Atoms or particles produced by nuclear fission
Nuclear fission products
are the atomic fragments left after a large atomic nucleus undergoes
nuclear fission
. Typically, a large
nucleus
like that of
uranium
fissions by splitting into two smaller nuclei, along with a few
neutrons
, the release of heat energy (
kinetic energy
of the nuclei), and
gamma rays
. The two smaller nuclei are the
fission products
. (See also
Fission products (by element)
).
About 0.2% to 0.4% of fissions are
ternary fissions
, producing a third light nucleus such as
helium-4
(90%) or
tritium
(7%).
The fission products themselves are usually unstable and therefore radioactive. Due to being relatively neutron-rich for their atomic number, many of them quickly undergo
beta decay
. This releases additional energy in the form of
beta particles
,
antineutrinos
, and
gamma rays
. Thus, fission events normally result in beta and additional gamma radiation that begins immediately after, even though this radiation is not produced directly by the fission event itself.
The produced
radionuclides
have varying
half-lives
, and therefore vary in
radioactivity
. For instance,
strontium-89
and
strontium-90
are produced in similar quantities in fission, and each nucleus decays by
beta
emission. But
90
Sr has a 30-year half-life, and
89
Sr a 50.5-day half-life. Thus in the 50.5 days it takes half the
89
Sr atoms to decay, emitting the same number of beta particles as there were decays, less than 0.4% of the
90
Sr atoms have decayed, emitting only 0.4% of the betas. The radioactive emission rate is highest for the shortest lived radionuclides, although they also decay the fastest. Additionally, less stable fission products are less likely to decay to stable nuclides, instead decaying to other radionuclides, which undergo further decay and radiation emission, adding to the radiation output. It is these short lived fission products that are the immediate hazard of spent fuel, and the energy output of the radiation also generates significant heat which must be considered when storing spent fuel. As there are hundreds of different radionuclides created, the initial radioactivity level fades quickly as short lived radionuclides decay, but never ceases completely as longer lived radionuclides make up more and more of the remaining unstable atoms.
[1]
In fact the short lived products are so predominant that 87 percent decay to stable isotopes within the first month after removal from the reactor core.
[2]
Formation and decay
[
edit
]
The sum of the
atomic mass
of the two atoms produced by the fission of one
fissile
atom
is always less than the atomic mass of the original atom. This is because some of the mass is lost as free
neutrons
, and once kinetic energy of the fission products has been removed (i.e., the products have been cooled to extract the heat provided by the reaction), then the mass associated with this energy is lost to the system also, and thus appears to be "missing" from the cooled fission products.
Since the nuclei that can readily undergo fission are particularly neutron-rich (e.g. 61% of the
nucleons
in
uranium-235
are neutrons), the initial fission products are often more neutron-rich than stable nuclei of the same mass as the fission product (e.g. stable
zirconium
-90 is 56% neutrons compared to unstable
strontium
-90 at 58%). The initial fission products therefore may be unstable and typically undergo
beta decay
to move towards a stable configuration, converting a neutron to a
proton
with each beta emission. (Fission products do not decay via
alpha decay
.)
A few neutron-rich and short-lived initial fission products decay by ordinary beta decay (this is the source of perceptible half life, typically a few tenths of a second to a few seconds), followed by immediate emission of a neutron by the excited daughter-product. This process is the source of so-called
delayed neutrons
, which play an important role in control of a
nuclear reactor
.
The first beta decays are rapid and may release high energy
beta particles
or
gamma radiation
. However, as the fission products approach stable nuclear conditions, the last one or two decays may have a long
half-life
and release less energy.
Radioactivity over time
[
edit
]
Fission products have half-lives of 90 years (
samarium-151
) or less, except for seven
long-lived fission products
that have half lives of 211,100 years (
technetium-99
) or more. Therefore, the total radioactivity of a mixture of pure fission products decreases rapidly for the first several hundred years (controlled by the short-lived products) before stabilizing at a low level that changes little for hundreds of thousands of years (controlled by the seven long-lived products).
