Chemical compound (CH₃CH=CH₂)
For the unrelated compound sold as "Propolene", see
glucomannan
.
Propylene
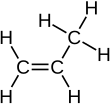
 Skeletal formula of propene Skeletal formula of propene
|
|
|

|
 Propylene Propylene
|
|
| Names
|
| Preferred IUPAC name
|
| Identifiers
|
|
|
|
|
|
|
|
|
1696878
|
| ChEBI
|
|
| ChEMBL
|
|
| ChemSpider
|
|
| ECHA InfoCard
|
100.003.693

|
| EC Number
|
|
|
|
852
|
| KEGG
|
|
|
|
|
| RTECS number
|
|
| UNII
|
|
| UN number
|
1077
In
Liquefied petroleum gas
:
1075
|
|
|
|
InChI=1S/C3H6/c1-3-2/h3H,1H2,2H3
 Y Y
Key: QQONPFPTGQHPMA-UHFFFAOYSA-N
 Y Y
InChI=1/C3H6/c1-3-2/h3H,1H2,2H3
Key: QQONPFPTGQHPMA-UHFFFAOYAA
|
|
|
| Properties
|
|
|
C
3
H
6
|
| Molar mass
|
42.081
g·mol
?1
|
| Appearance
|
Colorless gas
|
| Density
|
1.81 kg/m
3
, gas (1.013 bar, 15 °C)
1.745 kg/m
3
, gas (1.013 bar, 25 °C)
613.9 kg/m
3
, liquid
|
| Melting point
|
?185.2 °C (?301.4 °F; 88.0 K)
|
| Boiling point
|
?47.6 °C (?53.7 °F; 225.6 K)
|
|
|
0.61 g/m
3
|
|
|
-31.5·10
?6
cm
3
/mol
|
| Viscosity
|
8.34
μPa·s
at 16.7 °C
|
| Structure
|
|
|
0.366
D
(gas)
|
| Hazards
|
| GHS
labelling
:
[2]
|
|
|
 
|
|
|
Danger
|
|
|
H220
|
|
|
P210
,
P377
,
P381
,
P403
|
| NFPA 704
(fire diamond)
|
|
| Flash point
|
?108 °C (?162 °F; 165 K)
|
| Safety data sheet
(SDS)
|
External MSDS
|
| Related compounds
|
|
|
Ethylene
,
Isomers of Butylene
;
Allyl
,
Propenyl
|
Related compounds
|
Propane
,
Propyne
Propadiene
,
1-Propanol
2-Propanol
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25 °C [77 °F], 100 kPa).
|
Chemical compound
Propylene
, also known as
propene
, is an
unsaturated
organic compound
with the
chemical formula
CH
3
CH=CH
2
. It has one
double bond
, and is the second simplest member of the
alkene
class of
hydrocarbons
. It is a colorless gas with a faint petroleum-like odor.
[3]
Propylene is a product of combustion from forest fires, cigarette smoke, and motor vehicle and aircraft exhaust.
[4]
It was discovered in 1850 by
A. W. von Hoffman
's student Captain (later Major General
[5]
)
John Williams Reynolds
as the only gaseous product of
thermal decomposition
of
amyl alcohol
to react with chlorine and bromine.
[6]
Production
[
edit
]
Steam cracking
[
edit
]
The dominant technology for producing propylene is
steam cracking
, using
propane
as the
feedstock
. Cracking propane yields a mixture of
ethylene
, propylene,
methane
,
hydrogen gas
, and other related compounds. The yield of propylene is about 15%. The other principal feedstock is naphtha, especially in the
Middle East
and Asia.
[7]
Propylene can be separated by
fractional distillation
from the hydrocarbon mixtures obtained from cracking and other refining processes; refinery-grade propene is about 50 to 70%.
[8]
In the United States,
shale gas
is a major source of propane.
Olefin conversion technology
[
edit
]
In the Phillips triolefin or
olefin conversion technology
, propylene is interconverted with
ethylene
and
2-butenes
.
