Atmospheric chemistry
is a branch of
science
in which the
chemistry
of the
Earth's atmosphere
and that of other planets is studied. It is a
multidisciplinary
field of research and draws on
environmental chemistry
,
physics
,
meteorology
,
computer modeling
,
oceanography
,
geology
and
volcanology
and other disciplines. Research is connected with other areas of study such as
climatology
.
Studying the
atmosphere
includes studying the interactions between the atmosphere and living organisms. The composition of the
Earth
's atmosphere changes as result of natural processes such as
volcano
emissions,
lightning
and bombardment by solar particles from the
Sun
's
corona
. It has also been changed by human activity. Some of these changes are harmful to human health,
crops
and
ecosystems
. Examples of problems include
acid rain
,
ozone depletion
,
photochemical
smog
,
greenhouse gases
and
global warming
. Atmospheric chemists study the causes of these problems. Atmospheric chemists offer
theories
about these problems, then test the theories and possible solutions. Atmospheric chemists also note the effects of changes in government policy.
 Make-up of Earth's atmosphere. Water vapor is not included as it changes a lot over time. Each tiny cube (such as the one representing
krypton
) has one millionth of the volume of the entire block.
Make-up of Earth's atmosphere. Water vapor is not included as it changes a lot over time. Each tiny cube (such as the one representing
krypton
) has one millionth of the volume of the entire block.
Data is from
NASA Langley
.
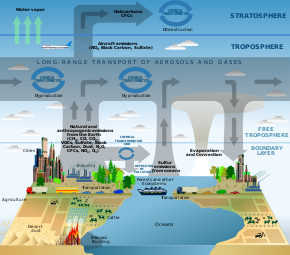 Schematic of chemical and transport processes related to atmospheric composition.
Schematic of chemical and transport processes related to atmospheric composition.
Notes: the
concentration
of CO
2
and CH
4
vary by
season
and location. The mean
molecular mass
of
air
is 28.97 g/mol.
The ancient
Greeks
regarded air as one of the four elements. The first scientific studies of atmospheric composition began in the 18th century. Chemists such as
Joseph Priestley
,
Antoine Lavoisier
and
Henry Cavendish
made the first measurements of the composition of the atmosphere.
In the late 19th and early 20th centuries interest shifted towards trace constituents with very small concentrations. One important discovery for atmospheric chemistry was the discovery of
ozone
by
Christian Friedrich Schonbein
in 1840.
The concentrations of trace gases in the atmosphere have changed over time and so have the chemical processes which make and destroy compounds in the air. Two important examples of this were the explanation by
Sydney Chapman
and
Gordon Dobson
of how the
ozone layer
is created and maintained, and the explanation of photochemical smog by
Arie Jan Haagen-Smit
. Further studies on ozone issues led to the 1995
Nobel Prize in Chemistry
award shared between
Paul Crutzen
,
Mario Molina
and
Frank Sherwood Rowland
.
[2]
In the 21st century the focus is now
shifting
again. Atmospheric chemistry is increasingly studied as one part of the
Earth system
. Before, scientists focused on atmospheric chemistry in isolation. Now, scientists study atmospheric chemistry as one part of a single system with the rest of the
atmosphere
,
biosphere
and
geosphere
. A reason for this is the links between chemistry and
climate
. For example, changing climate and the recovery of the ozone hole affect each other. Also, the composition of the atmosphere interact with the oceans and terrestrial
ecosystems
.
Observations, lab measurements and modeling are the three central elements in atmospheric chemistry. All three methods are used together. For example, observations may tell that more of a chemical compound exists than previously thought possible. This will stimulate new modelling and laboratory studies which will increase scientific understanding to a point where the observations can be explained.
Observations of atmospheric chemistry are important. Scientist record data about the chemical composition of air over time to watch for any changes. One example of this is the
Keeling Curve
- a series of measurements from 1958 to today which show a steady rise in of the concentration of
carbon dioxide
. Observations of atmospheric chemistry are made in observatories such as that on
Mauna Loa
and on mobile platforms such as aircraft, ships and
balloons
. Observations of atmospheric composition are increasingly made by
satellites
giving a global picture of
air pollution
and chemistry.
[3]
Surface observations have the advantage that they provide long term records at high time resolution but provide data from a limited vertical and horizontal space. Some surface based instruments such as
LIDAR
can provide concentration profiles of chemical compounds and
aerosol
but are still restricted in the horizontal region they cover. Many observations are shared on-line.
Measurements made in the laboratory are essential to our understanding of the sources and
sinks
of
pollutants
and compounds found in nature. Lab studies tell which gases react with each other and how fast they react. Scientists measure reactions in the gas phase, on surfaces and in water. Scientists also study
photochemistry
which quantifies how quickly molecules are split apart by sunlight and what the products are. Scientists also study
thermodynamic
data such as
Henry's law
coefficients.
- Brasseur, Guy P.; Orlando, John J.; Tyndall, Geoffrey S. (1999).
Atmospheric Chemistry and Global Change
. Oxford University Press.
ISBN
0-19-510521-4
.
- Finlayson-Pitts, Barbara J.; Pitts, James N., Jr. (2000).
Chemistry of the Upper and Lower Atmosphere
. Academic Press.
ISBN
0-12-257060-X
.
- Seinfeld, John H.; Pandis, Spyros N. (2006).
Atmospheric Chemistry and Physics: From Air Pollution to Climate Change
(2nd Ed.). John Wiley and Sons, Inc.
ISBN
0471828572
.
- Warneck, Peter (2000).
Chemistry of the Natural Atmosphere
(2nd Ed.). Academic Press.
ISBN
0-12-735632-0
.
- Wayne, Richard P. (2000).
Chemistry of Atmospheres
(3rd Ed.). Oxford University Press.
ISBN
0-19-850375-X
.