Electrically neutral group of two or more atoms
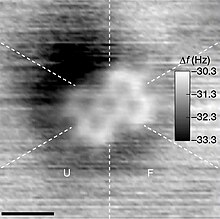 Atomic force microscopy
(AFM) image of a
PTCDA
molecule, in which the five six-carbon rings are visible.
[1]
Atomic force microscopy
(AFM) image of a
PTCDA
molecule, in which the five six-carbon rings are visible.
[1]
 A
scanning tunneling microscopy
image of
pentacene
molecules, which consist of linear chains of five carbon rings.
[2]
A
scanning tunneling microscopy
image of
pentacene
molecules, which consist of linear chains of five carbon rings.
[2]
 AFM image of 1,5,9-trioxo-13-azatriangulene and its chemical structure.
[3]
AFM image of 1,5,9-trioxo-13-azatriangulene and its chemical structure.
[3]
A
molecule
is a group of two or more
atoms
held together by
attractive forces
known as
chemical bonds
; depending on context, the term may or may not include
ions
which satisfy this criterion.
[4]
[5]
[6]
[7]
[8]
In
quantum physics
,
organic chemistry
, and
biochemistry
, the distinction from ions is dropped and
molecule
is often used when referring to
polyatomic ions
.
A molecule may be
homonuclear
, that is, it consists of atoms of one
chemical element
, e.g. two atoms in the
oxygen
molecule (O
2
); or it may be
heteronuclear
, a
chemical compound
composed of more than one element, e.g.
water
(two hydrogen atoms and one oxygen atom; H
2
O). In the
kinetic theory of gases
, the term
molecule
is often used for any gaseous
particle
regardless of its composition. This relaxes the requirement that a molecule contains two or more atoms, since the
noble gases
are individual atoms.
[9]
Atoms and complexes connected by
non-covalent interactions
, such as
hydrogen bonds
or
ionic bonds
, are typically not considered single molecules.
[10]
Concepts similar to molecules have been discussed since ancient times, but modern investigation into the nature of molecules and their bonds began in the 17th century. Refined over time by scientists such as
Robert Boyle
,
Amedeo Avogadro
,
Jean Perrin
, and
Linus Pauling
, the study of molecules is today known as
molecular physics
or molecular chemistry.
Etymology
According to
Merriam-Webster
and the
Online Etymology Dictionary
, the word "molecule" derives from the
Latin
"
moles
" or small unit of mass. The word is derived from French
molecule
(1678), from
Neo-Latin
molecula
, diminutive of Latin
moles
"mass, barrier". The word, which until the late 18th century was used only in Latin form, became popular after being used in works of philosophy by
Descartes
.
[11]
[12]
History
The definition of the molecule has evolved as knowledge of the structure of molecules has increased. Earlier definitions were less precise, defining molecules as the smallest
particles
of pure
chemical substances
that still retain their
composition
and chemical properties.
[13]
This definition often breaks down since many substances in ordinary experience, such as
rocks
,
salts
, and
metals
, are composed of large crystalline networks of
chemically bonded
atoms or
ions
, but are not made of discrete molecules.
The modern concept of molecules can be traced back towards pre-scientific and Greek philosophers such as
Leucippus
and
Democritus
who argued that all the universe is composed of
atoms and voids
. Circa 450 BC
Empedocles
imagined
fundamental elements
(
fire
(
 ),
earth
(
),
earth
(
 ),
air
(
),
air
(
 ), and
water
(
), and
water
(
 )) and "forces" of attraction and repulsion allowing the elements to interact.
)) and "forces" of attraction and repulsion allowing the elements to interact.
A fifth element, the incorruptible quintessence
aether
, was considered to be the fundamental building block of the heavenly bodies. The viewpoint of Leucippus and Empedocles, along with the aether, was accepted by
Aristotle
and passed to medieval and renaissance Europe.
In a more concrete manner, however, the concept of aggregates or units of bonded atoms, i.e. "molecules", traces its origins to
Robert Boyle
's 1661 hypothesis, in his famous treatise
The Sceptical Chymist
, that matter is composed of
clusters of particles
and that chemical change results from the rearrangement of the clusters. Boyle argued that matter's basic elements consisted of various sorts and sizes of particles, called "corpuscles", which were capable of arranging themselves into groups. In 1789,
William Higgins
published views on what he called combinations of "ultimate" particles, which foreshadowed the concept of
valency bonds
. If, for example, according to Higgins, the force between the ultimate particle of oxygen and the ultimate particle of nitrogen were 6, then the strength of the force would be divided accordingly, and similarly for the other combinations of ultimate particles.
