear. They may promote uterine blood flow, myometrial growth, stimulate breast growth and at term, promote cervical softening and expression of myometrial
oxytocin
receptors.
[
citation needed
]
In baboons, blocking of estrogen production leads to pregnancy loss, suggesting estradiol has a role in the maintenance of pregnancy. Research is investigating the role of estrogens in the process of initiation of
labor
. Actions of estradiol are required before the exposure of progesterone in the luteal phase.
[
citation needed
]
Male reproductive system
[
edit
]
The effect of estradiol (and estrogens in general) upon male reproduction is complex. Estradiol is produced by action of
aromatase
mainly in the
Leydig cells
of the
mammalian
testis
, but also by some
germ cells
and the
Sertoli cells
of immature mammals.
[14]
It functions (
in vitro
) to prevent
apoptosis
of male
sperm
cells.
[15]
While some studies in the early 1990s claimed a connection between globally declining
sperm counts
and estrogen exposure in the environment,
[16]
later studies found no such connection, nor evidence of a general decline in sperm counts.
[17]
[18]
Suppression of estradiol production in a subpopulation of subfertile men may improve the
semen
analysis.
[19]
Males with certain
sex chromosome
genetic conditions
, such as
Klinefelter's syndrome
, will have a higher level of estradiol.
[20]
Skeletal system
[
edit
]
Estradiol has a profound effect on bone. Individuals without it (or other estrogens) will become tall and
eunuchoid
, as
epiphyseal
closure is delayed or may not take place.
[21]
Bone density
is also affected, resulting in early
osteopenia
and
osteoporosis
.
[22]
Low levels of estradiol may also predict fractures, with post-menopausal women having the highest incidence of
bone fracture
.
[23]
Women past menopause experience an accelerated loss of bone mass due to a relative estrogen deficiency.
[24]
Skin health
[
edit
]
The
estrogen receptor
, as well as the
progesterone receptor
, have been detected in the
skin
, including in
keratinocytes
and
fibroblasts
.
[25]
[26]
At
menopause
and thereafter, decreased levels of female
sex hormones
result in
atrophy
, thinning, and increased
wrinkling
of the skin and a reduction in skin
elasticity
, firmness, and strength.
[25]
[26]
These skin changes constitute an acceleration in
skin aging
and are the result of decreased
collagen
content, irregularities in the
morphology
of
epidermal
skin cells
, decreased
ground substance
between
skin fibers
, and reduced
capillaries
and
blood flow
.
[25]
[26]
The skin also becomes more
dry
during menopause, which is due to reduced skin
hydration
and
surface lipids
(sebum production).
[25]
Along with chronological aging and photoaging, estrogen deficiency in menopause is one of the three main factors that predominantly influences skin aging.
[25]
Hormone replacement therapy
consisting of systemic treatment with estrogen alone or in combination with a progestogen, has well-documented and considerable beneficial effects on the skin of postmenopausal women.
[25]
[26]
These benefits include increased skin collagen content, skin thickness and elasticity, and skin hydration and surface lipids.
[25]
[26]
Topical estrogen has been found to have similar beneficial effects on the skin.
[25]
In addition, a study has found that topical 2% progesterone cream significantly increases skin elasticity and firmness and observably decreases wrinkles in peri- and postmenopausal women.
[26]
Skin hydration and surface lipids, on the other hand, did not significantly change with topical progesterone.
[26]
These findings suggest that progesterone, like estrogen, also has beneficial effects on the skin, and may be independently protective against skin aging.
[26]
Nervous system
[
edit
]
Estrogens can be produced in the
brain
from steroid precursors. As
antioxidants
, they have been found to have
neuroprotective
function.
[27]
The positive and negative
feedback loops
of the
menstrual cycle
involve ovarian estradiol as the link to the hypothalamic-pituitary system to regulate
gonadotropins
.
[28]
Estrogen is considered to play a significant role in women's mental health, with links suggested between the hormone level, mood and well-being. Sudden drops or fluctuations in, or long periods of sustained low levels of estrogen may be correlated with significant mood-lowering. Clinical recovery from depression postpartum, perimenopause, and postmenopause was shown to be effective after levels of estrogen were stabilized and/or restored.
[29]
[30]
The volumes of
sexually dimorphic
brain structures in
transgender women
were found to change and approximate typical female brain structures when exposed to estrogen concomitantly with androgen deprivation over a period of months,
[31]
suggesting that estrogen and/or androgens have a significant part to play in sex differentiation of the brain, both
prenatally
and later in life.
There is also evidence the programming of adult male sexual behavior in many vertebrates is largely dependent on estradiol produced during prenatal life and early infancy.
[32]
It is not yet known whether this process plays a significant role in human sexual behavior, although evidence from other mammals tends to indicate a connection.
[33]
Estrogen has been found to increase the
secretion
of oxytocin and to increase the
expression
of its
receptor
, the
oxytocin receptor
, in the
brain
.
[34]
In women, a single dose of estradiol has been found to be sufficient to increase circulating oxytocin concentrations.
[35]
Gynecological cancers
[
edit
]
Estradiol has been tied to the development and progression of cancers such as breast cancer, ovarian cancer and endometrial cancer. Estradiol affects target tissues mainly by interacting with two
nuclear receptors
called
estrogen receptor α
(ERα) and
estrogen receptor β
(ERβ).
[36]
[37]
One of the functions of these estrogen receptors is the modulation of
gene expression
. Once estradiol binds to the ERs, the receptor complexes then bind to specific
DNA sequences
, possibly causing damage to the DNA and an increase in cell division and
DNA replication
.
Eukaryotic cells
respond to damaged DNA by stimulating or impairing G1, S, or G2 phases of the cell cycle to initiate
DNA repair
. As a result, cellular transformation and cancer cell proliferation occurs.
[38]
Cardiovascular system
[
edit
]
Estrogen affects certain
blood vessels
. Improvement in arterial blood flow has been demonstrated in
coronary arteries
.
[39]
17-beta-estradiol (E2) is considered the most potent estrogen found in humans. E2 influences vascular function, apoptosis, and damage during cardiac ischemia and reperfusion. E2 can protect the heart and individual cardiac myocytes from injuries related to ischemia. After a heart attack or long periods of hypertension, E2 inhibits the adverse effects of pathologic remodeling of the heart.
[40]
During
pregnancy
, high levels of estrogens, namely estradiol, increase
coagulation
and the risk of
venous thromboembolism
.
Absolute and relative incidence of venous thromboembolism (VTE) during pregnancy and the postpartum period
| Absolute incidence of first VTE per 10,000 person?years during pregnancy and the postpartum period
|
|
|
Swedish data A
|
Swedish data B
|
English data
|
Danish data
|
| Time period
|
N
|
Rate (95% CI)
|
N
|
Rate (95% CI)
|
N
|
Rate (95% CI)
|
N
|
Rate (95% CI)
|
| Outside pregnancy
|
1105
|
4.2 (4.0?4.4)
|
1015
|
3.8 (?)
|
1480
|
3.2 (3.0?3.3)
|
2895
|
3.6 (3.4?3.7)
|
| Antepartum
|
995
|
20.5 (19.2?21.8)
|
690
|
14.2 (13.2?15.3)
|
156
|
9.9 (8.5?11.6)
|
491
|
10.7 (9.7?11.6)
|
| Trimester 1
|
207
|
13.6 (11.8?15.5)
|
172
|
11.3 (9.7?13.1)
|
23
|
4.6 (3.1?7.0)
|
61
|
4.1 (3.2?5.2)
|
| Trimester 2
|
275
|
17.4 (15.4?19.6)
|
178
|
11.2 (9.7?13.0)
|
30
|
5.8 (4.1?8.3)
|
75
|
5.7 (4.6?7.2)
|
| Trimester 3
|
513
|
29.2 (26.8?31.9)
|
340
|
19.4 (17.4?21.6)
|
103
|
18.2 (15.0?22.1)
|
355
|
19.7 (17.7?21.9)
|
| Around delivery
|
115
|
154.6 (128.8?185.6)
|
79
|
106.1 (85.1?132.3)
|
34
|
142.8 (102.0?199.8)
|
?