Rhenium
and
molybdenum
catalysts are used:
[9]
![{\displaystyle {\ce {CH2=CH2{}+CH3CH=CHCH3->[][{\text{Re, Mo}} \atop {\text{catalyst}}]2CH2=CHCH3}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/acdd4b00da58144bb78872e0663521fe689b07b1)
The technology is founded on an
olefin metathesis
reaction discovered at
Phillips Petroleum Company
.
[10]
[11]
Propylene yields of about 90 wt% are achieved.
Related is the
Methanol-to-Olefins/Methanol-to-Propene
process. It converts
synthesis gas (syngas)
to
methanol
, and then
converts the methanol to ethylene and/or propene
. The process produces water as a by-product.
Synthesis gas
is produced from the reformation of natural gas or by the steam-induced reformation of petroleum products such as naphtha, or by
gasification of coal
.
Fluid catalytic cracking
[
edit
]
High severity
fluid catalytic cracking
(FCC) uses traditional FCC technology under severe conditions (higher catalyst-to-oil ratios, higher steam injection rates, higher temperatures, etc.) in order to maximize the amount of propene and other light products. A high severity FCC unit is usually fed with gas oils (paraffins) and residues, and produces about 20?25% (by mass) of propene on feedstock together with greater volumes of motor gasoline and distillate byproducts. These high temperature processes are expensive and have a high carbon footprint. For these reasons, alternative routes to propylene continue to attract attention.
[12]
Other commercialized methods
[
edit
]
On-purpose propylene production technologies were developed throughout the twentieth century. Of these, propane dehydrogenation technologies such as the CATOFIN and OLEFLEX processes have become common, although they still make up a minority of the market, with most of the olefin being sourced from the above mentioned cracking technologies. Platinum, chromia, and vanadium catalysts are common in propane dehydrogenation processes.
Market
[
edit
]
Propene production has remained static at around 35 million
tonnes
(Europe and North America only) from 2000 to 2008, but it has been increasing in East Asia, most notably Singapore and China.
[13]
Total world production of propene is currently about half that of ethylene.
Research
[
edit
]
The use of engineered
enzymes
has been explored but has not been commercialized.
[14]
There is ongoing research into the use of oxygen carrier catalysts for the oxidative dehydrogenation of propane. This poses several advantages, as this reaction mechanism can occur at lower temperatures than conventional dehydrogenation, and may not be equilibrium-limited because oxygen is used to combust the hydrogen by-product.
[15]
Uses
[
edit
]
Propene is the second most important starting product in the
petrochemical industry
after
ethylene
. It is the raw material for a wide variety of products.
Polypropylene
manufacturers consume nearly two thirds of global production.
[16]
Polypropylene end uses include films, fibers, containers, packaging, and caps and closures. Propene is also used for the production of important chemicals such as
propylene oxide
,
acrylonitrile
,
cumene
,
butyraldehyde
, and
acrylic acid
. In the year 2013 about 85 million tonnes of propene were processed worldwide.
[16]
Propene and
benzene
are converted to
acetone
and
phenol
via the
cumene process
.
 Overview of the cumene process
Overview of the cumene process
Propene is also used to produce
isopropyl alcohol
(propan-2-ol),
acrylonitrile
,
propylene oxide
, and
epichlorohydrin
.
[17]
The industrial production of
acrylic acid
involves the catalytic partial oxidation of propene.
[18]
Propylene is an intermediate in the oxidation to acrylic acid.
In industry and workshops, propene is used as an alternative fuel to acetylene in
Oxy-fuel welding and cutting
, brazing and heating of metal for the purpose of bending. It has become a standard in
BernzOmatic
products and others in MAPP substitutes,
[19]
now that true
MAPP gas
is no longer available.
Reactions
[
edit
]
Propene resembles other alkenes in that it undergoes
addition
reactions relatively easily at room temperature. The relative weakness of its double bond explains its tendency to react with substances that can achieve this transformation. Alkene reactions include: 1)
polymerization
, 2)
oxidation
, 3)
halogenation
and
hydrohalogenation
, 4)
alkylation
, 5)
hydration
, 6)
oligomerization
, and 7)
hydroformylation
.