Amedeo Avogadro
created the word "molecule".
[14]
His 1811 paper "Essay on Determining the Relative Masses of the Elementary Molecules of Bodies", he essentially states, i.e. according to
Partington
's
A Short History of Chemistry
, that:
[15]
The smallest particles of gases are not necessarily simple atoms, but are made up of a certain number of these atoms united by attraction to form a single
molecule
.
In coordination with these concepts, in 1833 the French chemist
Marc Antoine Auguste Gaudin
presented a clear account of Avogadro's hypothesis,
[16]
regarding atomic weights, by making use of "volume diagrams", which clearly show both semi-correct molecular geometries, such as a linear water molecule, and correct molecular formulas, such as H
2
O:
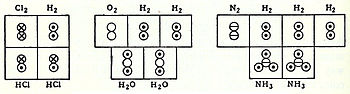 Marc Antoine Auguste Gaudin's volume diagrams of molecules in the gas phase (1833)
Marc Antoine Auguste Gaudin's volume diagrams of molecules in the gas phase (1833)
In 1917, an unknown American undergraduate chemical engineer named
Linus Pauling
was learning the
Dalton hook-and-eye bonding method
, which was the mainstream description of bonds between atoms at the time. Pauling, however, was not satisfied with this method and looked to the newly emerging field of quantum physics for a new method. In 1926, French physicist
Jean Perrin
received the Nobel Prize in physics for proving, conclusively, the existence of molecules. He did this by calculating the
Avogadro constant
using three different methods, all involving liquid phase systems. First, he used a
gamboge
soap-like emulsion, second by doing experimental work on
Brownian motion
, and third by confirming Einstein's theory of particle rotation in the liquid phase.
[17]
In 1927, the physicists
Fritz London
and
Walter Heitler
applied the new quantum mechanics to the deal with the saturable, nondynamic forces of attraction and repulsion, i.e., exchange forces, of the hydrogen molecule. Their valence bond treatment of this problem, in their joint paper,
[18]
was a landmark in that it brought chemistry under quantum mechanics. Their work was an influence on Pauling, who had just received his doctorate and visited Heitler and London in Zurich on a
Guggenheim Fellowship
.
Subsequently, in 1931, building on the work of Heitler and London and on theories found in Lewis' famous article, Pauling published his ground-breaking article "The Nature of the Chemical Bond"
[19]
in which he used
quantum mechanics
to calculate properties and structures of molecules, such as angles between bonds and rotation about bonds. On these concepts, Pauling developed
hybridization theory
to account for bonds in molecules such as CH
4
, in which four sp³ hybridised orbitals are overlapped by
hydrogen
's
1s
orbital, yielding four
sigma (σ) bonds
. The four bonds are of the same length and strength, which yields a molecular structure as shown below:
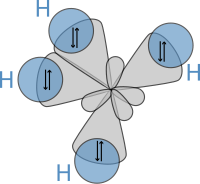 A schematic presentation of hybrid orbitals overlapping hydrogens' s orbitals
A schematic presentation of hybrid orbitals overlapping hydrogens' s orbitals
Molecular science
The science of molecules is called
molecular chemistry
or
molecular physics
, depending on whether the focus is on chemistry or physics. Molecular chemistry deals with the laws governing the interaction between molecules that results in the formation and breakage of chemical bonds, while molecular physics deals with the laws governing their structure and properties. In practice, however, this distinction is vague. In molecular sciences, a molecule consists of a stable system (
bound state
) composed of two or more atoms.
Polyatomic ions
may sometimes be usefully thought of as electrically charged molecules. The term
unstable molecule
is used for very
reactive
species, i.e., short-lived assemblies (
resonances
) of electrons and
nuclei
, such as
radicals
,
molecular ions
,
Rydberg molecules
,
transition states
,
van der Waals complexes
, or systems of colliding atoms as in
Bose?Einstein condensate
.