|
| Postpartum
|
649
|
42.3 (39.2?45.7)
|
509
|
33.1 (30.4?36.1)
|
135
|
27.4 (23.1?32.4)
|
218
|
17.5 (15.3?20.0)
|
| Early postpartum
|
584
|
75.4 (69.6?81.8)
|
460
|
59.3 (54.1?65.0)
|
177
|
46.8 (39.1?56.1)
|
199
|
30.4 (26.4?35.0)
|
| Late postpartum
|
65
|
8.5 (7.0?10.9)
|
49
|
6.4 (4.9?8.5)
|
18
|
7.3 (4.6?11.6)
|
319
|
3.2 (1.9?5.0)
|
| Incidence rate ratios (IRRs) of first VTE during pregnancy and the postpartum period
|
|
|
Swedish data A
|
Swedish data B
|
English data
|
Danish data
|
| Time period
|
IRR* (95% CI)
|
IRR* (95% CI)
|
IRR (95% CI)†
|
IRR (95% CI)†
|
| Outside pregnancy
|
Reference (i.e., 1.00)
|
| Antepartum
|
5.08 (4.66?5.54)
|
3.80 (3.44?4.19)
|
3.10 (2.63?3.66)
|
2.95 (2.68?3.25)
|
| Trimester 1
|
3.42 (2.95?3.98)
|
3.04 (2.58?3.56)
|
1.46 (0.96?2.20)
|
1.12 (0.86?1.45)
|
| Trimester 2
|
4.31 (3.78?4.93)
|
3.01 (2.56?3.53)
|
1.82 (1.27?2.62)
|
1.58 (1.24?1.99)
|
| Trimester 3
|
7.14 (6.43?7.94)
|
5.12 (4.53?5.80)
|
5.69 (4.66?6.95)
|
5.48 (4.89?6.12)
|
| Around delivery
|
37.5 (30.9?44.45)
|
27.97 (22.24?35.17)
|
44.5 (31.68?62.54)
|
?
|
| Postpartum
|
10.21 (9.27?11.25)
|
8.72 (7.83?9.70)
|
8.54 (7.16?10.19)
|
4.85 (4.21?5.57)
|
| Early postpartum
|
19.27 (16.53?20.21)
|
15.62 (14.00?17.45)
|
14.61 (12.10?17.67)
|
8.44 (7.27?9.75)
|
| Late postpartum
|
2.06 (1.60?2.64)
|
1.69 (1.26?2.25)
|
2.29 (1.44?3.65)
|
0.89 (0.53?1.39)
|
| Notes:
Swedish data A = Using any code for VTE regardless of confirmation. Swedish data B = Using only algorithm-confirmed VTE. Early postpartum = First 6 weeks after delivery. Late postpartum = More than 6 weeks after delivery. * = Adjusted for age and calendar year. † = Unadjusted ratio calculated based on the data provided.
Source:
[41]
|
Other functions
[
edit
]
Estradiol has complex effects on the
liver
. It affects the production of multiple
proteins
, including
lipoproteins
, binding proteins, and proteins responsible for
blood clotting
.
[
citation needed
]
In high amounts, estradiol can lead to
cholestasis
, for instance
cholestasis of pregnancy
.
Certain gynecological conditions are dependent on estrogen, such as
endometriosis
,
leiomyomata
uteri, and
uterine bleeding
.
[
citation needed
]
Biological activity
[
edit
]
Estradiol acts primarily as an
agonist
of the
estrogen receptor
(ER), a
nuclear
steroid hormone receptor
. There are two subtypes of the ER,
ERα
and
ERβ
, and estradiol potently binds to and activates both of these receptors. The result of ER activation is a modulation of
gene transcription
and
expression
in ER-expressing
cells
, which is the predominant mechanism by which estradiol mediates its biological effects in the body. Estradiol also acts as an agonist of
membrane estrogen receptors
(mERs), such as
GPER
(GPR30), a recently discovered non-nuclear receptor for estradiol, via which it can mediate a variety of rapid, non-
genomic
effects.
[42]
Unlike the case of the ER, GPER appears to be
selective
for estradiol, and shows very low
affinities
for other endogenous estrogens, such as estrone and
estriol
.
[43]
Additional mERs besides GPER include
ER-X
,
ERx
, and
G
q
-mER
.
[44]
[45]
ERα/ERβ are in inactive state trapped in multimolecular chaperone complexes organized around the heat shock protein 90 (HSP90), containing p23 protein, and immunophilin, and located in majority in cytoplasm and partially in nucleus. In the E2 classical pathway or estrogen classical pathway, estradiol enters the
cytoplasm
, where it interacts with ERs. Once bound E2, ERs dissociate from the molecular chaperone complexes and become competent to dimerize, migrate to nucleus, and to bind to specific DNA sequences (
estrogen response element
, ERE), allowing for gene transcription which can take place over hours and days.
Given by
subcutaneous injection
in mice, estradiol is about 10-fold more potent than estrone and about 100-fold more potent than estriol.
[46]
[47]
[48]
As such, estradiol is the main estrogen in the body, although the roles of estrone and estriol as estrogens are said not to be negligible.
[48]
Biochemistry
[
edit
]
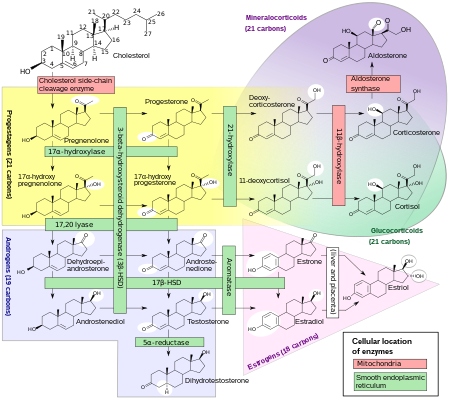 Human
steroidogenesis
, showing estradiol at bottom right.
[49]
Human
steroidogenesis
, showing estradiol at bottom right.
[49]
Biosynthesis
[
edit
]
Estradiol, like other
steroid hormones
, is derived from
cholesterol
. After
side chain
cleavage and using the Δ
5
or the Δ
4
- pathway,
androstenedione
is the key intermediary. A portion of the androstenedione is converted to testosterone, which in turn undergoes conversion to estradiol by aromatase. In an alternative pathway, androstenedione is
aromatized
to
estrone
, which is subsequently converted to estradiol via
17β-hydroxysteroid dehydrogenase
(17β-HSD).
[50]
During the reproductive years, most estradiol in women is produced by the
granulosa cells
of the ovaries by the aromatization of androstenedione (produced in the theca folliculi cells) to estrone, followed by conversion of estrone to estradiol by 17β-HSD. Smaller amounts of estradiol are also produced by the
adrenal cortex
, and, in men, by the testes.
[
medical citation needed
]
Estradiol is not produced in the
gonads
only; in particular,
fat cells
produce active precursors to estradiol, and will continue to do so even after menopause.
[51]
Estradiol is also produced in the
brain
and in
arterial walls
.
In men, approximately 15 to 25% of circulating estradiol is produced in the
testicles
.
[52]
[53]
The rest is synthesized via peripheral aromatization of testosterone into estradiol and of androstenedione into estrone (which is then transformed into estradiol via peripheral 17β-HSD).
[52]
[53]
This peripheral aromatization occurs predominantly in
adipose tissue
, but also occurs in other
tissues
such as
bone
,
liver
, and the
brain
.
[52]
Approximately 40 to 50 μg of estradiol is produced per day in men.
[52]
Distribution
[
edit
]
In plasma, estradiol is largely bound to SHBG and
albumin
. Only about 2.21% (± 0.04%) of estradiol is free and biologically active. The percentage remains constant throughout the
menstrual cycle
.
[54]
Metabolism
[
edit
]
Estradiol
unidentified
unidentified
|
Inactivation of estradiol includes conversion to less-active estrogens, such as estrone and estriol. Estriol is the major urinary
metabolite
.
[
citation needed
]
Estradiol is
conjugated
in the
liver
to form
estrogen conjugates
like
estradiol sulfate
,
estradiol glucuronide
and, as such, excreted via the
kidneys
. Some of the water-soluble conjugates are excreted via the
bile duct
, and partly reabsorbed after
hydrolysis
from the
intestinal tract
. This
enterohepatic circulation
contributes to maintaining estradiol levels.
Estradiol is also metabolized via
hydroxylation
into
catechol estrogens
. In the liver, it is non-specifically metabolized by
CYP1A2
,
CYP3A4
, and
CYP2C9
via 2-hydroxylation into
2-hydroxyestradiol
, and by
CYP2C9
,
CYP2C19
, and
CYP2C8
via 17β-hydroxy dehydrogenation into
estrone
,
[55]
with various other
cytochrome P450
(CYP)
enzymes
and
metabolic transformations
also being involved.
[56]
Estradiol is additionally
conjugated
with an
ester
into
lipoidal estradiol
forms like
estradiol palmitate
and
estradiol stearate
to a certain extent; these esters are stored in
adipose tissue
and may act as a very long-lasting reservoir of estradiol.