This behavior of pure fission products with actinides removed, contrasts with the decay of fuel that still contains
actinides
. This fuel is produced in the so-called "open" (i.e., no
nuclear reprocessing
)
nuclear fuel cycle
. A number of these actinides have half lives in the missing range of about 100 to 200,000 years, causing some difficulty with storage plans in this time-range for open cycle non-reprocessed fuels.
Proponents of nuclear fuel cycles which aim to consume all their actinides by fission, such as the
Integral Fast Reactor
and
molten salt reactor
, use this fact to claim that within 200 years, their fuel wastes are no more radioactive than the original
uranium ore
.
[3]
Fission products emit
beta radiation
, while actinides primarily emit
alpha radiation
. Many of each also emit
gamma radiation
.
Yield
[
edit
]
 Fission product yields by mass for
thermal neutron
fission of
uranium-235
,
plutonium-239
, a combination of the two typical of current nuclear power reactors, and
uranium-233
used in the
thorium cycle
.
Fission product yields by mass for
thermal neutron
fission of
uranium-235
,
plutonium-239
, a combination of the two typical of current nuclear power reactors, and
uranium-233
used in the
thorium cycle
.
Each fission of a parent atom produces a different set of fission product atoms. However, while an individual fission is not predictable, the fission products are statistically predictable. The amount of any particular isotope produced per fission is called its yield, typically expressed as percent per parent fission; therefore, yields total to 200%, not 100%. (The true total is in fact slightly greater than 200%, owing to rare cases of
ternary fission
.)
While fission products include every element from
zinc
through the
lanthanides
, the majority of the fission products occur in two peaks. One peak occurs at about (expressed by atomic masses 85 through 105)
strontium
to
ruthenium
while the other peak is at about
tellurium
to
neodymium
(expressed by atomic masses 130 through 145). The yield is somewhat dependent on the parent atom and also on the energy of the initiating neutron.
In general the higher the energy of the state that undergoes nuclear fission, the more likely that the two fission products have similar mass. Hence, as the neutron energy increases and/or the energy of the
fissile
atom increases, the valley between the two peaks becomes more shallow.
[4]
For instance, the curve of yield against mass for
239
Pu has a more shallow valley than that observed for
235
U when the neutrons are
thermal neutrons
. The curves for the fission of the later
actinides
tend to make even more shallow valleys. In extreme cases such as
259
Fm
, only one peak is seen; this is a consequence of symmetric fission becoming dominant due to
shell effects
.
[5]
The adjacent figure shows a typical fission product distribution from the fission of uranium. Note that in the calculations used to make this graph, the activation of fission products was ignored and the fission was assumed to occur in a single moment rather than a length of time. In this bar chart results are shown for different cooling times (time after fission).
Because of the stability of nuclei with
even numbers of protons and/or neutrons
, the curve of yield against element is not a smooth curve but tends to alternate. Note that the curve against mass number is smooth.
[6]
Production
[
edit
]
Small amounts of fission products are naturally formed as the result of either
spontaneous fission
of natural uranium, which occurs at a low rate, or as a result of neutrons from
radioactive decay
or reactions with
cosmic ray
particles. The microscopic tracks left by these fission products in some natural minerals (mainly
apatite
and
zircon
) are used in
fission track dating
to provide the cooling (crystallization) ages of natural rocks. The technique has an effective
dating
range of 0.1 Ma to >1.0 Ga depending on the mineral used and the concentration of uranium in that mineral.
About 1.5 billion years ago in a uranium ore body in Africa, a
natural nuclear fission reactor
operated for a few hundred thousand years and produced approximately 5 tonnes of fission products. These fission products were important in providing proof that the natural reactor had occurred.
Fission products are produced in
nuclear weapon
explosions, with the amount depending on the type of weapon.
The largest source of fission products is from
nuclear reactors
. In current
nuclear power
reactors, about 3% of the uranium in the fuel is converted into fission products as a by-product of energy generation. Most of these fission products remain in the fuel unless there is
fuel element failure
or a
nuclear accident
, or the fuel is
reprocessed
.