Prevalence
Molecules as components of matter are common. They also make up most of the oceans and atmosphere. Most organic substances are molecules. The substances of life are molecules, e.g. proteins, the amino acids of which they are composed, the nucleic acids (DNA and RNA), sugars, carbohydrates, fats, and vitamins. The nutrient minerals are generally ionic compounds, thus they are not molecules, e.g. iron sulfate.
However, the majority of familiar solid substances on Earth are made partly or completely of crystals or ionic compounds, which are not made of molecules. These include all of the minerals that make up the substance of the Earth, sand, clay, pebbles, rocks, boulders,
bedrock
, the
molten interior
, and the
core of the Earth
. All of these contain many chemical bonds, but are
not
made of identifiable molecules.
No typical molecule can be defined for salts nor for
covalent crystals
, although these are often composed of repeating
unit cells
that extend either in a
plane
, e.g.
graphene
; or three-dimensionally e.g.
diamond
,
quartz
,
sodium chloride
. The theme of repeated unit-cellular-structure also holds for most metals which are condensed phases with
metallic bonding
. Thus solid metals are not made of molecules. In
glasses
, which are solids that exist in a vitreous disordered state, the atoms are held together by chemical bonds with no presence of any definable molecule, nor any of the regularity of repeating unit-cellular-structure that characterizes salts, covalent crystals, and metals.
Bonding
Molecules are generally held together by
covalent bonding
. Several non-metallic elements exist only as molecules in the environment either in compounds or as homonuclear molecules, not as free atoms: for example, hydrogen.
While some people say a metallic crystal can be considered a single giant molecule held together by
metallic bonding
,
[20]
others point out that metals behave very differently than molecules.
[21]
Covalent
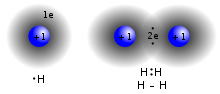 A covalent bond forming H
2
(right) where two
hydrogen atoms
share the two electrons
A covalent bond forming H
2
(right) where two
hydrogen atoms
share the two electrons
A covalent bond is a chemical bond that involves the sharing of
electron pairs
between atoms. These electron pairs are termed
shared pairs
or
bonding pairs
, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is termed
covalent bonding
.
[22]
Ionic
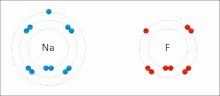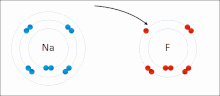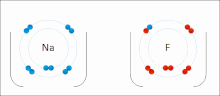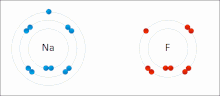 Sodium
and
fluorine
undergoing a redox reaction to form
sodium fluoride
. Sodium loses its outer
electron
to give it a stable
electron configuration
, and this electron enters the fluorine atom
exothermically
.
Sodium
and
fluorine
undergoing a redox reaction to form
sodium fluoride
. Sodium loses its outer
electron
to give it a stable
electron configuration
, and this electron enters the fluorine atom
exothermically
.
Ionic bonding is a type of chemical bond that involves the
electrostatic
attraction between oppositely charged ions, and is the primary interaction occurring in
ionic compounds
. The ions are atoms that have lost one or more
electrons
(termed
cations
) and atoms that have gained one or more electrons (termed
anions
).
[23]
This transfer of electrons is termed
electrovalence
in contrast to
covalence
. In the simplest case, the cation is a
metal
atom and the anion is a
nonmetal
atom, but these ions can be of a more complicated nature, e.g. molecular ions like NH
4
+
or SO
4
2?
. At normal temperatures and pressures, ionic bonding mostly creates solids (or occasionally liquids) without separate identifiable molecules, but the vaporization/sublimation of such materials does produce separate molecules where electrons are still transferred fully enough for the bonds to be considered ionic rather than covalent.
Molecular size
Most molecules are far too small to be seen with the naked eye, although molecules of many
polymers
can reach
macroscopic
sizes, including
biopolymers
such as
DNA
. Molecules commonly used as building blocks for organic synthesis have a dimension of a few
angstroms
(A) to several dozen A, or around one billionth of a meter. Single molecules cannot usually be observed by
light
(as noted above), but small molecules and even the outlines of individual atoms may be traced in some circumstances by use of an
atomic force microscope
. Some of the largest molecules are
macromolecules
or
supermolecules
.
The smallest molecule is the
diatomic
hydrogen (H
2
), with a bond length of 0.74 A.
[24]
Effective molecular radius is the size a molecule displays in solution.