[57]
[58]
Excretion
[
edit
]
Estradiol is
excreted
in the form of
glucuronide
and
sulfate
estrogen conjugates
in
urine
. Following an
intravenous injection
of
labeled
estradiol in women, almost 90% is excreted in urine and
feces
within 4 to 5 days.
[59]
[60]
Enterohepatic recirculation
causes a delay in excretion of estradiol.
[59]
Levels
[
edit
]
 Estradiol levels across the menstrual cycle in 36 normally cycling, ovulatory women, based on 956 specimens.
[61]
The horizontal dashed lines are the mean integrated levels for each curve. The vertical dashed line in the center is mid-cycle.
Estradiol levels across the menstrual cycle in 36 normally cycling, ovulatory women, based on 956 specimens.
[61]
The horizontal dashed lines are the mean integrated levels for each curve. The vertical dashed line in the center is mid-cycle.
Levels of estradiol in premenopausal women are highly variable throughout the menstrual cycle and reference ranges widely vary from source to source.
[62]
Estradiol levels are minimal and according to most laboratories range from 20 to 80 pg/mL during the early to mid follicular phase (or the first week of the menstrual cycle, also known as menses).
[63]
[64]
Levels of estradiol gradually increase during this time and through the mid to late follicular phase (or the second week of the menstrual cycle) until the pre-ovulatory phase.
[62]
[63]
At the time of pre-ovulation (a period of about 24 to 48 hours), estradiol levels briefly surge and reach their highest concentrations of any other time during the menstrual cycle.
[62]
Circulating levels are typically between 130 and 200 pg/mL at this time, but in some women may be as high as 300 to 400 pg/mL, and the upper limit of the reference range of some laboratories are even greater (for instance, 750 pg/mL).
[62]
[63]
[65]
[66]
[67]
Following ovulation (or mid-cycle) and during the latter half of the menstrual cycle or the luteal phase, estradiol levels plateau and fluctuate between around 100 and 150 pg/mL during the early and mid luteal phase, and at the time of the late luteal phase, or a few days before menstruation, reach a low of around 40 pg/mL.
[62]
[64]
The mean integrated levels of estradiol during a full menstrual cycle have variously been reported by different sources as 80, 120, and 150 pg/mL.
[64]
[68]
[69]
Although contradictory reports exist, one study found mean integrated estradiol levels of 150 pg/mL in younger women whereas mean integrated levels ranged from 50 to 120 pg/mL in older women.
[69]
During the reproductive years of human females, levels of estradiol are somewhat higher than that of estrone, except during the early follicular phase of the menstrual cycle; thus, estradiol may be considered the predominant estrogen during human female reproductive years in terms of absolute serum levels and estrogenic activity.
[
citation needed
]
During pregnancy, estriol becomes the predominant circulating estrogen, and this is the only time at which estetrol occurs in the body, while during menopause, estrone predominates (both based on serum levels).
[
citation needed
]
The estradiol produced by male humans, from testosterone, is present at serum levels roughly comparable to those of
postmenopausal
women (14?55 versus <35 pg/mL, respectively).
[
citation needed
]
It has also been reported that if concentrations of estradiol in a 70-year-old man are compared to those of a 70-year-old woman, levels are approximately 2- to 4-fold higher in the man.
[70]
Measurement
[
edit
]
In women, serum estradiol is measured in a
clinical laboratory
and reflects primarily the activity of the ovaries. The Estradiol blood test measures the amount of estradiol in the blood.
[77]
It is used to check the function of the ovaries, placenta, adrenal glands.
[77]
This can detect baseline estrogen in women with
amenorrhea
or menstrual dysfunction, and to detect the state of hypoestrogenicity and menopause. Furthermore, estrogen monitoring during fertility therapy assesses follicular growth and is useful in monitoring the treatment. Estrogen-producing tumors will demonstrate persistent high levels of estradiol and other estrogens. In
precocious puberty
, estradiol levels are inappropriately increased.
Ranges
[
edit
]
Individual laboratory results should always be interpreted using the ranges provided by the laboratory that performed the test.
Reference ranges for the blood content of estradiol during the menstrual cycle
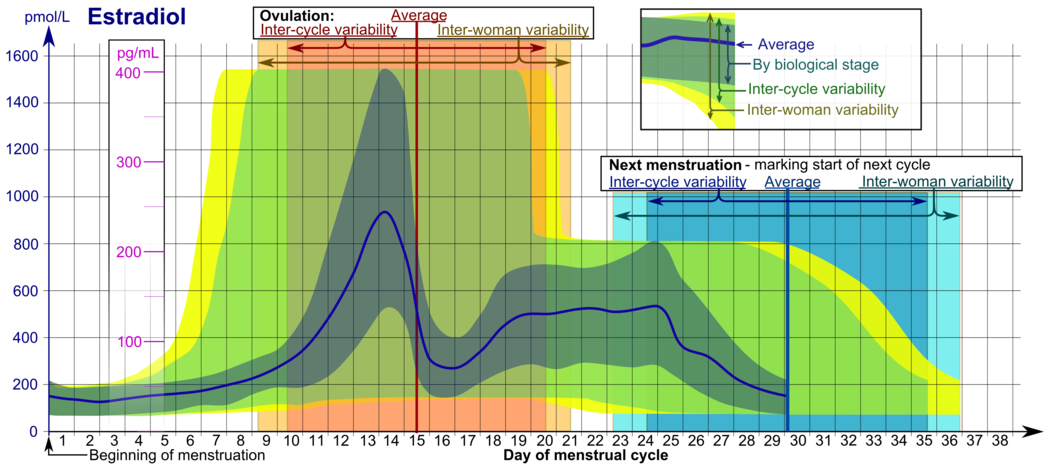 Reference ranges for the blood content
of estradiol during the
menstrual cycle
Reference ranges for the blood content
of estradiol during the
menstrual cycle
- The ranges denoted
By biological stage
may be used in closely monitored menstrual cycles in regard to other markers of its biological progression, with the time scale being compressed or stretched to how much faster or slower, respectively, the cycle progresses compared to an average cycle.
- The ranges denoted
Inter-cycle variability
are more appropriate to use in unmonitored cycles with only the beginning of menstruation known, but where the woman accurately knows her average cycle lengths and time of ovulation, and that they are somewhat averagely regular, with the time scale being compressed or stretched to how much a woman's average cycle length is shorter or longer, respectively, than the average of the population.
- The ranges denoted
Inter-woman variability
are more appropriate to use when the average cycle lengths and time of ovulation are unknown, but only the beginning of menstruation is given.
[81]
In the normal menstrual cycle, estradiol levels measure typically <50 pg/mL at menstruation, rise with follicular development (peak: 200 pg/mL), drop briefly at ovulation, and rise again during the luteal phase for a second peak. At the end of the luteal phase, estradiol levels drop to their menstrual levels unless there is a pregnancy.
During pregnancy, estrogen levels, including estradiol, rise steadily toward term. The source of these estrogens is the
placenta
, which aromatizes
prohormones
produced in the fetal adrenal gland.
Production rates, secretion rates, clearance rates, and blood levels of major sex hormones
| Sex
|
Sex hormone
|
Reproductive
phase
|
Blood
production rate
|
Gonadal
secretion rate
|
Metabolic
clearance rate
|
Reference range (serum levels)
|
| SI
units
|
Non-
SI
units
|
| Men
|
Androstenedione
|
?
|
2.8 mg/day
|
1.6 mg/day
|
2200 L/day
|
2.8?7.3 nmol/L
|
80?210 ng/dL
|
| Testosterone
|
?
|
6.5 mg/day
|
6.2 mg/day
|
950 L/day
|
6.9?34.7 nmol/L
|
200?1000 ng/dL
|
| Estrone
|
?
|
150 μg/day
|
110 μg/day
|
2050 L/day
|
37?250 pmol/L
|
10?70 pg/mL
|
| Estradiol
|
?
|
60 μg/day
|
50 μg/day
|
1600 L/day
|
<37?210 pmol/L
|
10?57 pg/mL
|
| Estrone sulfate
|
?
|
80 μg/day
|
Insignificant
|
167 L/day
|
600?2500 pmol/L
|
200?900 pg/mL
|
| Women
|
Androstenedione
|
?
|
3.2 mg/day
|
2.8 mg/day
|
2000 L/day
|
3.1?12.2 nmol/L
|
89?350 ng/dL
|
| Testosterone
|
?
