From Simple English Wikipedia, the free encyclopedia
| This article
does not have any
sources
.
You can help Wikipedia by finding
good
sources, and adding them.
|
In
chemistry
,
alcohol
is a general term which refers to many
organic compounds
used in industry and science as
reagents
,
solvents
, and
fuels
. Alcohols are
carbohydrates
which are made of an
alkyl
group
with one or more
hydroxyl
(-
O
H
) groups bound to its
carbon
atoms
. Alcohol is
colorless
, and also
transparent
.
There are two ways of naming alcohols: Common names, and IUPAC names.
- Common names often are made by taking the name of the
alkyl
group, and adding the word "alcohol". For example, "methyl alcohol" or "ethyl alcohol".
- IUPAC names are made by taking the name of the alkane chain, removing the last "e", and adding "ol". Examples of this are "methanol" and "ethanol".
The
hydroxyl
(OH) group makes alcohols
polar
. Alcohols are very weakly
acidic
. Most alcohols are highly flammable.
The simplest two alcohols are
methanol
(or
methyl
alcohol) and
ethanol
(or
ethyl
alcohol), which have the following structures:
IUPAC nomenclature
is used when describing more complex alcohols.
In common usage, "alcohol" often means ethanol or "grain alcohol". (See also:
alcoholic proof
).
Other commonly used alcohols include:
- Isopropyl alcohol
(
sec
-propyl alcohol, propan-2-ol, 2-propanol) H
3
C-CH(OH)-CH
3
, or "rubbing alcohol"
- ethylene glycol
(ethane-1,2-diol) HO-CH
2
-CH
2
-OH, which is the main substance in
antifreeze
- glycerin
(or
glycerol
, propane-1,2,3-triol) HO-CH
2
-CH(OH)-CH
2
-OH bound in natural
fats
and oils, which are
triglycerides
(triacylglycerols)
- phenol
, an alcohol where the hydroxyl group is bound to a
benzene ring
.
Possible long term side effects
[
change
|
change source
]
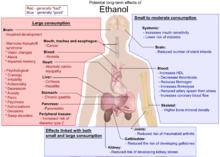 Possible long term side effects in humans
Possible long term side effects in humans
|
|---|
Hydrocarbons
(only C and H)
| |
|---|
Only
carbon
,
hydrogen
,
and
oxygen
(only C, H and O)
| |
|---|
Only one
element,
not being
carbon,
hydrogen,
or oxygen
(one element,
not C, H or O)
| |
|---|
| Other
| |
|---|