Power reactors
[
edit
]
Commercial
nuclear fission reactors
are operated in the otherwise self-extinguishing
prompt subcritical
state. Certain fission products decay over seconds to minutes, producing additional
delayed neutrons
crucial to sustaining criticality.
[7]
[8]
An example is
bromine-87
with a half-life of about a minute.
[9]
Operating in this
delayed critical
state, power changes slowly enough to permit human and automatic control. Analogous to
fire dampers
varying the movement of wood embers towards new fuel,
control rods
are moved as the nuclear
fuel burns up
over time.
[10]
[11]
[12]
[13]
In a nuclear power reactor, the main sources of radioactivity are fission products along with
actinides
and
activation products
. Fission products are most of the radioactivity for the first several hundred years, while actinides dominate roughly 10
3
to 10
5
years after fuel use.
Most fission products are retained near their points of production. They are important to reactor operation not only because some contribute delayed neutrons useful for reactor control, but some are neutron poisons that inhibit the nuclear reaction. Buildup of neutron poisons is a key to
how long a given fuel element can be kept in the reactor
. Fission product decay also generates heat that continues even after the reactor has been shut down and fission stopped. This
decay heat
requires removal after shutdown; loss of this cooling damaged the reactors at
Three Mile Island
and
Fukushima
.
If the fuel
cladding
around the fuel develops holes, fission products can leak into the primary
coolant
. Depending on the chemistry, they may settle within the
reactor core
or travel through the coolant system and chemistry control systems are provided to remove them. In a well-designed power reactor running under normal conditions, coolant radioactivity is very low.
The isotope responsible for most of the gamma exposure in
fuel reprocessing
plants (and the Chernobyl site in 2005) is
caesium-137
.
Iodine-129
is a major radioactive isotope released from reprocessing plants. In nuclear reactors both caesium-137 and
strontium-90
are found in locations away from the fuel because they're formed by the
beta decay
of
noble gases
(
xenon-137
, with a 3.8-minute half-life, and
krypton-90
, with a 32-second half-life) which enable them to be deposited away from the fuel, e.g. on
control rods
.
Nuclear reactor poisons
[
edit
]
Some fission products decay with the release of
delayed neutrons
, important to nuclear reactor control.
Other fission products, such as
xenon-135
and
samarium-149
, have a high neutron absorption
cross section
. Since a nuclear reactor must balance neutron production and absorption rates, fission products that absorb neutrons tend to "poison" or shut the reactor down; this is controlled with burnable poisons and control rods. Build-up of xenon-135 during shutdown or low-power operation may poison the reactor enough to
impede restart
or interfere with normal control of the reaction during restart or restoration of full power. This played a major role in the
Chernobyl disaster
.
Nuclear weapons
[
edit
]
Nuclear weapons
use fission as either the partial or the main energy source. Depending on the weapon design and where it is exploded, the relative importance of the fission product radioactivity will vary compared to the activation product radioactivity in the total fallout radioactivity.
The immediate fission products from nuclear weapon fission are essentially the same as those from any other fission source, depending slightly on the particular nuclide that is fissioning. However, the very short time scale for the reaction makes a difference in the particular mix of isotopes produced from an atomic bomb.
For example, the
134
Cs/
137
Cs ratio provides an easy method of distinguishing between fallout from a bomb and the fission products from a power reactor. Almost no
caesium-134
is formed by nuclear fission (because
xenon
-134 is stable). The
134
Cs is formed by the
neutron activation
of the stable
133
Cs which is formed by the decay of isotopes in the
isobar
(A = 133). So in a momentary criticality, by the time that the
neutron
flux becomes zero too little time will have passed for any
133
Cs to be present. While in a power reactor plenty of time exists for the decay of the isotopes in the
isobar
to form
133
Cs, the
133
Cs thus formed can then be activated to form
134
Cs only if the time between the start and the end of the criticality is long.