|
190 μg/day
|
60 μg/day
|
500 L/day
|
0.7?2.8 nmol/L
|
20?81 ng/dL
|
| Estrone
|
Follicular phase
|
110 μg/day
|
80 μg/day
|
2200 L/day
|
110?400 pmol/L
|
30?110 pg/mL
|
| Luteal phase
|
260 μg/day
|
150 μg/day
|
2200 L/day
|
310?660 pmol/L
|
80?180 pg/mL
|
| Postmenopause
|
40 μg/day
|
Insignificant
|
1610 L/day
|
22?230 pmol/L
|
6?60 pg/mL
|
| Estradiol
|
Follicular phase
|
90 μg/day
|
80 μg/day
|
1200 L/day
|
<37?360 pmol/L
|
10?98 pg/mL
|
| Luteal phase
|
250 μg/day
|
240 μg/day
|
1200 L/day
|
699?1250 pmol/L
|
190?341 pg/mL
|
| Postmenopause
|
6 μg/day
|
Insignificant
|
910 L/day
|
<37?140 pmol/L
|
10?38 pg/mL
|
| Estrone sulfate
|
Follicular phase
|
100 μg/day
|
Insignificant
|
146 L/day
|
700?3600 pmol/L
|
250?1300 pg/mL
|
| Luteal phase
|
180 μg/day
|
Insignificant
|
146 L/day
|
1100?7300 pmol/L
|
400?2600 pg/mL
|
| Progesterone
|
Follicular phase
|
2 mg/day
|
1.7 mg/day
|
2100 L/day
|
0.3?3 nmol/L
|
0.1?0.9 ng/mL
|
| Luteal phase
|
25 mg/day
|
24 mg/day
|
2100 L/day
|
19?45 nmol/L
|
6?14 ng/mL
|
Notes and sources
Notes:
"The
concentration
of a steroid in the circulation is determined by the rate at which it is secreted from glands, the rate of metabolism of precursor or prehormones into the steroid, and the rate at which it is extracted by tissues and metabolized. The
secretion rate
of a steroid refers to the total secretion of the compound from a gland per unit time. Secretion rates have been assessed by sampling the venous effluent from a gland over time and subtracting out the arterial and peripheral venous hormone concentration. The
metabolic clearance rate
of a steroid is defined as the volume of blood that has been completely cleared of the hormone per unit time. The
production rate
of a steroid hormone refers to entry into the blood of the compound from all possible sources, including secretion from glands and conversion of prohormones into the steroid of interest. At steady state, the amount of hormone entering the blood from all sources will be equal to the rate at which it is being cleared (metabolic clearance rate) multiplied by blood concentration (production rate = metabolic clearance rate × concentration). If there is little contribution of prohormone metabolism to the circulating pool of steroid, then the production rate will approximate the secretion rate."
Sources:
See template.
|
Medical use
[
edit
]
Estradiol is used as a
medication
, primarily in
hormone therapy
for
menopausal
symptoms
as well as
feminizing hormone therapy
for trans individuals.
[82]
Chemistry
[
edit
]
Structures of major endogenous estrogens
Note the
hydroxyl
(?OH)
groups
: estrone (E1) has one, estradiol (E2) has two, estriol (E3) has three, and estetrol (E4) has four.
|
Estradiol is an
estrane
steroid
.
[82]
It is also known as 17β-estradiol (to distinguish it from
17α-estradiol
) or as estra-1,3,5(10)-triene-3,17β-diol. It has two
hydroxyl groups
, one at the C3 position and the other at the 17β position, as well as three
double bonds
in the A
ring
. Due to its two hydroxyl groups, estradiol is often abbreviated as E2. The structurally related estrogens, estrone (E1), estriol (E3), and
estetrol
(E4) have one, three, and four hydroxyl groups, respectively.
Neuropsychopharmacology
[
edit
]
In a randomized, double-blind, placebo-controlled study, estradiol was shown to have gender-specific effects on fairness sensitivity. Overall, when the division of a given amount of money was framed as either fair or unfair in a modified version of the
ultimatum game
, estradiol increased the acceptance rate of fair-framed proposals among men and decreased it among women. However, among the placebo-group "the mere belief of receiving estradiol treatment significantly increased the acceptance of unfair-framed offers in both sexes", indicating that so-called "environmental" factors played a role in organising the responses towards these presentations of the
ultimatum game
.
[83]
History
[
edit
]
The discovery of estrogen is usually credited to the
American
scientists
Edgar Allen
and
Edward A. Doisy
.
[84]
[85]
In 1923, they observed that injection of fluid from
porcine
ovarian follicles
produced
pubertal
- and
estrus
-type changes (including
vaginal
,
uterine
, and
mammary gland
changes and
sexual receptivity
) in
sexually immature
,
ovariectomized
mice and rats.
[84]
[85]
[86]
These findings demonstrated the existence of a
hormone
which is produced by the
ovaries
and is involved in
sexual maturation
and
reproduction
.
[84]
[85]
[86]
At the time of its discovery, Allen and Doisy did not name the hormone, and simply referred to it as an "ovarian hormone" or "follicular hormone";
[85]
others referred to it variously as
feminin
,
folliculin
,
menformon
,
thelykinin
, and
emmenin
.
[87]
[88]
In 1926, Parkes and Bellerby coined the term
estrin
to describe the hormone on the basis of it inducing estrus in animals.
[89]
[87]
Estrone
was isolated and purified independently by Allen and Doisy and
German
scientist
Adolf Butenandt
in 1929, and
estriol
was isolated and purified by Marrian in 1930; they were the first estrogens to be identified.
[85]
[90]
[91]
Estradiol, the most potent of the three major estrogens, was the last of the three to be identified.
[85]
[89]
It was discovered by Schwenk and Hildebrant in 1933, who
synthesized
it via
reduction
of estrone.
[85]
Estradiol was subsequently isolated and purified from sow ovaries by Doisy in 1935, with its
chemical structure
determined simultaneously,
[92]
and was referred to variously as
dihydrotheelin
,
dihydrofolliculin
,
dihydrofollicular hormone
, and
dihydroxyestrin
.
[85]
[93]
[94]
In 1935, the name
estradiol
and the term
estrogen
were formally established by the Sex Hormone Committee of the Health Organization of the
League of Nations
; this followed the names estrone (which was initially called theelin, progynon, folliculin, and ketohydroxyestrin) and estriol (initially called theelol and trihydroxyestrin) having been established in 1932 at the first meeting of the International Conference on the Standardization of Sex Hormones in
London
.
[89]
[95]
Following its discovery, a
partial synthesis
of estradiol from
cholesterol
was developed by Inhoffen and Hohlweg in 1940, and a
total synthesis
was developed by Anner and Miescher in 1948.
[85]
Society and culture
[
edit
]
Etymology
[
edit
]
The name
estradiol
derives from
estra-
,
Gk.
ο?στρο?
(
oistros
, literally meaning "verve or inspiration"),
[96]
which refers to the
estrane
steroid
ring
system, and
-diol
, a chemical term and suffix indicating that the compound is a type of
alcohol
bearing two
hydroxyl
groups
.
References
[
edit
]
- ^
Ford SR, Roach SS (7 October 2013).
Roach's Introductory Clinical Pharmacology
. Lippincott Williams & Wilkins. pp. 525?.
ISBN
978-1-4698-3214-2
.
- ^
Hochadel M (1 April 2015).
Mosby's Drug Reference for Health Professions
. Elsevier Health Sciences. pp. 602?.
ISBN
978-0-323-31103-8
.
- ^
a
b
c
d
e
Stanczyk FZ, Archer DF, Bhavnani BR (June 2013). "Ethinyl estradiol and 17β-estradiol in combined oral contraceptives: pharmacokinetics, pharmacodynamics and risk assessment".
Contraception
.
87
(6): 706?27.
doi
:
10.1016/j.contraception.2012.12.011
.
PMID
23375353
.
- ^
Falcone T, Hurd WW (2007).
Clinical Reproductive Medicine and Surgery
. Elsevier Health Sciences. pp. 22?.
ISBN
978-0-323-03309-1
.
- ^
Price TM, Blauer KL, Hansen M, Stanczyk F, Lobo R, Bates GW (March 1997). "Single-dose pharmacokinetics of sublingual versus oral administration of micronized 17 beta-estradiol".
Obstetrics and Gynecology
.
89
(3): 340?5.
doi
:
10.1016/S0029-7844(96)00513-3
.
PMID
9052581
.
S2CID
71641652
.
- ^
Naunton M, Al Hadithy AF, Brouwers JR, Archer DF (2006). "Estradiol gel: review of the pharmacology, pharmacokinetics, efficacy, and safety in menopausal women".
