Series of chemical elements
In chemistry, a
transition metal
(or
transition element
) is a
chemical element
in the
d-block
of the
periodic table
(groups 3 to 12), though the elements of
group 12
(and less often
group 3
) are sometimes excluded. The
lanthanide
and
actinide
elements (the
f-block
) are called
inner transition metals
and are sometimes considered to be transition metals as well.
Since they are metals, they are lustrous and have good electrical and thermal conductivity. Most (with the exception of
group 11
and group 12) are hard and strong, and have high melting and boiling temperatures. They form compounds in any of two or more different
oxidation states
and bind to a variety of
ligands
to form
coordination complexes
that are often coloured. They form many useful
alloys
and are often employed as
catalysts
in elemental form or in compounds such as coordination complexes and
oxides
. Most are strongly
paramagnetic
because of their
unpaired d electrons
, as are many of their compounds. All of the elements that are
ferromagnetic
near room temperature are transition metals (
iron
,
cobalt
and
nickel
) or inner transition metals (
gadolinium
).
English chemist
Charles Rugeley Bury
(1890?1968) first used the word
transition
in this context in 1921, when he referred to a
transition series of elements
during the change of an inner layer of electrons (for example
n
= 3 in the 4th row of the periodic table) from a stable group of 8 to one of 18, or from 18 to 32.
[1]
[2]
[3]
These elements are now known as the d-block.
 The first row of transition metals, in order
The first row of transition metals, in order
Definition and classification
The 2011
IUPAC
Principles of Chemical Nomenclature
describe a "transition metal" as any element in groups 3 to 12 on the
periodic table
.
[4]
This corresponds exactly to the
d-block
elements, and many scientists use this definition.
[5]
[6]
In actual practice, the
f-block
lanthanide
and
actinide
series are called "inner transition metals". The 2005
Red Book
allows for the group 12 elements to be excluded, but not the 2011
Principles
.
[7]
The IUPAC
Gold Book
[8]
defines a transition metal as "an
element
whose atom has a partially filled
d
sub-shell, or which can give rise to
cations
with an incomplete d sub-shell", but this definition is taken from an old edition of the
Red Book
and is no longer present in the current edition.
[7]
In the d-block, the atoms of the elements have between zero and ten d electrons.
Published texts and periodic tables show
variation regarding the heavier members of group 3
.
[9]
The common placement of
lanthanum
and
actinium
in these positions is not supported by physical, chemical, and electronic
evidence
,
[10]
[11]
[12]
which overwhelmingly favour putting
lutetium
and
lawrencium
in those places.
[13]
[14]
Some authors prefer to leave the spaces below
yttrium
blank as a third option, but there is confusion on whether this format implies that group 3 contains only
scandium
and yttrium, or if it also contains all the lanthanides and actinides;
[15]
[16]
[17]
[18]
[19]
additionally, it creates a 15-element-wide f-block, when
quantum mechanics
dictates that the f-block should only be 14 elements wide.
[15]
The form with lutetium and lawrencium in group 3 is supported by a 1988
IUPAC
report on physical, chemical, and electronic grounds,
[20]
and again by a 2021 IUPAC preliminary report as it is the only form that allows simultaneous (1) preservation of the sequence of increasing atomic numbers, (2) a 14-element-wide f-block, and (3) avoidance of the split in the d-block.
[15]
Argumentation can still be found in the contemporary literature purporting to defend the form with lanthanum and actinium in group 3, but many authors consider it to be logically inconsistent (a particular point of contention being the differing treatment of
actinium
and
thorium
, which both can use 5f as a
valence
orbital
but have no 5f occupancy as single atoms);
[14]
[21]
[22]
the majority of investigators considering the problem agree with the updated form with lutetium and lawrencium.
[14]
The group 12 elements
zinc
,
cadmium
, and
mercury
are sometimes excluded from the transition metals.
