Chemical reaction in which a nucleophile is affixed to the substrate
In
chemistry
, a
nucleophilic substitution
(
S
N
) is a class of
chemical reactions
in which an
electron
-rich
chemical species
(known as a
nucleophile
) replaces a
functional group
within another electron-deficient molecule (known as the
electrophile
). The molecule that contains the electrophile and the leaving functional group is called the
substrate
.
[1]
[2]
The most general form of the reaction may be given as the following:

The electron pair (
:
) from the nucleophile (Nuc)
attacks
the substrate (
R?LG
) and bonds with it. Simultaneously, the leaving group (LG) departs with an electron pair. The principal product in this case is
R?Nuc
. The nucleophile may be electrically neutral or negatively charged, whereas the substrate is typically neutral or positively charged.
An example of nucleophilic substitution is the
hydrolysis
of an
alkyl
bromide
, R-Br under basic conditions, where the attacking nucleophile is
hydroxyl
(
OH
?
) and the
leaving group
is
bromide
(
Br
?
).

Nucleophilic substitution reactions are common in
organic chemistry
. Nucleophiles often attack a
saturated
aliphatic
carbon. Less often, they may attack an
aromatic
or unsaturated carbon.
[3]
Saturated carbon centres
[
edit
]
S
N
1 and S
N
2 reactions
[
edit
]
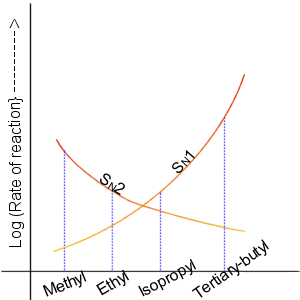 A graph showing the relative reactivities of the different alkyl halides towards S
N
1 and S
N
2 reactions (also see Table 1).
A graph showing the relative reactivities of the different alkyl halides towards S
N
1 and S
N
2 reactions (also see Table 1).
In 1935,
Edward D. Hughes
and
Sir Christopher Ingold
studied nucleophilic substitution reactions of
alkyl halides
and related compounds. They proposed that there were two main mechanisms at work, both of them competing with each other. The two main mechanisms were the
S
N
1 reaction
and the
S
N
2 reaction
, where
S
stands for substitution,
N
stands for nucleophilic, and the number represents the
kinetic order
of the reaction.
[4]
In the S
N
2 reaction, the addition of the nucleophile and the elimination of leaving group take place simultaneously (i.e. a
concerted reaction
). S
N
2 occurs when the central carbon atom is easily accessible to the nucleophile.
[5]
In S
N
2 reactions, there are a few conditions that affect the rate of the reaction. First of all, the 2 in S
N
2 implies that there are two concentrations of substances that affect the rate of reaction: substrate (Sub) and nucleophile. The rate equation for this reaction would be Rate=k[Sub][Nuc]. For a S
N
2 reaction, an
aprotic solvent
is best, such as acetone, DMF, or DMSO. Aprotic solvents do not add protons (H
+
ions) into solution; if protons were present in S
N
2 reactions, they would react with the nucleophile and severely limit the reaction rate. Since this reaction occurs in one step,
steric effects
drive the reaction speed. In the intermediate step, the nucleophile is 185 degrees from the leaving group and the stereochemistry is inverted as the nucleophile bonds to make the product. Also, because the intermediate is partially bonded to the nucleophile and leaving group, there is no time for the substrate to rearrange itself: the nucleophile will bond to the same carbon that the leaving group was attached to. A final factor that affects reaction rate is nucleophilicity; the nucleophile must attack an atom other than a hydrogen.
By contrast the S
N
1 reaction involves two steps. S
N
1 reactions tend to be important when the central carbon atom of the substrate is surrounded by bulky groups, both because such groups interfere sterically with the S
N
2 reaction (discussed above) and because a highly substituted carbon forms a stable
carbocation
.
Like S
N
2 reactions, there are quite a few factors that affect the reaction rate of S
N
1 reactions. Instead of having two concentrations that affect the reaction rate, there is only one, substrate. The rate equation for this would be Rate=k[Sub]. Since the rate of a reaction is only determined by its slowest step, the rate at which the leaving group "leaves" determines the speed of the reaction. This means that the better the leaving group, the faster the reaction rate. A general rule for what makes a good leaving group is the weaker the conjugate base, the better the leaving group. In this case, halogens are going to be the best leaving groups, while compounds such as amines, hydrogen, and alkanes are going to be quite poor leaving groups. As S
N
2 reactions were affected by sterics, S
N
1 reactions are determined by bulky groups attached to the carbocation. Since there is an intermediate that actually contains a positive charge, bulky groups attached are going to help stabilize the charge on the carbocation through resonance and distribution of charge. In this case, tertiary carbocation will react faster than a secondary which will react much faster than a primary. It is also due to this carbocation intermediate that the product does not have to have inversion. The nucleophile can attack from the top or the bottom and therefore create a racemic product. It is important to use a protic solvent, water and alcohols, since an aprotic solvent could attack the intermediate and cause unwanted product. It does not matter if the hydrogens from the protic solvent react with the nucleophile since the nucleophile is not involved in the rate determining step.
Reactions
[
edit
]
There are many reactions in organic chemistry involving this type of mechanism. Common examples include:
- R?X
→
R?H
using
LiAlH
4
(S
N
2)
- R?Br + OH
?
→
R?OH
+
Br
?
(S
N
2) or
- R?Br + H
2
O → R?OH +
HBr
(S
N
1)
- R?Br +
OR'
?
→
R?OR'
+ Br
?
(S
N
2)
Borderline mechanism
[
edit
]
An example of a substitution reaction taking place by a so-called
borderline mechanism
as originally studied by Hughes and Ingold
[6]
is the reaction of
1-phenylethyl chloride
with
sodium methoxide
in methanol.

