From Wikipedia, the free encyclopedia
Ratio of neutrons to protons in an atomic nucleus
The
neutron?proton ratio
(
N/Z ratio
or
nuclear ratio
) of an
atomic nucleus
is the
ratio
of its number of
neutrons
to its number of
protons
. Among stable nuclei and naturally occurring nuclei, this ratio generally increases with increasing atomic number.
[1]
This is because electrical repulsive forces between protons scale with distance differently than
strong nuclear force
attractions. In particular, most pairs of protons in large nuclei are not far enough apart, such that electrical repulsion dominates over the strong nuclear force, and thus proton density in stable larger nuclei must be lower than in stable smaller nuclei where more pairs of protons have appreciable short-range nuclear force attractions.
For many elements with atomic number
Z
small enough to occupy only the first three
nuclear shells
, that is up to that of
calcium
(
Z
= 20), there exists a stable isotope with
N
/
Z
ratio of one. The exceptions are
beryllium
(
N
/
Z
= 1.25) and every element with odd atomic number between 9 and 19 inclusive (though in those cases
N
=
Z
+ 1 always allows for stability).
Hydrogen-1
(
N
/
Z
ratio = 0) and
helium-3
(
N
/
Z
ratio = 0.5) are the only stable isotopes with neutron?proton ratio under one.
Uranium-238
has the highest
N
/
Z
ratio of any
primordial nuclide
at 1.587,
[2]
while
mercury-204
has the highest
N
/
Z
ratio of any known stable isotope at 1.55.
Radioactive decay
generally proceeds so as to change the
N
/
Z
ratio to increase stability. If the
N
/
Z
ratio is greater than 1,
alpha decay
increases the
N
/
Z
ratio, and hence provides a common pathway towards stability for decays involving large nuclei with too few neutrons.
Positron emission
and
electron capture
also increase the ratio, while
beta decay
decreases the ratio.
Nuclear waste
exists mainly because nuclear fuel has a higher stable
N
/
Z
ratio than its
fission products
.
Semi-empirical description
[
edit
]
For stable nuclei, the neutron-proton ratio is such that the
binding energy
is at a
local minimum
or close to a minimum.
From the liquid drop model, this bonding energy is approximated by empirical
Bethe?Weizsacker formula

Given a value of
 and ignoring the contributions of nucleon spin pairing (i.e. ignoring the
and ignoring the contributions of nucleon spin pairing (i.e. ignoring the
 term), the binding energy is a quadratic expression in
term), the binding energy is a quadratic expression in
 that is minimized when the neutron-proton ratio is
that is minimized when the neutron-proton ratio is
 .
.
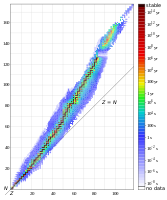 Isotope half-lives. Note that the plot for stable isotopes diverges from the line
Z
=
N
as the element number
Z
becomes larger
Isotope half-lives. Note that the plot for stable isotopes diverges from the line
Z
=
N
as the element number
Z
becomes larger
See also
[
edit
]
References
[
edit
]