[25]
[26]
The
table of permselectivity for different substances
contains examples.
Molecular formulas
Chemical formula types
The
chemical formula
for a molecule uses one line of chemical element symbols, numbers, and sometimes also other symbols, such as parentheses, dashes, brackets, and
plus
(+) and
minus
(?) signs. These are limited to one typographic line of symbols, which may include subscripts and superscripts.
A compound's
empirical formula
is a very simple type of chemical formula.
[27]
It is the simplest
integer
ratio
of the chemical elements that constitute it.
[28]
For example, water is always composed of a 2:1 ratio of hydrogen to oxygen atoms, and
ethanol
(ethyl alcohol) is always composed of carbon, hydrogen, and oxygen in a 2:6:1 ratio. However, this does not determine the kind of molecule uniquely ?
dimethyl ether
has the same ratios as ethanol, for instance. Molecules with the same
atoms
in different arrangements are called
isomers
. Also carbohydrates, for example, have the same ratio (carbon:hydrogen:oxygen= 1:2:1) (and thus the same empirical formula) but different total numbers of atoms in the molecule.
The
molecular formula
reflects the exact number of atoms that compose the molecule and so characterizes different molecules. However different isomers can have the same atomic composition while being different molecules.
The empirical formula is often the same as the molecular formula but not always. For example, the molecule
acetylene
has molecular formula C
2
H
2
, but the simplest integer ratio of elements is CH.
The
molecular mass
can be calculated from the chemical formula and is expressed in conventional
atomic mass units
equal to 1/12 of the mass of a neutral carbon-12 (
12
C
isotope
) atom. For
network solids
, the term
formula unit
is used in
stoichiometric
calculations.
Structural formula
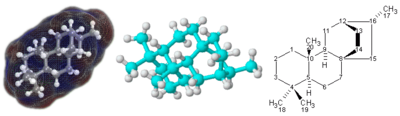 3D
(left and center) and
2D
(right) representations of the
terpenoid
molecule atisane
3D
(left and center) and
2D
(right) representations of the
terpenoid
molecule atisane
For molecules with a complicated 3-dimensional structure, especially involving atoms bonded to four different substituents, a simple molecular formula or even semi-structural chemical formula may not be enough to completely specify the molecule. In this case, a graphical type of formula called a
structural formula
may be needed. Structural formulas may in turn be represented with a one-dimensional chemical name, but such
chemical nomenclature
requires many words and terms which are not part of chemical formulas.
Molecular geometry
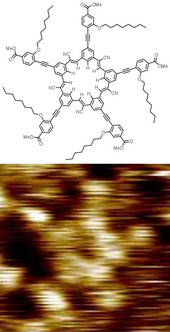 Structure and
STM
image of a "cyanostar"
dendrimer
molecule.
[29]
Structure and
STM
image of a "cyanostar"
dendrimer
molecule.
[29]
Molecules have fixed
equilibrium
geometries?bond lengths and angles? about which they continuously oscillate through vibrational and rotational motions. A pure substance is composed of molecules with the same average geometrical structure. The chemical formula and the structure of a molecule are the two important factors that determine its properties, particularly its
reactivity
.
Isomers
share a chemical formula but normally have very different properties because of their different structures.
Stereoisomers
, a particular type of isomer, may have very similar physico-chemical properties and at the same time different
biochemical
activities.
Molecular spectroscopy
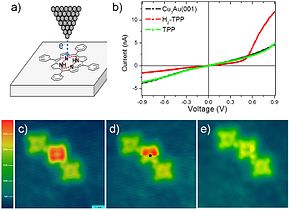 Hydrogen can be removed from individual
H
2
TPP
molecules by applying excess voltage to the tip of a
scanning tunneling microscope
(STM, a); this removal alters the current-voltage (I-V) curves of TPP molecules, measured using the same STM tip, from
diode
like (red curve in b) to
resistor
like (green curve). Image (c) shows a row of TPP, H
2
TPP and TPP molecules. While scanning image (d), excess voltage was applied to H
2
TPP at the black dot, which instantly removed hydrogen, as shown in the bottom part of (d) and in the rescan image (e). Such manipulations can be used in
single-molecule electronics
.