According to Jiri Hala's textbook,
[14]
the radioactivity in the fission product mixture in an
atom bomb
is mostly caused by short-lived isotopes such as
iodine-131
and
barium-140
. After about four months,
cerium-141
,
zirconium-95
/
niobium-95
, and
strontium-89
represent the largest share of radioactive material. After two to three years,
cerium-144
/
praseodymium-144
,
ruthenium-106
/
rhodium-106
, and
promethium-147
are responsible for the bulk of the radioactivity. After a few years, the radiation is dominated by strontium-90 and caesium-137, whereas in the period between 10,000 and a million years it is
technetium-99
that dominates.
Application
[
edit
]
Some fission products (such as
137
Cs) are used in medical and industrial
radioactive sources
.
99
TcO
4
?
(
pertechnetate
) ion can react with steel surfaces to form a
corrosion resistant layer
. In this way these metaloxo anions act as
anodic
corrosion inhibitors
- it renders the steel surface passive. The formation of
99
TcO
2
on
steel
surfaces is one effect which will retard the release of
99
Tc from
nuclear waste
drums and nuclear equipment which has become lost prior to
decontamination
(e.g.
nuclear submarine
reactors which have been lost at sea).
In a similar way the release of radio-iodine in a serious power reactor accident could be retarded by
adsorption
on metal surfaces within the nuclear plant.
[15]
Much of the other work on the iodine chemistry which would occur during a bad accident has been done.
[16]
Decay
[
edit
]
 The external
gamma
dose for a person in the open near the
Chernobyl disaster
site.
The external
gamma
dose for a person in the open near the
Chernobyl disaster
site.
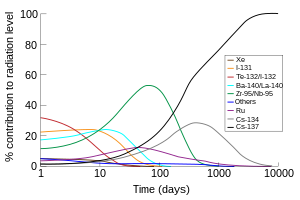 The portion of the total radiation dose (in air) contributed by each isotope versus time after the
Chernobyl disaster
, at the site thereof. Note that this image was drawn using data from the OECD report, and the second edition of "The radiochemical manual".
[17]
The portion of the total radiation dose (in air) contributed by each isotope versus time after the
Chernobyl disaster
, at the site thereof. Note that this image was drawn using data from the OECD report, and the second edition of "The radiochemical manual".
[17]
For fission of
uranium-235
, the predominant radioactive fission products include isotopes of
iodine
,
caesium
,
strontium
,
xenon
and
barium
. The threat becomes smaller with the passage of time. Locations where radiation fields once posed immediate mortal threats, such as much of the
Chernobyl Nuclear Power Plant
on day one of the
accident
and the
ground zero
sites of
U.S. atomic bombings in Japan
(6 hours after detonation) are now relatively safe because the radioactivity has decreased to a low level.
Many of the fission products decay through very short-lived isotopes to form
stable isotopes
, but a considerable number of the
radioisotopes
have
half-lives
longer than a day.
The radioactivity in the fission product mixture is initially mostly caused by short lived isotopes such as
131
I and
140
Ba; after about four months
141
Ce,
95
Zr/
95
Nb and
89
Sr take the largest share, while after about two or three years the largest share is taken by
144
Ce/
144
Pr,
106
Ru/
106
Rh and
147
Pm. Later
90
Sr and
137
Cs are the main radioisotopes, being succeeded by
99
Tc. In the case of a release of radioactivity from a power reactor or used fuel, only some elements are released; as a result, the isotopic signature of the radioactivity is very different from an open air
nuclear detonation
, where all the fission products are dispersed.
Fallout countermeasures
[
edit
]
The purpose of radiological emergency preparedness is to protect people from the effects of radiation exposure after a nuclear accident or bomb.
Evacuation
is the most effective protective measure. However, if evacuation is impossible or even uncertain, then local
fallout shelters
and other measures provide the best protection.
[18]
Iodine
[
edit
]
 Per capita thyroid doses in the continental United States of
iodine-131
resulting from all exposure routes from all atmospheric nuclear tests conducted at the
Nevada Test Site
. See also
Downwinders
.