[1]
This is because they have the
electronic configuration
[ ]d
10
s
2
, where the d shell is complete,
[23]
and they still have a complete d shell in all their known
oxidation states
. The group 12 elements Zn, Cd and Hg may therefore, under certain criteria, be classed as
post-transition metals
in this case. However, it is often convenient to include these elements in a discussion of the transition elements. For example, when discussing the
crystal field stabilization energy
of first-row transition elements, it is convenient to also include the elements
calcium
and zinc, as both
Ca
2+
and
Zn
2+
have a value of zero, against which the value for other transition metal ions may be compared. Another example occurs in the
Irving?Williams series
of stability constants of complexes. Moreover, Zn, Cd, and Hg can use their d orbitals for
bonding
even though they are not known in oxidation states that would formally require breaking open the d-subshell, which sets them apart from the p-block elements.
[24]
[25]
[26]
The recent (though disputed and so far not reproduced independently) synthesis of
mercury(IV) fluoride
(
HgF
4
) has been taken by some to reinforce the view that the group 12 elements should be considered transition metals,
[27]
but some authors still consider this compound to be exceptional.
[28]
Copernicium
is expected to be able to use its d electrons for chemistry as its 6d
subshell
is destabilised by strong
relativistic effects
due to its very high atomic number, and as such is expected to have transition-metal-like behaviour and show higher oxidation states than +2 (which are not definitely known for the lighter group 12 elements). Even in bare dications, Cn
2+
is predicted to be 6d
8
7s
2
, unlike Hg
2+
which is 5d
10
6s
0
.
Although
meitnerium
,
darmstadtium
, and
roentgenium
are within the d-block and are expected to behave as transition metals analogous to their lighter
congeners
iridium
,
platinum
, and
gold
, this has not yet been experimentally confirmed. Whether
copernicium
behaves more like
mercury
or has properties more similar to those of the
noble gas
radon
is not clear. Relative inertness of Cn would come from the relativistically expanded 7s?7p
1/2
energy gap, which is already adumbrated in the 6s?6p
1/2
gap for Hg, weakening metallic bonding and causing its well-known low melting and boiling points.
Transition metals with lower or higher group numbers are described as 'earlier' or 'later', respectively. When described in a two-way classification scheme, early transition metals are on the left side of the d-block from group 3 to group 7. Late transition metals are on the right side of the d-block, from group 8 to 11 (or 12, if they are counted as transition metals). In an alternative three-way scheme, groups 3, 4, and 5 are classified as early transition metals, 6, 7, and 8 are classified as middle transition metals, and 9, 10, and 11 (and sometimes group 12) are classified as late transition metals.
The heavy group 2 elements
calcium
,
strontium
, and
barium
do not have filled d-orbitals as single atoms, but are known to have d-orbital bonding participation in some
compounds
, and for that reason have been called "honorary" transition metals.
[29]
Probably the same is true of
radium
.
[30]
The f-block elements La?Yb and Ac?No have chemical activity of the (n?1)d shell, but importantly also have chemical activity of the (n?2)f shell that is absent in d-block elements. Hence they are often treated separately as inner transition elements.
Electronic configuration
The general electronic configuration of the d-block atoms is [noble gas](
n
? 1)d
0?10
n
s
0?2
n
p
0?1
. Here "[noble gas]" is the electronic configuration of the last
noble gas
preceding the atom in question, and
n
is the highest
principal quantum number
of an occupied orbital in that atom. For example, Ti (
Z
= 22) is in period 4 so that
n
= 4, the first 18 electrons have the same configuration of Ar at the end of period 3, and the overall configuration is [Ar]3d
2
4s
2
. The period 6 and 7 transition metals also add core (
n
? 2)f
14
electrons, which are omitted from the tables below. The p orbitals are almost never filled in free atoms (the one exception being lawrencium due to relativistic effects that become important at such high
Z
), but they can contribute to the chemical bonding in transition metal compounds.
The
Madelung rule
predicts that the inner d orbital is filled after the
valence-shell
s orbital. The typical
electronic structure
of transition metal atoms is then written as [noble gas]
n
s
2
(
n
? 1)d
m
. This rule is approximate, but holds for most of the transition metals. Even when it fails for the neutral ground state, it accurately describes a low-lying excited state.
The d subshell is the next-to-last subshell and is denoted as (
n
? 1)d subshell. The number of s electrons in the outermost s subshell is generally one or two except
palladium
(Pd), with no electron in that s sub shell in its ground state. The s subshell in the valence shell is represented as the
n
s subshell, e.g. 4s. In the periodic table, the transition metals are present in ten groups (3 to 12).