Menopause
.
13
(3): 517?27.
doi
:
10.1097/01.gme.0000191881.52175.8c
.
PMID
16735950
.
S2CID
42748448
.
- ^
Ryan KJ (August 1982). "Biochemistry of aromatase: significance to female reproductive physiology".
Cancer Research
.
42
(8 Suppl): 3342s?3344s.
PMID
7083198
.
- ^
Mechoulam R, Brueggemeier RW, Denlinger DL (September 1984). "Estrogens in insects".
Cellular and Molecular Life Sciences
.
40
(9): 942?944.
doi
:
10.1007/BF01946450
.
S2CID
31950471
.
- ^
Ozon R (1972).
"Estrogens in Fishes, Amphibians, Reptiles, and Birds"
. In Idler DR (ed.).
Steroids In Nonmammalian Vertebrates
. Oxford: Elsevier Science. pp. 390?414.
ISBN
978-0323140980
.
- ^
Saldanha, Colin J., Luke Remage-Healey, and Barney A. Schlinger. "Synaptocrine signaling: steroid synthesis and action at the synapse." Endocrine reviews 32.4 (2011): 532?549.
- ^
a
b
c
McMillan JA, Feigin RD, DeAngelis C, Jones MD (2006).
Oski's Pediatrics: Principles & Practice
. Lippincott Williams & Wilkins. pp. 550?.
ISBN
978-0-7817-3894-1
.
- ^
a
b
c
Craig CR, Stitzel RE (2004).
Modern Pharmacology with Clinical Applications
. Lippincott Williams & Wilkins. pp. 706?.
ISBN
978-0-7817-3762-3
.
- ^
Preedy VR (2 December 2011).
Handbook of Growth and Growth Monitoring in Health and Disease
. Springer Science & Business Media. pp. 2661?.
ISBN
978-1-4419-1794-2
.
- ^
Carreau S, Lambard S, Delalande C, Denis-Galeraud I, Bilinska B, Bourguiba S (April 2003).
"Aromatase expression and role of estrogens in male gonad : a review"
.
Reproductive Biology and Endocrinology
.
1
: 35.
doi
:
10.1186/1477-7827-1-35
.
PMC
155680
.
PMID
12747806
.
- ^
Pentikainen V, Erkkila K, Suomalainen L, Parvinen M, Dunkel L (May 2000).
"Estradiol acts as a germ cell survival factor in the human testis in vitro"
.
The Journal of Clinical Endocrinology and Metabolism
.
85
(5): 2057?67.
doi
:
10.1210/jcem.85.5.6600
.
PMID
10843196
.
- ^
Sharpe RM, Skakkebaek NE (May 1993). "Are oestrogens involved in falling sperm counts and disorders of the male reproductive tract?".
Lancet
.
341
(8857): 1392?5.
doi
:
10.1016/0140-6736(93)90953-E
.
PMID
8098802
.
S2CID
33135527
.
- ^
Handelsman DJ (2001). "Estrogens and falling sperm counts".
Reproduction, Fertility, and Development
.
13
(4): 317?24.
doi
:
10.1071/rd00103
.
PMID
11800170
.
- ^
Fisch H, Goldstedin R (2003).
"Environmental estrogens and sperm counts"
(PDF)
.
Pure and Applied Chemistry
.
75
(11?12): 2181?2193.
doi
:
10.1351/pac200375112181
.
S2CID
11068097
. Archived from
the original
(PDF)
on 4 March 2016
. Retrieved
29 December
2015
.
- ^
Raman JD, Schlegel PN (February 2002). "Aromatase inhibitors for male infertility".
The Journal of Urology
.
167
(2 Pt 1): 624?9.
doi
:
10.1016/S0022-5347(01)69099-2
.
PMID
11792932
.
- ^
Visootsak J, Graham JM (October 2006).
"Klinefelter syndrome and other sex chromosomal aneuploidies"
.
Orphanet Journal of Rare Diseases
.
1
(42): 42.
doi
:
10.1186/1750-1172-1-42
.
PMC
1634840
.
PMID
17062147
.
- ^
Vanderschueren D, Laurent MR, Claessens F, Gielen E, Lagerquist MK, Vandenput L, et al. (December 2014).
"Sex steroid actions in male bone"
.
Endocrine Reviews
.
35
(6): 906?60.
doi
:
10.1210/er.2014-1024
.
PMC
4234776
.
PMID
25202834
.
- ^
Carani C, Qin K, Simoni M, Faustini-Fustini M, Serpente S, Boyd J, et al. (July 1997).
"Effect of testosterone and estradiol in a man with aromatase deficiency"
.
The New England Journal of Medicine
.
337
(2): 91?5.
doi
:
10.1056/NEJM199707103370204
.
PMID
9211678
.
- ^
Bergh C, Wennergren D, Moller M, Brisby H (21 December 2020).
"Fracture incidence in adults in relation to age and gender: A study of 27,169 fractures in the Swedish Fracture Register in a well-defined catchment area"
.
PLOS ONE
.
15
(12): e0244291.
Bibcode
:
2020PLoSO..1544291B
.
doi
:
10.1371/journal.pone.0244291
.
PMC
7751975
.
PMID
33347485
.
- ^
Albright F
, Smith PH, Richardson AM (31 May 1941). "Postmenopausal Osteoporosis: Its Clinical Features".
JAMA
.
116
(22): 2465?2474.
doi
:
10.1001/jama.1941.02820220007002
.
- ^
a
b
c
d
e
f
g
h
Raine-Fenning NJ, Brincat MP, Muscat-Baron Y (2003). "Skin aging and menopause : implications for treatment".
American Journal of Clinical Dermatology
.
4
(6): 371?8.
doi
:
10.2165/00128071-200304060-00001
.
PMID
12762829
.
S2CID
20392538
.
- ^
a
b
c
d
e
f
g
h
Holzer G, Riegler E, Honigsmann H, Farokhnia S, Schmidt JB, Schmidt B (September 2005). "Effects and side-effects of 2% progesterone cream on the skin of peri- and postmenopausal women: results from a double-blind, vehicle-controlled, randomized study".
The British Journal of Dermatology
.
153
(3): 626?34.
doi
:
10.1111/j.1365-2133.2005.06685.x
.
PMID
16120154
.
S2CID
6077829
.
- ^
Behl C, Widmann M, Trapp T, Holsboer F (November 1995). "17-beta estradiol protects neurons from oxidative stress-induced cell death in vitro".
Biochemical and Biophysical Research Communications
.
216
(2): 473?82.
doi
:
10.1006/bbrc.1995.2647
.
PMID
7488136
.
- ^
Meethal SV, Liu T, Chan HW, Ginsburg E, Wilson AC, Gray DN, Bowen RL, Vonderhaar BK, Atwood CS (August 2009).
"Identification of a regulatory loop for the synthesis of neurosteroids: a steroidogenic acute regulatory protein-dependent mechanism involving hypothalamic-pituitary-gonadal axis receptors"
.
Journal of Neurochemistry
.
110
(3): 1014?27.
doi
:
10.1111/j.1471-4159.2009.06192.x
.
PMC
2789665
.
PMID
19493163
.
- ^
Douma SL, Husband C, O'Donnell ME, Barwin BN, Woodend AK (2005). "Estrogen-related mood disorders: reproductive life cycle factors".
Advances in Nursing Science
.
28
(4): 364?75.
doi
:
10.1097/00012272-200510000-00008
.
PMID
16292022
.
S2CID
9172877
.
- ^
Lasiuk GC, Hegadoren KM (October 2007). "The effects of estradiol on central serotonergic systems and its relationship to mood in women".
Biological Research for Nursing
.
9
(2): 147?60.
doi
:
10.1177/1099800407305600
.
PMID
17909167
.
S2CID
37965502
.
- ^
Hulshoff HE, Cohen-Kettenis PT, Van Haren NE, Peper JS, Brans RG, Cahn W, Schnack HG, Gooren LJ, Kahn RS (July 2006).
"Changing your sex changes your brain: influences of testosterone and estrogen on adult human brain structure"
.
European Journal of Endocrinology
.
155
(suppl_1): 107?114.
doi
:
10.1530/eje.1.02248
.
- ^
Harding CF (June 2004).
"Hormonal modulation of singing: hormonal modulation of the songbird brain and singing behavior"
.
Annals of the New York Academy of Sciences
.
1016
(1): 524?39.
Bibcode
:
2004NYASA1016..524H
.
doi
:
10.1196/annals.1298.030
.
PMID
15313793
.
S2CID
12457330
. Archived from
the original
on 27 September 2007.