Per capita thyroid doses in the continental United States of
iodine-131
resulting from all exposure routes from all atmospheric nuclear tests conducted at the
Nevada Test Site
. See also
Downwinders
.
At least three
isotopes of iodine
are important.
129
I
,
131
I
(radioiodine) and
132
I. Open air
nuclear testing
and the
Chernobyl disaster
both released iodine-131.
The short-lived
isotopes
of iodine are particularly harmful because the
thyroid
collects and concentrates
iodide
? radioactive as well as stable. Absorption of radioiodine can lead to acute, chronic, and delayed effects. Acute effects from high doses include
thyroiditis
, while chronic and delayed effects include
hypothyroidism
,
thyroid nodules
, and
thyroid cancer
. It has been shown that the active iodine released from
Chernobyl
and
Mayak
[19]
has resulted in an increase in the incidence of thyroid cancer in the former
Soviet Union
.
One measure which protects against the risk from radio-iodine is taking a dose of
potassium iodide
(KI) before exposure to radioiodine. The non-radioactive iodide "saturates" the thyroid, causing less of the radioiodine to be stored in the body.
Administering potassium iodide reduces the effects of radio-iodine by 99% and is a prudent, inexpensive supplement to
fallout shelters
. A low-cost alternative to commercially available iodine pills is a
saturated solution
of potassium iodide. Long-term storage of KI is normally in the form of
reagent-grade
crystals.
[20]
The administration of known
goitrogen
substances can also be used as a
prophylaxis
in reducing the bio-uptake of iodine, (whether it be the nutritional non-radioactive
iodine-127
or radioactive iodine, radioiodine - most commonly
iodine-131
, as the body cannot discern between different iodine
isotopes
).
Perchlorate
ions, a common water contaminant in the USA due to the
aerospace industry
, has been shown to reduce iodine uptake and thus is classified as a
goitrogen
. Perchlorate ions are a competitive inhibitor of the process by which iodide is actively deposited into thyroid follicular cells. Studies involving healthy adult volunteers determined that at levels above 0.007 milligrams per kilogram per day (mg/(kg·d)), perchlorate begins to temporarily inhibit the thyroid gland's ability to absorb iodine from the bloodstream ("iodide uptake inhibition", thus perchlorate is a known goitrogen).
[21]
The reduction of the iodide pool by perchlorate has dual effects ? reduction of excess hormone synthesis and hyperthyroidism, on the one hand, and reduction of thyroid inhibitor synthesis and hypothyroidism on the other. Perchlorate remains very useful as a single dose application in tests measuring the discharge of radioiodide accumulated in the thyroid as a result of many different disruptions in the further metabolism of iodide in the thyroid gland.
[22]
Treatment of thyrotoxicosis (including Graves' disease) with 600?2,000 mg potassium perchlorate (430-1,400 mg perchlorate) daily for periods of several months or longer was once common practice, particularly in Europe,
[21]
[23]
and perchlorate use at lower doses to treat thyroid problems continues to this day.
[24]
Although 400 mg of potassium perchlorate divided into four or five daily doses was used initially and found effective, higher doses were introduced when 400 mg/day was discovered not to control thyrotoxicosis in all subjects.
[21]
[22]
Current regimens for treatment of
thyrotoxicosis
(including Graves' disease), when a patient is exposed to additional sources of iodine, commonly include 500 mg potassium perchlorate twice per day for 18?40 days.
[21]
[25]
Prophylaxis with perchlorate-containing water at concentrations of 17
ppm
, which corresponds to 0.5 mg/kg-day personal intake, if one is 70 kg and consumes 2 litres of water per day, was found to reduce baseline radioiodine uptake by 67%
[21]
This is equivalent to ingesting a total of just 35 mg of perchlorate ions per day. In another related study where subjects drank just 1 litre of perchlorate-containing water per day at a concentration of 10 ppm, i.e. daily 10 mg of perchlorate ions were ingested, an average 38% reduction in the uptake of iodine was observed.