The elements in group 3 have an
n
s
2
(
n
? 1)d
1
configuration, except for
lawrencium
(Lr): its 7s
2
7p
1
configuration exceptionally does not fill the 6d orbitals at all. The first transition series is present in the 4th period, and starts after Ca (
Z
= 20) of group 2 with the configuration [Ar]4s
2
, or
scandium
(Sc), the first element of group 3 with atomic number
Z
= 21 and configuration [Ar]4s
2
3d
1
, depending on the definition used. As we move from left to right, electrons are added to the same d subshell till it is complete. Since the electrons added fill the (
n
? 1)d orbitals, the properties of the d-block elements are quite different from those of s and p block elements in which the filling occurs either in s or in p orbitals of the valence shell.
The electronic configuration of the individual elements present in all the d-block series are given below:
[31]
First (3d) d-block Series (Sc?Zn)
| Group
|
3
|
4
|
5
|
6
|
7
|
8
|
9
|
10
|
11
|
12
|
| Atomic number
|
21
|
22
|
23
|
24
|
25
|
26
|
27
|
28
|
29
|
30
|
| Element
|
Sc
|
Ti
|
V
|
Cr
|
Mn
|
Fe
|
Co
|
Ni
|
Cu
|
Zn
|
Electron
configuration
|
3d
1
4s
2
|
3d
2
4s
2
|
3d
3
4s
2
|
3d
5
4s
1
|
3d
5
4s
2
|
3d
6
4s
2
|
3d
7
4s
2
|
3d
8
4s
2
|
3d
10
4s
1
|
3d
10
4s
2
|
Second (4d) d-block Series (Y?Cd)
| Atomic number
|
39
|
40
|
41
|
42
|
43
|
44
|
45
|
46
|
47
|
48
|
| Element
|
Y
|
Zr
|
Nb
|
Mo
|
Tc
|
Ru
|
Rh
|
Pd
|
Ag
|
Cd
|
Electron
configuration
|
4d
1
5s
2
|
4d
2
5s
2
|
4d
4
5s
1
|
4d
5
5s
1
|
4d
5
5s
2
|
4d
7
5s
1
|
4d
8
5s
1
|
4d
10
5s
0
|
4d
10
5s
1
|
4d
10
5s
2
|
Third (5d) d-block Series (Lu?Hg)
| Atomic number
|
71
|
72
|
73
|
74
|
75
|
76
|
77
|
78
|
79
|
80
|
| Element
|
Lu
|
Hf
|
Ta
|
W
|
Re
|
Os
|
Ir
|
Pt
|
Au
|
Hg
|
Electron
configuration
|
5d
1
6s
2
|
5d
2
6s
2
|
5d
3
6s
2
|
5d
4
6s
2
|
5d
5
6s
2
|
5d
6
6s
2
|
5d
7
6s
2
|
5d
9
6s
1
|
5d
10
6s
1
|
5d
10
6s
2
|
Fourth (6d) d-block Series (Lr?Cn)
(Configurations predicted for Mt?Cn)
| Atomic number
|
103
|
104
|
105
|
106
|
107
|
108
|
109
|
110
|
111
|
112
|
| Element
|
Lr
|
Rf
|
Db
|
Sg
|
Bh
|
Hs
|
Mt
|
Ds
|
Rg
|
Cn
|
Electron
configuration
|
7s
2
7p
1
|
6d
2
7s
2
|
6d
3
7s
2
|
6d
4
7s
2
|
6d
5
7s
2
|
6d
6
7s
2
|
6d
7
7s
2
|
6d
8
7s
2
|
6d
9
7s
2
|
6d
10
7s
2
|
A careful look at the electronic configuration of the elements reveals that there are certain exceptions to the
Madelung rule
. For Cr as an example the rule predicts the configuration 3d
4
4s
2
, but the observed atomic spectra show that the real
ground state
is 3d
5
4s
1
. To explain such exceptions, it is necessary to consider the effects of increasing
nuclear charge
on the orbital energies, as well as the electron?electron interactions including both
Coulomb repulsion
and
exchange energy
.