The
reaction rate
is found to the sum of S
N
1 and S
N
2 components with 61% (3,5 M, 70 °C) taking place by the latter.
Other mechanisms
[
edit
]
Besides S
N
1 and S
N
2, other mechanisms are known, although they are less common. The
S
N
i
mechanism is observed in reactions of
thionyl chloride
with
alcohols
, and it is similar to S
N
1 except that the nucleophile is delivered from the same side as the leaving group.
Nucleophilic substitutions can be accompanied by an
allylic rearrangement
as seen in reactions such as the
Ferrier rearrangement
. This type of mechanism is called an S
N
1' or S
N
2' reaction (depending on the kinetics). With
allylic
halides or sulphonates, for example, the nucleophile may attack at the γ unsaturated carbon in place of the carbon bearing the leaving group. This may be seen in the reaction of 1-chloro-2-butene with
sodium hydroxide
to give a mixture of 2-buten-1-ol and 1-buten-3-ol:

The
Sn1CB mechanism
appears in
inorganic chemistry
. Competing mechanisms exist.
[7]
[8]
In
organometallic chemistry
the
nucleophilic abstraction
reaction occurs with a nucleophilic substitution mechanism.
Unsaturated carbon centres
[
edit
]
Nucleophilic substitution via the S
N
1 or S
N
2 mechanism does not generally occur with vinyl or aryl halides or related compounds. Under certain conditions nucleophilic substitutions may occur, via other mechanisms such as those described in the
nucleophilic aromatic substitution
article.
When the substitution occurs at the
carbonyl
group, the
acyl
group may undergo
nucleophilic acyl substitution
. This is the normal mode of substitution with
carboxylic acid
derivatives such as
acyl chlorides
,
esters
and
amides
.
References
[
edit
]
- ^
March, J. (1992).
Advanced Organic Chemistry
(4th ed.). New York: Wiley.
ISBN
9780471601807
.
- ^
R. A. Rossi, R. H. de Rossi,
Aromatic Substitution by the S
RN
1 Mechanism, ACS Monograph Series No. 178, American Chemical Society, 1983.
ISBN
0-8412-0648-1
.
- ^
L. G. Wade,
Organic Chemistry
, 5th ed., Prentice Hall, Upper Saddle River, New Jersey, 2003.
- ^
S. R. Hartshorn,
Aliphatic Nucleophilic Substitution
, Cambridge University Press, London, 1973.
ISBN
0-521-09801-7
- ^
Introducing Aliphatic Substitution with a Discovery Experiment Using Competing Electrophiles
Timothy P. Curran, Amelia J. Mostovoy, Margaret E. Curran, and Clara Berger Journal of Chemical Education 2016 93 (4), 757-761
doi
:
10.1021/acs.jchemed.5b00394
- ^
253. Reaction kinetics and the Walden inversion. Part II. Homogeneous hydrolysis, alcoholysis, and ammonolysis of -phenylethyl halides
Edward D. Hughes, Christopher K. Ingold and Alan D. Scott,
J. Chem. Soc.
,
1937
, 1201
doi
:
10.1039/JR9370001201
- ^
N.S.Imyanitov.
Electrophilic Bimolecular Substitution as an Alternative to Nucleophilic Monomolecular Substitution in Inorganic and Organic Chemistry
. J. Gen. Chem. USSR (Engl. Transl.)
1990
; 60 (3); 417-419.
- ^
Unimolecular Nucleophilic Substitution does not Exist! / N.S.Imyanitov.
SciTecLibrary
External links
[
edit
]