[26]
However, when the average perchlorate absorption in perchlorate plant workers subjected to the highest exposure has been estimated as approximately 0.5 mg/kg-day, as in the above paragraph, a 67% reduction of iodine uptake would be expected. Studies of chronically exposed workers though have thus far failed to detect any abnormalities of thyroid function, including the uptake of iodine.
[27]
this may well be attributable to sufficient daily exposure or intake of healthy iodine-127 among the workers and the short 8 hr
biological half life
of perchlorate in the body.
[21]
To completely block the uptake of iodine-131 by the purposeful addition of perchlorate ions to a populace's water supply, aiming at dosages of 0.5 mg/kg-day, or a water concentration of 17 ppm, would therefore be grossly inadequate at truly reducing radioiodine uptake. Perchlorate ion concentrations in a region's water supply would need to be much higher, at least 7.15 mg/kg of body weight per day, or a water concentration of 250
ppm
, assuming people drink 2 liters of water per day, to be truly beneficial to the population at preventing
bioaccumulation
when exposed to a radioiodine environment,
[21]
[25]
independent of the availability of
iodate
or
iodide
drugs.
The continual distribution of perchlorate tablets or the addition of perchlorate to the water supply would need to continue for no less than 80?90 days, beginning immediately after the initial release of radioiodine was detected. After 80?90 days passed, released radioactive iodine-131 would have decayed to less than 0.1% of its initial quantity, at which time the danger from biouptake of iodine-131 is essentially over.
[28]
In the event of a radioiodine release, the ingestion of prophylaxis potassium iodide, if available, or even iodate, would rightly take precedence over perchlorate administration, and would be the first line of defense in protecting the population from a radioiodine release. However, in the event of a radioiodine release too massive and widespread to be controlled by the limited stock of iodide and iodate prophylaxis drugs, then the addition of perchlorate ions to the water supply, or distribution of perchlorate tablets would serve as a cheap, efficacious, second line of defense against
carcinogenic
radioiodine bioaccumulation.
The ingestion of goitrogen drugs is, much like potassium iodide also not without its dangers, such as
hypothyroidism
. In all these cases however, despite the risks, the prophylaxis benefits of intervention with iodide, iodate, or perchlorate outweigh the serious cancer risk from radioiodine bioaccumulation in regions where radioiodine has sufficiently contaminated the environment.
Caesium
[
edit
]
The Chernobyl accident released a large amount of
caesium isotopes
which were dispersed over a wide area.
137
Cs is an isotope which is of long-term concern as it remains in the top layers of soil. Plants with shallow root systems tend to absorb it for many years. Hence grass and mushrooms can carry a considerable amount of
137
Cs, which can be transferred to humans through the
food chain
.
One of the best countermeasures in
dairy farming
against
137
Cs is to mix up the soil by deeply ploughing the soil. This has the effect of putting the
137
Cs out of reach of the shallow roots of the grass, hence the level of radioactivity in the grass will be lowered. Also the removal of top few centimeters of soil and its burial in a shallow trench will reduce the dose to humans and animals as the
gamma rays
from
137
Cs will be attenuated by their passage through the soil. The deeper and more remote the trench is, the better the degree of protection.
Fertilizers
containing
potassium
can be used to dilute cesium and limit its uptake by plants.
In
livestock
farming, another countermeasure against
137
Cs is to feed to animals
prussian blue
. This compound acts as an
ion-exchanger
. The
cyanide
is so tightly bonded to the iron that it is safe for a human to consume several grams of prussian blue per day. The prussian blue reduces the
biological half-life
(different from the
nuclear half-life
) of the caesium. The physical or nuclear half-life of
137
Cs is about 30 years. Caesium in humans normally has a biological half-life of between one and four months. An added advantage of the prussian blue is that the caesium which is stripped from the animal in the droppings is in a form which is not available to plants. Hence it prevents the caesium from being recycled. The form of prussian blue required for the treatment of animals, including humans is a special grade. Attempts to use the
pigment
grade used in
paints
have not been successful.