- ^
Simerly RB (27 March 2002).
"Wired for reproduction: organization and development of sexually dimorphic circuits in the mammalian forebrain"
(PDF)
.
Annual Review of Neuroscience
.
25
: 507?36.
doi
:
10.1146/annurev.neuro.25.112701.142745
.
PMID
12052919
. Archived from
the original
(PDF)
on 1 October 2008
. Retrieved
7 March
2007
.
- ^
Goldstein I, Meston CM, Davis S, Traish A (17 November 2005).
Women's Sexual Function and Dysfunction: Study, Diagnosis and Treatment
. CRC Press. pp. 205?.
ISBN
978-1-84214-263-9
.
- ^
Acevedo-Rodriguez A, Mani SK, Handa RJ (2015).
"Oxytocin and Estrogen Receptor β in the Brain: An Overview"
.
Frontiers in Endocrinology
.
6
: 160.
doi
:
10.3389/fendo.2015.00160
.
PMC
4606117
.
PMID
26528239
.
- ^
Bulzomi P, Bolli A, Galluzzo P, Leone S, Acconcia F, Marino M (January 2010).
"Naringenin and 17beta-estradiol coadministration prevents hormone-induced human cancer cell growth"
.
IUBMB Life
.
62
(1): 51?60.
doi
:
10.1002/iub.279
.
PMID
19960539
.
S2CID
7903757
.
- ^
Sreeja S, Santhosh Kumar TR, Lakshmi BS, Sreeja S (July 2012). "Pomegranate extract demonstrate a selective estrogen receptor modulator profile in human tumor cell lines and in vivo models of estrogen deprivation".
The Journal of Nutritional Biochemistry
.
23
(7): 725?32.
doi
:
10.1016/j.jnutbio.2011.03.015
.
PMID
21839626
.
- ^
Thomas CG, Strom A, Lindberg K, Gustafsson JA (June 2011). "Estrogen receptor beta decreases survival of p53-defective cancer cells after DNA damage by impairing G₂/M checkpoint signaling".
Breast Cancer Research and Treatment
.
127
(2): 417?27.
doi
:
10.1007/s10549-010-1011-z
.
PMID
20623183
.
S2CID
6752694
.
- ^
Collins P, Rosano GM, Sarrel PM, Ulrich L, Adamopoulos S, Beale CM, McNeill JG, Poole-Wilson PA (July 1995). "17 beta-Estradiol attenuates acetylcholine-induced coronary arterial constriction in women but not men with coronary heart disease".
Circulation
.
92
(1): 24?30.
doi
:
10.1161/01.CIR.92.1.24
.
PMID
7788912
.
- ^
Knowlton, A.A.; Lee, A.R. (July 2012).
"Estrogen and the cardiovascular system"
.
Pharmacology & Therapeutics
.
135
(1): 54?70.
doi
:
10.1016/j.pharmthera.2012.03.007
.
PMC
5688223
.
PMID
22484805
.
- ^
Abdul Sultan A, West J, Stephansson O, Grainge MJ, Tata LJ, Fleming KM, Humes D, Ludvigsson JF (November 2015).
"Defining venous thromboembolism and measuring its incidence using Swedish health registries: a nationwide pregnancy cohort study"
.
BMJ Open
.
5
(11): e008864.
doi
:
10.1136/bmjopen-2015-008864
.
PMC
4654387
.
PMID
26560059
.
- ^
Prossnitz ER, Barton M (May 2014).
"Estrogen biology: new insights into GPER function and clinical opportunities"
.
Molecular and Cellular Endocrinology
.
389
(1?2): 71?83.
doi
:
10.1016/j.mce.2014.02.002
.
PMC
4040308
.
PMID
24530924
.
- ^
Prossnitz ER, Arterburn JB, Sklar LA (2007).
"GPR30: A G protein-coupled receptor for estrogen"
.
Mol. Cell. Endocrinol
. 265?266: 138?42.
doi
:
10.1016/j.mce.2006.12.010
.
PMC
1847610
.
PMID
17222505
.
- ^
Soltysik K, Czekaj P (April 2013). "Membrane estrogen receptors - is it an alternative way of estrogen action?".
Journal of Physiology and Pharmacology
.
64
(2): 129?42.
PMID
23756388
.
- ^
Micevych PE, Kelly MJ (2012).
"Membrane estrogen receptor regulation of hypothalamic function"
.
Neuroendocrinology
.
96
(2): 103?10.
doi
:
10.1159/000338400
.
PMC
3496782
.
PMID
22538318
.
- ^
Labhart A (6 December 2012).
Clinical Endocrinology: Theory and Practice
. Springer Science & Business Media. pp. 548?.
ISBN
978-3-642-96158-8
.
- ^
Tucker SB (2007).
Maternal, Fetal, & Neonatal Physiology: A Clinical Perspective
. Elsevier Health Sciences. pp. 43?.
ISBN
978-1-4160-2944-1
.
- ^
a
b
Hall JE (31 May 2015).
Guyton and Hall Textbook of Medical Physiology E-Book
. Elsevier Health Sciences. pp. 1043?.
ISBN
978-0-323-38930-3
.
- ^
Haggstrom M, Richfield D (2014).
"Diagram of the pathways of human steroidogenesis"
.
WikiJournal of Medicine
.
1
(1).
doi
:
10.15347/wjm/2014.005
.
ISSN
2002-4436
.
- ^
Boron WF, Boulpaep EL (2003).
Medical Physiology: A Cellular And Molecular Approach
. Elsevier/Saunders. p. 1300.
ISBN
978-1-4160-2328-9
.
- ^
Mutschler E, Schafer-Korting M (2001).
Arzneimittelwirkungen
(in German) (8 ed.). Stuttgart: Wissenschaftliche Verlagsgesellschaft. pp. 434, 444.
ISBN
978-3-8047-1763-3
.
- ^
a
b
c
d
Melmed S (1 January 2016).
Williams Textbook of Endocrinology
. Elsevier Health Sciences. pp. 710?.
ISBN
978-0-323-29738-7
.
- ^
a
b
Marcus R, Feldman D, Dempster DW, Luckey M, Cauley JA (13 June 2013).
Osteoporosis
. Academic Press. pp. 331?.
ISBN
978-0-12-398252-0
.
- ^
Wu CH, Motohashi T, Abdel-Rahman HA, Flickinger GL, Mikhail G (August 1976). "Free and protein-bound plasma estradiol-17 beta during the menstrual cycle".
The Journal of Clinical Endocrinology and Metabolism
.
43
(2): 436?45.
doi
:
10.1210/jcem-43-2-436
.
PMID
950372
.
- ^
Cheng ZN, Shu Y, Liu ZQ, Wang LS, Ou-Yang DS, Zhou HH (February 2001). "Role of cytochrome P450 in estradiol metabolism in vitro".
Acta Pharmacologica Sinica
.
22
(2): 148?54.
PMID
11741520
.
- ^
Lee AJ, Cai MX, Thomas PE, Conney AH, Zhu BT (August 2003).
"Characterization of the oxidative metabolites of 17beta-estradiol and estrone formed by 15 selectively expressed human cytochrome p450 isoforms"
.
Endocrinology
.
144
(8): 3382?98.
doi
:
10.1210/en.2003-0192
.
PMID
12865317
.
- ^
Oettel M, Schillinger E (6 December 2012).
Estrogens and Antiestrogens I: Physiology and Mechanisms of Action of Estrogens and Antiestrogens
. Springer Science & Business Media. pp. 235?237.
ISBN
978-3-642-58616-3
.
- ^
Oettel M, Schillinger E (6 December 2012).
Estrogens and Antiestrogens II: Pharmacology and Clinical Application of Estrogens and Antiestrogen
. Springer Science & Business Media. pp. 268, 271.
ISBN
978-3-642-60107-1
.
- ^
a
b
Dorfman, Ralph I. (1961). "Steroid Hormone Metabolism".
Radioactive Isotopes in Physiology Diagnostics and Therapy / Kunstliche Radioaktive Isotope in Physiologie Diagnostik und Therapie
. pp. 1223?1241.
doi
:
10.1007/978-3-642-49761-2_39
.
ISBN
978-3-642-49477-2
.
- ^
Sandberg AA, Slaunwhite WR (August 1957).
"Studies on phenolic steroids in human subjects. II. The metabolic fate and hepato-biliary-enteric circulation of C14-estrone and C14-estradiol in women"
.
J. Clin. Invest
.
36
(8): 1266?78.
doi
:
10.1172/JCI103524
.
PMC
1072719
.
PMID
13463090
.