[31]
The exceptions are in any case not very relevant for chemistry because the energy difference between them and the expected configuration is always quite low.
[32]
The (
n
? 1)d orbitals that are involved in the transition metals are very significant because they influence such properties as magnetic character, variable oxidation states, formation of coloured compounds etc. The valence s and p orbitals (
n
s and
n
p) have very little contribution in this regard since they hardly change in the moving from left to the right in a transition series.
In transition metals, there are greater horizontal similarities in the properties of the elements in a period in comparison to the periods in which the d orbitals are not involved. This is because in a transition series, the valence shell electronic configuration of the elements do not change. However, there are some group similarities as well.
Characteristic properties
There are a number of properties shared by the transition elements that are not found in other elements, which results from the partially filled d shell. These include
- the formation of compounds whose colour is due to d?d electronic transitions
- the formation of compounds in many oxidation states, due to the relatively low energy gap between different possible oxidation states
[33]
- the formation of many
paramagnetic
compounds due to the presence of unpaired d electrons. A few compounds of main-group elements are also paramagnetic (e.g.
nitric oxide
,
oxygen
)
Most transition metals can be bound to a variety of
ligands
, allowing for a wide variety of transition metal complexes.
[34]
Coloured compounds
 From left to right, aqueous solutions of:
Co(NO
From left to right, aqueous solutions of:
Co(NO
3
)
2
(red);
K
2
Cr
2
O
7
(orange);
K
2
CrO
4
(yellow);
NiCl
2
(turquoise);
CuSO
4
(blue);
KMnO
4
(purple).
Colour in transition-series metal compounds is generally due to electronic transitions of two principal types.
- charge transfer
transitions. An electron may jump from a predominantly
ligand
orbital
to a predominantly metal orbital, giving rise to a ligand-to-metal charge-transfer (LMCT) transition. These can most easily occur when the metal is in a high oxidation state. For example, the colour of
chromate
,
dichromate
and
permanganate
ions is due to LMCT transitions. Another example is that
mercuric iodide
, HgI
2
, is red because of a LMCT transition.
A metal-to-ligand charge transfer (MLCT) transition will be most likely when the metal is in a low oxidation state and the ligand is easily reduced.
In general charge transfer transitions result in more intense colours than d?d transitions.
- d?d transitions. An electron jumps from one
d orbital
to another. In complexes of the transition metals the d orbitals do not all have the same energy. The pattern of splitting of the d orbitals can be calculated using
crystal field
theory. The extent of the splitting depends on the particular metal, its oxidation state and the nature of the ligands. The actual energy levels are shown on
Tanabe?Sugano diagrams
.
In
centrosymmetric
complexes, such as octahedral complexes, d?d transitions are forbidden by the
Laporte rule
and only occur because of
vibronic coupling
in which a
molecular vibration
occurs together with a d?d transition. Tetrahedral complexes have somewhat more intense colour because mixing d and p orbitals is possible when there is no centre of symmetry, so transitions are not pure d?d transitions. The
molar absorptivity
(ε) of bands caused by d?d transitions are relatively low, roughly in the range 5-500 M
?1
cm
?1
(where
M
= mol dm
?3
).
[35]
Some d?d transitions are
spin forbidden
. An example occurs in octahedral, high-spin complexes of
manganese
(II),
which has a d
5
configuration in which all five electrons have parallel spins; the colour of such complexes is much weaker than in complexes with spin-allowed transitions. Many compounds of manganese(II) appear almost colourless. The
spectrum of
[Mn(H
2
O)
6
]
2+
shows a maximum molar absorptivity of about 0.04 M
?1
cm
?1
in the
visible spectrum
.
Oxidation states
A characteristic of transition metals is that they exhibit two or more
oxidation states
, usually differing by one. For example, compounds of
vanadium
are known in all oxidation states between ?1, such as
[V(CO)
6
]
?
, and +5, such as
VO
3?
4
.