[29]
Strontium
[
edit
]
The addition of
lime
to soils which are poor in
calcium
can reduce the uptake of
strontium
by plants. Likewise in areas where the soil is low in
potassium
, the addition of a potassium fertilizer can discourage the uptake of cesium into plants. However such treatments with either lime or
potash
should not be undertaken lightly as they can alter the
soil chemistry
greatly, so resulting in a change in the plant
ecology
of the land.
[30]
Health concerns
[
edit
]
For introduction of radionuclides into organism, ingestion is the most important route. Insoluble compounds are not absorbed from the gut and cause only local irradiation before they are excreted. Soluble forms however show wide range of absorption percentages.
[31]
See also
[
edit
]
Notes
[
edit
]
References
[
edit
]
- ^
F. William Walker, Dr. George J. Kirouac, Francis M. Rourke. 1977.
Chart of the Nuclides
, twelfth edition. Knolls Atomic Power Laboratory, General Electric Company.
- ^
"What Happens to Nuclear Waste in the U.S.?"
. 19 November 2019.
- ^
"Introduction to ANL's IFR Program"
. 9 October 2007. Archived from
the original
on 9 October 2007.
- ^
Newton, Amos S. (1 January 1949).
"The Fission of Thorium with Alpha-Particles"
.
Physical Review
.
75
(1): 17?29.
Bibcode
:
1949PhRv...75...17N
.
doi
:
10.1103/PhysRev.75.17
.
S2CID
93655149
.
- ^
Pa?ca, H.; Andreev, A.V.; Adamian, G.G.; Antonenko, N.V. (2018).
"Charge distributions of fission fragments of low- and high-energy fission of Fm, No, and Rf isotopes"
.
Physical Review C
.
97
(3): 034621?1?034621?12.
Bibcode
:
2018PhRvC..97c4621P
.
doi
:
10.1103/PhysRevC.97.034621
.
- ^
"Nuclear Fission Yield"
. Archived from
the original
on 28 May 2007
. Retrieved
13 May
2009
.
- ^
"Elementary Physics of Reactor Control"
(PDF)
. Archived from
the original
(PDF)
on 12 July 2019
. Retrieved
15 April
2018
.
- ^
"Nuclear Fission - Fission Reaction"
.
Nuclear Power
.
- ^
"PROMPT AND DELAYED NEUTRONS"
.
nuclearpowertraining.tpub.com
.
- ^
Prompt and Delayed Neutrons
The fact the neutron is produced via this type of decay and this happens orders of magnitude later compared to the emission of the prompt neutrons, plays an extremely important role in the control of the reactor.
- ^
"
"In view of the very low concentration of Uranium used, it is not possible for a commercial nuclear reactor to explode like an atomic bomb from the perspective of physics. A good analogy would be to compare alcoholic spirits to beer. Alcoholic spirits, such as Vodka, typically have a 40% alcohol content, and are highly flammable. Beer, which generally has an alcohol content of less than 5%, does not burn."
"
. Archived from
the original
on 1 August 2018
. Retrieved
15 April
2018
.
- ^
"Nuclear Explained By Analogy Metaphor Examples"
.
www.metamia.com
.
- ^
nuclear education for K-12 students Myths About Nuclear Energy
It is impossible for a reactor to explode like a nuclear weapon; these weapons contain very special materials in very particular configurations, neither of which are present in a nuclear reactor
.
- ^
Hala, Jiri; James D. Navratil (2003).
Radioactivity, Ionizing Radiation, and Nuclear Energy
. Brno: Konvoj.
ISBN
80-7302-053-X
.
- ^
H. Glanneskog. Interactions of
I
2
and
CH
3
I with reactive metals under BWR severe-accident conditions,
Nucl. Engineering and Design
, 2004,
227
, 323-329
- ^
Workshop on iodine aspects of severe accident management. Summary and conclusions
. Nuclear Energy Agency. Committee on the safety of nuclear installations. OCDE. March 7, 2000.
- ^
"Nuclear Data Evaluation Lab"
. Retrieved
13 May
2009
.