Complexes of transition metals
[
edit
]
Foundational to hydroformylation, alkene metathesis, and polymerization are
metal-propylene complexes
, which are intermediates in these processes. Propylene is
prochiral
, meaning that binding of a reagent (such as a metal electrophile) to the C=C group yields one of two
enantiomers
.
Polymerization
[
edit
]
The majority of propene is used to form polypropylene, a very important commodity
thermoplastic
, through
chain-growth polymerization
.
[16]
In the presence of a suitable catalyst (typically a
Ziegler?Natta catalyst
), propene will polymerize. There are multiple ways to achieve this, such as using high pressures to suspending the catalyst in a solution of liquid propene, or running gaseous propene through a
fluidized bed reactor
.
[20]

Dimerization
[
edit
]
In the presence of
catalysts
, propylene
dimerizes
to give
2,3-dimethyl-1-butene
and/or
2,3-dimethyl-2-butene
.
[21]
Environmental safety
[
edit
]
Propene is a product of combustion from forest fires, cigarette smoke, and motor vehicle and aircraft exhaust.
[4]
It is an impurity in some heating gases. Observed concentrations have been in the range of 0.1?4.8 parts per billion (
ppb
) in rural air, 4?10.5 ppb in urban air, and 7?260 ppb in industrial air samples.
[8]
In the United States and some European countries a
threshold limit value
of 500 parts per million (
ppm
) was established for occupational (8-hour
time-weighted average
) exposure. It is considered a
volatile organic compound
(VOC) and emissions are regulated by many governments, but it is not listed by the U.S. Environmental Protection Agency (EPA) as a
hazardous air pollutant
under the
Clean Air Act
. With a relatively short half-life, it is not expected to bioaccumulate.
[8]
Propene has low acute toxicity from inhalation and is not considered to be carcinogenic. Chronic toxicity studies in mice did not yield significant evidence suggesting adverse effects. Humans briefly exposed to 4,000 ppm did not experience any noticeable effects.
[22]
Propene is dangerous from its potential to displace oxygen as an
asphyxiant gas
, and from its high flammability/explosion risk.
Bio-propylene
is the
bio-based
propylene.
[23]
[24]
It has been examined, motivated by diverse interests such a
carbon footprint
. Production from
glucose
has been considered.
[25]
More advanced ways of addressing such issues focus on electrification alternatives to
steam cracking
.
Storage and handling
[
edit
]
Propene is flammable. Propene is usually stored as liquid under pressure, although it is also possible to store it safely as gas at ambient temperature in approved containers.
[26]
Occurrence in nature
[
edit
]
Propene is detected in the
interstellar medium
through microwave spectroscopy.
[27]
On September 30, 2013,
NASA
also announced that the Cassini orbiter spacecraft, part of the
Cassini-Huygens
mission, had discovered small amounts of naturally occurring propene in the atmosphere of
Titan
using spectroscopy.
[28]
[29]
See also
[
edit
]
References
[
edit
]
- ^
"Front Matter".
Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013 (Blue Book)
. Cambridge:
The Royal Society of Chemistry
. 2014. p. 31.
doi
:
10.1039/9781849733069-FP001
.
ISBN
978-0-85404-182-4
.
- ^
"Propylene"
.
pubchem.ncbi.nlm.nih.gov
. Retrieved
14 December
2021
.
- ^
"Propylene"
.
- ^
a
b
Morgott, David (2018-01-04).
"The Human Exposure Potential from Propylene Releases to the Environment"
.
International Journal of Environmental Research and Public Health
.
15
(1): 66.
doi
:
10.3390/ijerph15010066
.
ISSN
1660-4601
.
PMC
5800165
.
PMID
29300328
.
- ^
"Maj Gen John Williams Reynolds, FCS"
.
geni_family_tree
. 1816-12-25
. Retrieved
2023-12-30
.