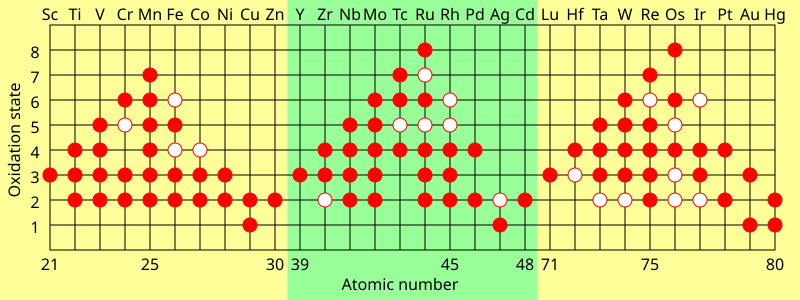 Oxidation states of the transition metals. The solid dots show common oxidation states, and the hollow dots show possible but unlikely states.
Oxidation states of the transition metals. The solid dots show common oxidation states, and the hollow dots show possible but unlikely states.
Main-group elements
in groups 13 to 18 also exhibit multiple oxidation states. The "common" oxidation states of these elements typically differ by two instead of one. For example, compounds of
gallium
in oxidation states +1 and +3 exist in which there is a single gallium atom. Compounds of Ga(II) would have an unpaired electron and would behave as a
free radical
and generally be destroyed rapidly, but some stable radicals of Ga(II) are known.
[36]
Gallium also has a formal oxidation state of +2 in dimeric compounds, such as
[Ga
2
Cl
6
]
2?
, which contain a Ga-Ga bond formed from the unpaired electron on each Ga atom.
[37]
Thus the main difference in oxidation states, between transition elements and other elements is that oxidation states are known in which there is a single atom of the element and one or more unpaired electrons.
The maximum oxidation state in the first row transition metals is equal to the number of valence electrons from
titanium
(+4) up to
manganese
(+7), but decreases in the later elements. In the second row, the maximum occurs with
ruthenium
(+8), and in the third row, the maximum occurs with
iridium
(+9). In compounds such as
[MnO
4
]
?
and
OsO
4
, the elements achieve a stable configuration by
covalent bonding
.
The lowest oxidation states are exhibited in
metal carbonyl
complexes such as
Cr(CO)
6
(oxidation state zero) and
[Fe(CO)
4
]
2?
(oxidation state ?2) in which the
18-electron rule
is obeyed. These complexes are also covalent.
Ionic compounds are mostly formed with oxidation states +2 and +3. In aqueous solution, the ions are hydrated by (usually) six water molecules arranged octahedrally.
Magnetism
Transition metal compounds are
paramagnetic
when they have one or more unpaired d electrons.
[38]
In octahedral complexes with between four and seven d electrons both
high spin
and
low spin
states are possible. Tetrahedral transition metal complexes such as
[FeCl
4
]
2?
are
high spin
because the crystal field splitting is small so that the energy to be gained by virtue of the electrons being in lower energy orbitals is always less than the energy needed to pair up the spins. Some compounds are
diamagnetic
. These include octahedral, low-spin, d
6
and square-planar d
8
complexes. In these cases,
crystal field
splitting is such that all the electrons are paired up.
Ferromagnetism
occurs when individual atoms are paramagnetic and the spin vectors are aligned parallel to each other in a crystalline material. Metallic iron and the alloy
alnico
are examples of ferromagnetic materials involving transition metals.
Antiferromagnetism
is another example of a magnetic property arising from a particular alignment of individual spins in the solid state.
Catalytic properties
The transition metals and their compounds are known for their homogeneous and heterogeneous
catalytic
activity. This activity is ascribed to their ability to adopt multiple oxidation states and to form complexes.
Vanadium
(V) oxide (in the
contact process
), finely divided
iron
(in the
Haber process
), and
nickel
(in
catalytic hydrogenation
) are some of the examples. Catalysts at a solid surface (
nanomaterial-based catalysts
) involve the formation of bonds between reactant molecules and atoms of the surface of the catalyst (first row transition metals utilize 3d and 4s electrons for bonding). This has the effect of increasing the concentration of the reactants at the catalyst surface and also weakening of the bonds in the reacting molecules (the activation energy is lowered). Also because the transition metal ions can change their oxidation states, they become more effective as
catalysts
.