- ^
C. Kearney, Nuclear War Survival Skills, Oregon Institute of Science and Medicine,
http://www.oism.org/
- ^
G. Mushkacheva, E. Rabinovich, V. Privalov, S. Povolotskaya, V. Shorokhova, S. Sokolova, V. Turdakova, E. Ryzhova, P. Hall, A. B. Schneider, D. L. Preston, and E. Ron, "Thyroid Abnormalities Associated with Protracted Childhood Exposure to 131I from Atmospheric Emissions from the Mayak Weapons Facility in Russia",
Radiation Research
, 2006,
166
(5), 715-722
- ^
C. Kearney, Nuclear War Survival Skills (Ch. 13), Oregon Institute of Science and Medicine,
http://www.oism.org/
- ^
a
b
c
d
e
f
g
Greer, Monte A.; Goodman, Gay; Pleus, Richard C.; Greer, Susan E. (2002).
"Health Effects Assessment for Environmental Perchlorate Contamination: The Dose Response for Inhibition of Thyroidal Radioiodine Uptake in Humans"
.
Environmental Health Perspectives
.
110
(9): 927?37.
doi
:
10.1289/ehp.02110927
.
PMC
1240994
.
PMID
12204829
.
- ^
a
b
Wolff, J (1998). "Perchlorate and the thyroid gland".
Pharmacological Reviews
.
50
(1): 89?105.
PMID
9549759
.
- ^
Barzilai, D; Sheinfeld, M (1966). "Fatal complications following use of potassium perchlorate in thyrotoxicosis. Report of two cases and a review of the literature".
Israel Journal of Medical Sciences
.
2
(4): 453?6.
PMID
4290684
.
- ^
Woenckhaus, U.; Girlich, C. (2005). "Therapie und Pravention der Hyperthyreose" [Therapy and prevention of hyperthyroidism].
Der Internist
(in German).
46
(12): 1318?23.
doi
:
10.1007/s00108-005-1508-4
.
PMID
16231171
.
S2CID
13214666
.
- ^
a
b
Bartalena, L.; Brogioni, S; Grasso, L; Bogazzi, F; Burelli, A; Martino, E (1996).
"Treatment of amiodarone-induced thyrotoxicosis, a difficult challenge: Results of a prospective study"
.
Journal of Clinical Endocrinology & Metabolism
.
81
(8): 2930?3.
doi
:
10.1210/jcem.81.8.8768854
.
PMID
8768854
.
- ^
Lawrence, J. E.; Lamm, S. H.; Pino, S.; Richman, K.; Braverman, L. E. (2000). "The Effect of Short-Term Low-Dose Perchlorate on Various Aspects of Thyroid Function".
Thyroid
.
10
(8): 659?63.
doi
:
10.1089/10507250050137734
.
PMID
11014310
.
- ^
Lamm, Steven H.; Braverman, Lewis E.; Li, Feng Xiao; Richman, Kent; Pino, Sam; Howearth, Gregory (1999). "Thyroid Health Status of Ammonium Perchlorate Workers: A Cross-Sectional Occupational Health Study".
Journal of Occupational & Environmental Medicine
.
41
(4): 248?60.
doi
:
10.1097/00043764-199904000-00006
.
PMID
10224590
.
- ^
"Nuclear Chemistry: Half-Lives and Radioactive Dating"
.
- ^
For further details of the use of prussian blue please see the
IAEA
report on the
Goiania accident
.
[1]
- ^
Development, Office of Research &.
"Full-Scale and Bench-Scale Studies on the Removal of Strontium from Water (abstract)"
.
cfpub.epa.gov
. Retrieved
14 June
2019
.
- ^
Baratta, Edmond J.; Nations, Food and Agriculture Organization of the United (10 February 1994).
Manual of Food Quality Control: Radionuclides in food
. Food & Agriculture Org.
ISBN
9789251035788
– via Google Books.
- ^
"Half-life, effective"
.
www.euronuclear.org
. Archived from
the original
on 9 July 2014
. Retrieved
25 December
2012
.
Bibliography
[
edit
]
Paul Reuss,
Neutron Physics
, chp 2.10.2, p 75
External links
[
edit
]