- ^
a
b
http://www.ilexmedical.com/files/PDF/Estradiol_ARC.pdf
[
bare URL PDF
]
- ^
a
b
c
d
e
Becker JB, Berkley KJ, Geary N, Hampson E,
Herman JP
, Young E (4 December 2007).
Sex Differences in the Brain: From Genes to Behavior
. Oxford University Press. pp. 64?.
ISBN
978-0-19-804255-6
.
Estradiol levels are minimal during the earliest days of the follicular phase, but increasing concentrations are released into the general circulation as the follicle matures. The highest levels are reached about 24 to 48 hours before the LH peak. In fact, the pre-ovulatory peak in estradiol represents its highest concentration during the entire menstrual cycle. Serum concentrations at this time are typically about 130?200 pg/mL, but concentrations as high as 300?400 pg/mL can be achieved in some women. Following a transient fall in association with ovulation, estradiol secretion is restored by production from the corpus luteum during the luteal phase. Plateau levels of around 100?150 pg/mL (Abraham, 1978; Thorneycroft et al., 1971) are most often seen during the period from ?10 to ?5 days before the onset of menses. With the regression of the corpus luteum, estradiol levels fall, gradually in some women and precipitously in others, during the last few days of the luteal phase. This ushers in the onset of menses, the sloughing of the endometrium. Serum estradiol during menses is approximately 30?50 pg/mL. (Source.)
- ^
a
b
c
Strauss JR, Barbieri RL (2009).
Yen and Jaffe's Reproductive Endocrinology: Physiology, Pathophysiology, and Clinical Management
. Elsevier Health Sciences. pp. 807?.
ISBN
978-1-4160-4907-4
.
In most laboratories, serum estradiol levels range from 20 to 80 pg/mL during the early to midfollicular phase of the menstrual cycle and peak at 200 to 500 pg/mL during the preovulatory surge. During the midluteal phase, serum estradiol levels range from 60 to 200 pg/mL.
- ^
a
b
c
Christian C, von Schoultz B (15 March 1994).
Hormone Replacement Therapy: Standardized or Individually Adapted Doses?
. CRC Press. pp. 60?.
ISBN
978-1-85070-545-1
.
Plasma levels of estradiol range from 40 to 80 pg/mL during the 1st week of the ovarian cycle (early follicular phase) and from 80 to 300 pg/mL during the 2nd week (mid- and late follicular phase including periovulatory peak). Then during the 3rd and 4th weeks, estradiol fluctuates between 100 and 150 pg/mL (early and mid-luteal phase) to 40 pg/mL a few days before menstruation (late luteal phase). The mean integrated estradiol level during a full 28-day normal cycle is around 80 pg/mL.
- ^
Jameson JL, De Groot LJ (18 May 2010).
Endocrinology: Adult and Pediatric
. Elsevier Health Sciences. pp. 2812?.
ISBN
978-1-4557-1126-0
.
Midcycle: 150-750 pg/mL
- ^
Hay ID, Wass JA (26 January 2009).
Clinical Endocrine Oncology
. John Wiley & Sons. pp. 623?.
ISBN
978-1-4443-0023-9
.
Mid-cycle: 110-330 pg/mL
- ^
Dons RF (12 July 1994).
Endocrine and Metabolic Testing Manual
. CRC Press. pp. 8?.
ISBN
978-0-8493-7657-3
.
Ovulatory: 200-400 pg/mL
- ^
Notelovitz M, van Keep PA (6 December 2012).
The Climacteric in Perspective: Proceedings of the Fourth International Congress on the Menopause, held at Lake Buena Vista, Florida, October 28 ? November 2, 1984
. Springer Science & Business Media. pp. 397?.
ISBN
978-94-009-4145-8
.
[...] following the menopause, circulating estradiol levels decrease from a premenopausal mean of 120 pg/mL to only 13 pg/mL.
- ^
a
b
Muller EE, MacLeod RM (6 December 2012).
Neuroendocrine Perspectives
. Springer Science & Business Media. pp. 121?.
ISBN
978-1-4612-3554-5
.
[...] [premenopausal] mean [estradiol] concentration of 150 pg/mL [...]
- ^
Sayed Y, Taxel P (December 2003). "The use of estrogen therapy in men".
Current Opinion in Pharmacology
.
3
(6): 650?4.
doi
:
10.1016/j.coph.2003.07.004
.
PMID
14644018
.
- ^
Nichols KC, Schenkel L, Benson H (1984). "17 beta-estradiol for postmenopausal estrogen replacement therapy".
Obstet Gynecol Surv
.
39
(4): 230?45.
doi
:
10.1097/00006254-198404000-00022
.
PMID
6717863
.
- ^
Cynthia C. Chernecky; Barbara J. Berger (31 October 2012).
Laboratory Tests and Diagnostic Procedures ? E-Book
. Elsevier Health Sciences. pp. 488?.
ISBN
978-1-4557-4502-9
.
- ^
- ^
Kenneth L. Becker (2001).
Principles and Practice of Endocrinology and Metabolism
. Lippincott Williams & Wilkins. pp. 889, 1059?1060, 2153.
ISBN
978-0-7817-1750-2
.
- ^
Lalit Bajaj; Stephen Berman (1 January 2011).
Berman's Pediatric Decision Making
. Elsevier Health Sciences. pp. 160?.
ISBN
978-0-323-05405-8
.
- ^
Kuhl H (2003).
"Ostrogene fur den Mann?"
[Estrogens for the man?].
Blickpunkt der Mann
.
1
(3): 6?12.
ISSN
1727-0669
.
- ^
a
b
"Estradiol blood test: MedlinePlus Medical Encyclopedia"
.
medlineplus.gov
. Retrieved
6 May
2019
.
- ^
a
b
c
d
e
f
g
h
i
j
GPNotebook ? reference range (oestradiol)
Retrieved on 27 September 2009
- ^
a
b
Values taken from day 1 after LH surge in:
Stricker R, Eberhart R, Chevailler MC, Quinn FA, Bischof P, Stricker R (2006). "Establishment of detailed reference values for luteinizing hormone, follicle stimulating hormone, estradiol, and progesterone during different phases of the menstrual cycle on the Abbott ARCHITECT analyzer".
Clinical Chemistry and Laboratory Medicine
.
44
(7): 883?7.
doi
:
10.1515/CCLM.2006.160
.
PMID
16776638
.
S2CID
524952
.
as PDF
- ^
a
b
c
d
Total amount multiplied by 0.022 according to 2.2% presented in:
Wu CH, Motohashi T, Abdel-Rahman HA, Flickinger GL, Mikhail G (August 1976). "Free and protein-bound plasma estradiol-17 beta during the menstrual cycle".
The Journal of Clinical Endocrinology and Metabolism
.
43
(2): 436?45.
doi
:
10.1210/jcem-43-2-436
.
PMID
950372
.
[
original research?
]
- ^
Haggstrom M (2014).
"Reference ranges for estradiol, progesterone, luteinizing hormone and follicle-stimulating hormone during the menstrual cycle"
.
WikiJournal of Medicine
.
1
(1).
doi
:
10.15347/wjm/2014.001
.
ISSN
2002-4436
.
- ^
a
b
Kuhl H (August 2005). "Pharmacology of estrogens and progestogens: influence of different routes of administration".
Climacteric
.
8
(1 Suppl 1): 3?63.
doi
:
10.1080/13697130500148875
.
PMID
16112947
.
S2CID
24616324
.
- ^
Coenjaerts M, Pape F, Santoso V, Grau F, Stoffel-Wagner B, Philipsen A, Schultz J, Hurlemann R, Scheele D (September 2021). "Sex differences in economic decision-making: Exogenous estradiol has opposing effects on fairness framing in women and men".
Eur. Neuropsychopharmacol
.
50
(2): 46?54.
doi
:
10.1016/j.euroneuro.2021.04.006
.
hdl
:
20.500.11811/11066
.
ISSN
0924-977X
.
PMID
33957337
.
S2CID
233982738
.
- ^
a
b
c
Loriaux DL, Loriaux L (14 March 2016).
A Biographical History of Endocrinology
. John Wiley & Sons. pp. 345?.
ISBN
978-1-119-20246-2
.
- ^
a
b
c
d
e
f
g
h
i
Lauritzen C, Studd JW (22 June 2005).
Current Management of the Menopause
. CRC Press. pp. 44?.
ISBN
978-0-203-48612-2
.
- ^
a
b
Allen E, Doisy EA (1923). "An Ovarian Hormone".
Journal of the American Medical Association
.
81
(10): 819.
doi
:
10.1001/jama.1923.02650100027012
.