An interesting type of catalysis occurs when the products of a reaction catalyse the reaction producing more catalyst (
autocatalysis
). One example is the reaction of
oxalic acid
with acidified
potassium permanganate
(or manganate (VII)).
[39]
Once a little Mn
2+
has been produced, it can react with MnO
4
?
forming Mn
3+
. This then reacts with C
2
O
4
?
ions forming Mn
2+
again.
Physical properties
As implied by the name, all transition metals are
metals
and thus conductors of electricity.
In general, transition metals possess a high
density
and high
melting points
and
boiling points
. These properties are due to
metallic bonding
by delocalized d electrons, leading to
cohesion
which increases with the number of shared electrons. However the group 12 metals have much lower melting and boiling points since their full d subshells prevent d?d bonding, which again tends to differentiate them from the accepted transition metals. Mercury has a melting point of ?38.83 °C (?37.89 °F) and is a liquid at room temperature.
See also
References
- ^
a
b
Jensen, William B. (2003).
"The Place of Zinc, Cadmium, and Mercury in the Periodic Table"
(PDF)
.
Journal of Chemical Education
.
80
(8): 952?961.
Bibcode
:
2003JChEd..80..952J
.
doi
:
10.1021/ed080p952
.
- ^
Bury, C. R. (1921).
"Langmuir's theory of the arrangement of electrons in atoms and molecules"
.
J. Am. Chem. Soc
.
43
(7): 1602?1609.
doi
:
10.1021/ja01440a023
.
- ^
Bury, Charles Rugeley
. Encyclopedia.com Complete dictionary of scientific biography (2008).
- ^
Leigh, G. J., ed. (2011).
Principles of Chemical Nomenclature
(PDF)
. The Royal Society of Chemistry. p. 9.
ISBN
978-1-84973-007-5
.
- ^
Petrucci, Ralph H.; Harwood, William S.; Herring, F. Geoffrey (2002).
General chemistry: principles and modern applications
(8th ed.). Upper Saddle River, N.J: Prentice Hall. pp.
341?342
.
ISBN
978-0-13-014329-7
.
LCCN
2001032331
.
OCLC
46872308
.
- ^
Housecroft, C. E. and Sharpe, A. G. (2005)
Inorganic Chemistry
, 2nd ed, Pearson Prentice-Hall, pp. 20?21.
- ^
a
b
Connelly, N.G.; Damhus, T.; Hartshorn, R.M.; Hutton, A.T., eds. (2005).
Nomenclature of Inorganic Chemistry
(PDF)
.
RSC
?
IUPAC
.
ISBN
0-85404-438-8
.
- ^
IUPAC
,
Compendium of Chemical Terminology
, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
transition element
".
doi
:
10.1351/goldbook.T06456
- ^
Scerri, Eric R. (2020).
The Periodic Table: Its Story and Its Significance
. New York, NY.
ISBN
978-0-19-091436-3
.
OCLC
1096234740
.
{{
cite book
}}
: CS1 maint: location missing publisher (
link
)
- ^
L. D. Landau
,
E. M. Lifshitz
(1958).
Quantum Mechanics: Non-Relativistic Theory
. Vol. 3 (1st ed.).
Pergamon Press
. pp. 256?7.
- ^
Wittig, Jorg (1973). "The pressure variable in solid state physics: What about 4f-band superconductors?". In H. J. Queisser (ed.).
Festkorper Probleme: Plenary Lectures of the Divisions Semiconductor Physics, Surface Physics, Low Temperature Physics, High Polymers, Thermodynamics and Statistical Mechanics, of the German Physical Society, Munster, March 19?24, 1973
. Advances in Solid State Physics. Vol. 13. Berlin, Heidelberg: Springer. pp. 375?396.
doi
:
10.1007/BFb0108579
.
ISBN
978-3-528-08019-8
.
- ^
Matthias, B. T. (1969). "Systematics of Super Conductivity". In Wallace, P. R. (ed.).
Superconductivity
. Vol. 1. Gordon and Breach. pp. 225?294.
ISBN
9780677138107
.
- ^
William B. Jensen (1982). "The Positions of Lanthanum (Actinium) and Lutetium (Lawrencium) in the Periodic Table".
J. Chem. Educ
.
59
(8): 634?636.