- ^
Rasmussen, Seth C. (2018), Rasmussen, Seth C. (ed.),
"Introduction"
,
Acetylene and Its Polymers: 150+ Years of History
, SpringerBriefs in Molecular Science, Cham: Springer International Publishing, pp. 1?19,
doi
:
10.1007/978-3-319-95489-9_1
,
ISBN
978-3-319-95489-9
, retrieved
2023-12-30
- ^
Ashford's Dictionary of Industrial Chemicals, Third edition, 2011,
ISBN
978-0-9522674-3-0
, pages 7766-9
- ^
a
b
c
"Product Safety Assessment(PSA): Propylene"
. Dow Chemical Co. Archived from
the original
on 2013-08-28
. Retrieved
2011-07-11
.
- ^
Ghashghaee, Mohammad (2018). "Heterogeneous catalysts for gas-phase conversion of ethylene to higher olefins".
Rev. Chem. Eng
.
34
(5): 595?655.
doi
:
10.1515/revce-2017-0003
.
S2CID
103664623
.
- ^
Banks, R. L.; Bailey, G. C. (1964). "Olefin Disproportionation. A New Catalytic Process".
Industrial & Engineering Chemistry Product Research and Development
.
3
(3): 170?173.
doi
:
10.1021/i360011a002
.
- ^
Lionel Delaude; Alfred F. Noels (2005). "Metathesis".
Kirk-Othmer Encyclopedia of Chemical Technology
. Weinheim: Wiley-VCH.
doi
:
10.1002/0471238961.metanoel.a01
.
ISBN
978-0-471-23896-6
.
- ^
Schiffer, Zachary J.; Manthiram, Karthish (2017). "Electrification and Decarbonization of the Chemical Industry".
Joule
.
1
: 10?14.
doi
:
10.1016/j.joule.2017.07.008
.
hdl
:
1721.1/124019
.
S2CID
117360588
.
- ^
Amghizar, Ismael; Vandewalle, Laurien A.; Van Geem, Kevin M.; Marin, Guy B. (2017).
"New Trends in Olefin Production"
.
Engineering
.
3
(2): 171?178.
doi
:
10.1016/J.ENG.2017.02.006
.
- ^
de Guzman, Doris (October 12, 2012).
"Global Bioenergies in bio-propylene"
.
Green Chemicals Blog
.
- ^
Wu, Tianwei; Yu, Qingbo; Roghair; et al. (2020).
"Chemical looping oxidative dehydrogenation of propane: A comparative study of Ga-based, Mo-based, V-based oxygen carriers"
.
Chemical Engineering and Processing - Process Intensification
.
157
: 108137.
Bibcode
:
2020CEPPI.15708137W
.
doi
:
10.1016/j.cep.2020.108137
.
ISSN
0255-2701
.
- ^
a
b
c
"Market Study: Propylene (2nd edition), Ceresana, December 2014"
. ceresana.com
. Retrieved
2015-02-03
.
- ^
Budavari, Susan, ed. (1996). "8034. Propylene".
The Merck Index, Twelfth Edition
. New Jersey: Merck & Co. pp. 1348?1349.
- ^
J.G.L., Fierro (Ed.) (2006).
Metal Oxides, Chemistry and Applications
. CRC Press. pp. 414?455.
- ^
For example, "MAPP-Pro"
- ^
Heggs, T. Geoffrey (2011-10-15),
"Polypropylene"
, in Wiley-VCH Verlag GmbH & Co. KGaA (ed.),
Ullmann's Encyclopedia of Industrial Chemistry
, Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA, pp. o21_o04,
doi
:
10.1002/14356007.o21_o04
,
ISBN
978-3-527-30673-2
, retrieved
2021-07-09
- ^
Olivier-Bourbigou, H.; Breuil, P. A. R.; Magna, L.; Michel, T.; Espada Pastor, M. Fernandez; Delcroix, D. (2020).
"Nickel Catalyzed Olefin Oligomerization and Dimerization"
(PDF)
.