ISSN
0002-9955
.
- ^
a
b
Gruhn JG, Kazer RR (11 November 2013).
Hormonal Regulation of the Menstrual Cycle: The Evolution of Concepts
. Springer Science & Business Media. pp. 69?73.
ISBN
978-1-4899-3496-3
.
- ^
Newerla GJ (1944). "The History of the Discovery and Isolation of the Female Sex Hormones".
New England Journal of Medicine
.
230
(20): 595?604.
doi
:
10.1056/NEJM194405182302001
.
ISSN
0028-4793
.
- ^
a
b
c
Fritz MA, Speroff L (28 March 2012).
Clinical Gynecologic Endocrinology and Infertility
. Lippincott Williams & Wilkins. pp. 750?.
ISBN
978-1-4511-4847-3
.
- ^
Parl FF (2000).
Estrogens, Estrogen Receptor and Breast Cancer
. IOS Press. pp. 4?.
ISBN
978-0-9673355-4-4
.
- ^
Sartorelli AC, Johns DG (27 November 2013).
Antineoplastic and Immunosuppressive Agents
. Springer Science & Business Media. pp. 104?.
ISBN
978-3-642-65806-8
.
- ^
Shoupe D, Haseltine FP (6 December 2012).
Contraception
. Springer Science & Business Media. pp. 2?.
ISBN
978-1-4612-2730-4
.
- ^
MacCorquodale DW, Thayer SA, Doisy EA (1935). "The Crystalline Ovarian Follicular Hormone".
Experimental Biology and Medicine
.
32
(7): 1182.
doi
:
10.3181/00379727-32-8020P
.
ISSN
1535-3702
.
S2CID
83557813
.
- ^
Chemicals Identified in Human Biological Media: A Data Base
. Design and Development Branch, Survey and Analysis Division, Office of Program Integration and Information, Office of Pesticides and Toxic Substances, Environmental Protection Agency. 1981. pp. 114?.
- ^
Fausto-Sterling A (2000).
Sexing the Body: Gender Politics and the Construction of Sexuality
. Basic Books. pp.
189
?.
ISBN
978-0-465-07714-4
.
- ^
"Greek Word Study Tool: oistros"
.
Perseus Digital Library
. Retrieved
28 December
2011
.
|
|---|
| ER
Tooltip Estrogen receptor
| | Agonists
|
- Steroidal:
2-Hydroxyestradiol
- 2-Hydroxyestrone
- 3-Methyl-19-methyleneandrosta-3,5-dien-17β-ol
- 3α-Androstanediol
- 3α,5α-Dihydrolevonorgestrel
- 3β,5α-Dihydrolevonorgestrel
- 3α-Hydroxytibolone
- 3β-Hydroxytibolone
- 3β-Androstanediol
- 4-Androstenediol
- 4-Androstenedione
- 4-Fluoroestradiol
- 4-Hydroxyestradiol
- 4-Hydroxyestrone
- 4-Methoxyestradiol
- 4-Methoxyestrone
- 5-Androstenediol
- 7-Oxo-DHEA
- 7α-Hydroxy-DHEA
- 7α-Methylestradiol
- 7β-Hydroxyepiandrosterone
- 8,9-Dehydroestradiol
- 8,9-Dehydroestrone
- 8β-VE2
- 10β,17β-Dihydroxyestra-1,4-dien-3-one (DHED)
- 11β-Chloromethylestradiol
- 11β-Methoxyestradiol
- 15α-Hydroxyestradiol
- 16-Ketoestradiol
- 16-Ketoestrone
- 16α-Fluoroestradiol
- 16α-Hydroxy-DHEA
- 16α-Hydroxyestrone
- 16α-Iodoestradiol
- 16α-LE2
- 16β-Hydroxyestrone
- 16β,17α-Epiestriol (16β-hydroxy-17α-estradiol)
- 17α-Estradiol
(
alfatradiol
)
- 17α-Dihydroequilenin
- 17α-Dihydroequilin
- 17α-Epiestriol (16α-hydroxy-17α-estradiol)
- 17α-Ethynyl-3α-androstanediol
- 17α-Ethynyl-3β-androstanediol
- 17β-Dihydroequilenin
- 17β-Dihydroequilin
- 17β-Methyl-17α-dihydroequilenin
- Abiraterone
- Abiraterone acetate
- Alestramustine
- Almestrone
- Anabolic steroids
(e.g.,
testosterone
and
esters
,
methyltestosterone
,
metandienone (methandrostenolone)
,
nandrolone
and
esters
, many others; via estrogenic metabolites)
- Atrimustine
- Bolandiol
- Bolandiol dipropionate
- Butolame
- Clomestrone
- Cloxestradiol
- Conjugated estriol
- Conjugated estrogens
- Cyclodiol
- Cyclotriol
- DHEA
- DHEA-S
- ent
-Estradiol
- Epiestriol (16β-epiestriol, 16β-hydroxy-17β-estradiol)
- Epimestrol
- Equilenin
- Equilin
- ERA-63 (ORG-37663)
- Esterified estrogens
- Estetrol
- Estradiol
- Estramustine
- Estramustine phosphate
- Estrapronicate
- Estrazinol
- Estriol
- Estrofurate
- Estrogenic substances
- Estromustine
- Estrone
- Etamestrol (eptamestrol)
- Ethinylandrostenediol
- Ethinylestradiol
- Ethinylestriol
- Ethylestradiol
- Etynodiol
- Etynodiol diacetate
- Hexolame
- Hippulin
- Hydroxyestrone diacetate
- Lynestrenol
- Lynestrenol phenylpropionate
- Mestranol
- Methylestradiol
- Moxestrol
- Mytatrienediol
- Nilestriol
- Norethisterone
- Noretynodrel
- Orestrate
- Pentolame
- Prodiame
- Prolame
- Promestriene
- RU-16117
- Quinestradol
- Quinestrol
- Tibolone
- Xenoestrogens:
Anise
-related (e.g.,
anethole
,
anol
,
dianethole
,
dianol
,
photoanethole
)
- Chalconoids
(e.g.,
isoliquiritigenin
,
phloretin
,
phlorizin (phloridzin)
,
wedelolactone
)
- Coumestans
(e.g.,
coumestrol
,
psoralidin
)
- Flavonoids
(incl.
7,8-DHF
,
8-prenylnaringenin
,
apigenin
,
baicalein
,
baicalin
,
biochanin A
,
calycosin
,
catechin
,
daidzein
,
daidzin
,
ECG
,
EGCG
,
epicatechin
,
equol
,
formononetin
,
glabrene
,
glabridin
,
genistein
,
genistin
,
glycitein
,
kaempferol
,
liquiritigenin
,
mirificin
,
myricetin
,
naringenin
,
penduletin
,
pinocembrin
,
prunetin
,
puerarin
,
quercetin
,
tectoridin
,
tectorigenin
)
- Lavender oil
- Lignans
(e.g.,
enterodiol
,
enterolactone
,
nyasol (
cis
-hinokiresinol)
)
- Metalloestrogens
(e.g.,
cadmium
)
- Pesticides
(e.g.,
alternariol
,
dieldrin
,
endosulfan
,
fenarimol
,
HPTE
,
methiocarb
,
methoxychlor
,
triclocarban
,
triclosan
)
- Phytosteroids
(e.g.,
digitoxin
(
digitalis
),
diosgenin
,
guggulsterone
)
- Phytosterols
(e.g.,
β-sitosterol
,
campesterol
,
stigmasterol
)
- Resorcylic acid lactones
(e.g.,
zearalanone
,
α-zearalenol
,
β-zearalenol
,
zearalenone
,
zeranol (α-zearalanol)
,
taleranol (teranol, β-zearalanol)
)
- Steroid
-like (e.g.,
deoxymiroestrol
,
miroestrol
)
- Stilbenoids
(e.g.,
resveratrol
,
rhaponticin
)
- Synthetic xenoestrogens
(e.g.,
alkylphenols
,
bisphenols
(e.g.,
BPA
,
BPF
,
BPS
),
DDT
,
parabens
,
PBBs
,
PHBA
,
phthalates
,
PCBs
)
- Others (e.g.,
agnuside
,
rotundifuran
)
|
|---|
Mixed
(
SERMs
Tooltip Selective estrogen receptor modulators
)
| |
|---|
| Antagonists
|
- Coregulator-binding modulators:
ERX-11
|
|---|
|
|---|
| GPER
Tooltip G protein-coupled estrogen receptor
| | Agonists
| |
|---|
| Antagonists
| |
|---|
| Unknown
| |
|---|
|
|---|
|