Bibcode
:
1982JChEd..59..634J
.
doi
:
10.1021/ed059p634
.
- ^
a
b
c
Jensen, William B. (2015).
"The positions of lanthanum (actinium) and lutetium (lawrencium) in the periodic table: an update"
.
Foundations of Chemistry
.
17
: 23?31.
doi
:
10.1007/s10698-015-9216-1
.
S2CID
98624395
.
Archived
from the original on 30 January 2021
. Retrieved
28 January
2021
.
- ^
a
b
c
Scerri, Eric (18 January 2021).
"Provisional Report on Discussions on Group 3 of the Periodic Table"
(PDF)
.
Chemistry International
.
43
(1): 31?34.
doi
:
10.1515/ci-2021-0115
.
S2CID
231694898
.
Archived
(PDF)
from the original on 13 April 2021
. Retrieved
9 April
2021
.
- ^
Thyssen, P.; Binnemans, K. (2011). "Accommodation of the Rare Earths in the Periodic Table: A Historical Analysis". In Gschneidner, K. A. Jr.; Bunzli, J-C.G; Vecharsky, Bunzli (eds.).
Handbook on the Physics and Chemistry of Rare Earths
. Vol. 41. Amsterdam: Elsevier. pp. 1?94.
doi
:
10.1016/B978-0-444-53590-0.00001-7
.
ISBN
978-0-444-53590-0
.
- ^
Barber, Robert C.; Karol, Paul J; Nakahara, Hiromichi; Vardaci, Emanuele; Vogt, Erich W. (2011).
"Discovery of the elements with atomic numbers greater than or equal to 113 (IUPAC Technical Report)"
.
Pure Appl. Chem
.
83
(7): 1485.
doi
:
10.1351/PAC-REP-10-05-01
.
- ^
Karol, Paul J.; Barber, Robert C.; Sherrill, Bradley M.; Vardaci, Emanuele; Yamazaki, Toshimitsu (22 December 2015).
"Discovery of the elements with atomic numbers Z = 113, 115 and 117 (IUPAC Technical Report)"
.
Pure Appl. Chem
.
88
(1?2): 139?153.
doi
:
10.1515/pac-2015-0502
.
- ^
Pyykko, Pekka (2019).
"An essay on periodic tables"
(PDF)
.
Pure and Applied Chemistry
.
91
(12): 1959?1967.
doi
:
10.1515/pac-2019-0801
.
S2CID
203944816
. Retrieved
27 November
2022
.
- ^
Fluck, E. (1988).
"New Notations in the Periodic Table"
(PDF)
.
Pure Appl. Chem.
60
(3): 431?436.
doi
:
10.1351/pac198860030431
.
S2CID
96704008
.
Archived
(PDF)
from the original on 25 March 2012
. Retrieved
24 March
2012
.
- ^
Scerri, Eric (2009).
"Which Elements Belong in Group 3?"
.
Journal of Chemical Education
.
86
(10): 1188.
Bibcode
:
2009JChEd..86.1188S
.
doi
:
10.1021/ed086p1188
. Retrieved
1 January
2023
.
- ^
Chemey, Alexander T.; Albrecht-Schmitt, Thomas E. (2019). "Evolution of the periodic table through the synthesis of new elements".
Radiochimica Acta
.
107
(9?11): 771?801.
doi
:
10.1515/ract-2018-3082
.
S2CID
104470619
.
- ^
Cotton, F. Albert; Wilkinson, G.; Murillo, C. A. (1999).
Advanced Inorganic Chemistry
(6th ed.). New York: Wiley,
ISBN
978-0-471-19957-1
.
- ^
Tossell, J.A. (1 November 1977). "Theoretical studies of valence orbital binding energies in solid zinc sulfide, zinc oxide, and zinc fluoride".
Inorganic Chemistry
.
16
(11): 2944?2949.
doi
:
10.1021/ic50177a056
.
- ^
Farberovich, O. V.; Kurganskii, S. I.; Domashevskaya, E. P. (1980). "Problems of the OPW Method. II. Calculation of the Band Structure of ZnS and CdS".
Physica Status Solidi B
.
97
(2): 631?640.
Bibcode
:
1980PSSBR..97..631F
.
doi
:
10.1002/pssb.2220970230
.