Chemical Reviews
.
120
(15): 7919?7983.
doi
:
10.1021/acs.chemrev.0c00076
.
PMID
32786672
.
S2CID
221124789
.
- ^
PubChem.
"Hazardous Substances Data Bank (HSDB): 175"
.
pubchem.ncbi.nlm.nih.gov
. Retrieved
2021-07-09
.
- ^
Bio-based drop-in, smart drop-in and dedicated chemicals
- ^
Duurzame bioplastics op basis van hernieuwbare grondstoffen
- ^
Guzman, Doris de (12 October 2012).
"Global Bioenergies in bio-propylene"
.
Green Chemicals Blog
. Retrieved
2021-07-09
.
- ^
Encyclopedia of Chemical Technology, Fourth edition, 1996,
ISBN
0471-52689-4
(v.20), page 261
- ^
Marcelino, N.; Cernicharo, J.; Agundez, M.; et al. (2007-08-10).
"Discovery of Interstellar Propylene (CH2CHCH3): Missing Links in Interstellar Gas-Phase Chemistry"
.
The Astrophysical Journal
.
665
(2). IOP: L127?L130.
arXiv
:
0707.1308
.
Bibcode
:
2007ApJ...665L.127M
.
doi
:
10.1086/521398
.
S2CID
15832967
.
- ^
"Spacecraft finds propylene on Saturn moon, Titan"
. UPI.com. 2013-09-30
. Retrieved
2013-11-12
.
- ^
"Cassini finds ingredient of household plastic on Saturn moon"
. Spacedaily.com
. Retrieved
2013-11-12
.
|
|---|
| Alkenes
| |
|---|
| Preparations
| |
|---|
| Reactions
| |
|---|
|
|---|
| Alcohols
| |
|---|
| Barbiturates
| |
|---|
| Benzodiazepines
| |
|---|
| Carbamates
| |
|---|
| Flavonoids
| |
|---|
| Imidazoles
| |
|---|
| Kava
constituents
| |
|---|
| Monoureides
| |
|---|
| Neuroactive steroids
| |
|---|
| Nonbenzodiazepines
| |
|---|
| Phenols
| |
|---|
| Piperidinediones
| |
|---|
| Pyrazolopyridines
| |
|---|
| Quinazolinones
| |
|---|
| Volatiles
/
gases
| |
|---|
| Others/unsorted
|
- 3-Hydroxybutanal
- α-EMTBL
- AA-29504
- Alogabat
- Avermectins
(e.g.,
ivermectin
)
- Bromide
compounds (e.g.,
lithium bromide
,
potassium bromide
,
sodium bromide
)
- Carbamazepine
- Chloralose
- Chlormezanone
- Clomethiazole
- Darigabat
- DEABL
- Deuterated etifoxine
- Dihydroergolines
(e.g.,
dihydroergocryptine
,
dihydroergosine
,
dihydroergotamine
,
ergoloid (dihydroergotoxine)
)
- DS2
- Efavirenz
- Etazepine
- Etifoxine
- Fenamates
(e.g.,
flufenamic acid
,
mefenamic acid
,
niflumic acid
,
tolfenamic acid
)
- Fluoxetine
- Flupirtine
- Hopantenic acid
- KRM-II-81
- Lanthanum
- Lavender oil
- Lignans
(e.g.,
4-O-methylhonokiol
,
honokiol
,
magnolol
,
obovatol
)
- Loreclezole
- Menthyl isovalerate (validolum)
- Monastrol
- Niacin
- Niacinamide
- Org 25,435
- Phenytoin
- Propanidid
- Retigabine (ezogabine)
- Safranal
- Seproxetine
- Stiripentol
- Sulfonylalkanes
(e.g.,
sulfonmethane (sulfonal)
,
tetronal
,
trional
)
- Terpenoids
(e.g.,
borneol
)
- Topiramate
- Valerian
constituents (e.g.,
isovaleric acid
,
isovaleramide
,
valerenic acid
,
valerenol
)
|
|---|
|