- ^
Singh, Prabhakar P. (1994). "Relativistic effects in mercury: Atom, clusters, and bulk".
Physical Review B
.
49
(7): 4954?4958.
Bibcode
:
1994PhRvB..49.4954S
.
doi
:
10.1103/PhysRevB.49.4954
.
PMID
10011429
.
- ^
Wang, Xuefang; Andrews, Lester; Riedel, Sebastian; Kaupp, Martin (2007). "Mercury Is a Transition Metal: The First Experimental Evidence for HgF
4
".
Angew. Chem. Int. Ed
.
46
(44): 8371?8375.
doi
:
10.1002/anie.200703710
.
PMID
17899620
.
- ^
Jensen, William B. (2008).
"Is Mercury Now a Transition Element?"
.
J. Chem. Educ
.
85
(9): 1182?1183.
Bibcode
:
2008JChEd..85.1182J
.
doi
:
10.1021/ed085p1182
.
- ^
Fernandez, Israel; Holzmann, Nicole; Frenking, Gernot (2020).
"The Valence Orbitals of the Alkaline-Earth Atoms"
.
Chemistry: A European Journal
.
26
(62): 14194?14210.
doi
:
10.1002/chem.202002986
.
PMC
7702052
.
PMID
32666598
.
S2CID
220529532
.
- ^
Pyykko, Pekka; Desclaux, Jean-Paul (1979). "Relativity and the Periodic System of Elements".
Accounts of Chemical Research
.
12
(8): 276?281.
doi
:
10.1021/ar50140a002
.
- ^
a
b
Miessler, G. L. and Tarr, D. A. (1999)
Inorganic Chemistry
, 2nd edn, Prentice-Hall, p. 38-39
ISBN
978-0-13-841891-5
- ^
Jørgensen, Christian (1973). "The Loose Connection between Electron Configuration and the Chemical Behavior of the Heavy Elements (Transuranics)".
Angewandte Chemie International Edition
.
12
(1): 12?19.
doi
:
10.1002/anie.197300121
.
- ^
Matsumoto, Paul S (2005). "Trends in Ionization Energy of Transition-Metal Elements".
Journal of Chemical Education
.
82
(11): 1660.
Bibcode
:
2005JChEd..82.1660M
.
doi
:
10.1021/ed082p1660
.
- ^
Hogan, C. Michael (2010).
"Heavy metal"
in
Encyclopedia of Earth
. National Council for Science and the Environment. E. Monosson and C. Cleveland (eds.) Washington DC.
- ^
Orgel, L.E. (1966).
An Introduction to Transition-Metal Chemistry, Ligand field theory
(2nd. ed.). London: Methuen.
- ^
Protchenko, Andrey V.; Dange, Deepak; Harmer, Jeffrey R.; Tang, Christina Y.; Schwarz, Andrew D.; Kelly, Michael J.; Phillips, Nicholas; Tirfoin, Remi; Birjkumar, Krishna Hassomal; Jones, Cameron; Kaltsoyannis, Nikolas; Mountford, Philip; Aldridge, Simon (16 February 2014). "Stable GaX
2
, InX
2
and TlX
2
radicals".
Nature Chemistry
.
6
(4): 315?319.
Bibcode
:
2014NatCh...6..315P
.
doi
:
10.1038/nchem.1870
.
PMID
24651198
.
- ^
Greenwood, Norman N.
; Earnshaw, Alan (1997).
Chemistry of the Elements
(2nd ed.).
Butterworth-Heinemann
.
ISBN
978-0-08-037941-8
.
p. 240
- ^
Figgis, B.N.; Lewis, J. (1960). Lewis, J.; Wilkins, R.G. (eds.).
The Magnetochemistry of Complex Compounds
. Modern Coordination Chemistry. New York: Wiley Interscience. pp. 400?454.
- ^
Kovacs KA, Grof P, Burai L, Riedel M (2004). "Revising the Mechanism of the Permanganate/Oxalate Reaction".
J. Phys. Chem. A
.
108
(50): 11026?11031.
Bibcode
:
2004JPCA..10811026K
.
doi
:
10.1021/jp047061u
.