[30]
Hydrogen can be removed from individual
H
2
TPP
molecules by applying excess voltage to the tip of a
scanning tunneling microscope
(STM, a); this removal alters the current-voltage (I-V) curves of TPP molecules, measured using the same STM tip, from
diode
like (red curve in b) to
resistor
like (green curve). Image (c) shows a row of TPP, H
2
TPP and TPP molecules. While scanning image (d), excess voltage was applied to H
2
TPP at the black dot, which instantly removed hydrogen, as shown in the bottom part of (d) and in the rescan image (e). Such manipulations can be used in
single-molecule electronics
.
[30]
Molecular spectroscopy
deals with the response (
spectrum
) of molecules interacting with probing signals of known
energy
(or
frequency
, according to the
Planck relation
). Molecules have quantized energy levels that can be analyzed by detecting the molecule's energy exchange through
absorbance
or
emission
.
[31]
Spectroscopy does not generally refer to
diffraction
studies where particles such as
neutrons
, electrons, or high energy
X-rays
interact with a regular arrangement of molecules (as in a crystal).
Microwave spectroscopy
commonly measures changes in the rotation of molecules, and can be used to identify molecules in outer space.
Infrared spectroscopy
measures the vibration of molecules, including stretching, bending or twisting motions. It is commonly used to identify the kinds of bonds or
functional groups
in molecules. Changes in the arrangements of electrons yield absorption or emission lines in ultraviolet, visible or
near infrared
light, and result in colour.
Nuclear resonance spectroscopy
measures the environment of particular nuclei in the molecule, and can be used to characterise the numbers of atoms in different positions in a molecule.
Theoretical aspects
The study of molecules by molecular physics and
theoretical chemistry
is largely based on
quantum mechanics
and is essential for the understanding of the chemical bond. The simplest of molecules is the
hydrogen molecule-ion
, H
2
+
, and the simplest of all the chemical bonds is the
one-electron bond
. H
2
+
is composed of two positively charged
protons
and one negatively charged
electron
, which means that the
Schrodinger equation
for the system can be solved more easily due to the lack of electron?electron repulsion. With the development of fast digital computers, approximate solutions for more complicated molecules became possible and are one of the main aspects of
computational chemistry
.
When trying to define rigorously whether an arrangement of atoms is
sufficiently stable
to be considered a molecule, IUPAC suggests that it "must correspond to a depression on the
potential energy surface
that is deep enough to confine at least one vibrational state".
[4]
This definition does not depend on the nature of the interaction between the atoms, but only on the strength of the interaction. In fact, it includes weakly bound species that would not traditionally be considered molecules, such as the
helium
dimer
,
He
2
, which has one vibrational bound state
[32]
and is so loosely bound that it is only likely to be observed at very low temperatures.
Whether or not an arrangement of atoms is
sufficiently stable
to be considered a molecule is inherently an operational definition. Philosophically, therefore, a molecule is not a fundamental entity (in contrast, for instance, to an
elementary particle
); rather, the concept of a molecule is the chemist's way of making a useful statement about the strengths of atomic-scale interactions in the world that we observe.
See also
References
- ^
Iwata, Kota; Yamazaki, Shiro; Mutombo, Pingo; Hapala, Prokop; Ondra?ek, Martin; Jelinek, Pavel; Sugimoto, Yoshiaki (2015).
"Chemical structure imaging of a single molecule by atomic force microscopy at room temperature"
.
Nature Communications
.
6
: 7766.
Bibcode
:
2015NatCo...6.7766I
.
doi
:
10.1038/ncomms8766
.
PMC
4518281
.
PMID
26178193
.
- ^
Dinca, L.E.; De Marchi, F.; MacLeod, J.M.; Lipton-Duffin, J.; Gatti, R.; Ma, D.;
Perepichka, D.F.
; Rosei, F. (2015). "Pentacene on Ni(111): Room-temperature molecular packing and temperature-activated conversion to graphene".
Nanoscale
.
7
(7): 3263?9.
Bibcode
:
2015Nanos...7.3263D
.
doi
:
10.1039/C4NR07057G
.
PMID
25619890
.
- ^
Hapala, Prokop; ?vec, Martin; Stetsovych, Oleksandr; Van Der Heijden, Nadine J.; Ondra?ek, Martin; Van Der Lit, Joost; Mutombo, Pingo; Swart, Ingmar; Jelinek, Pavel (2016).
"Mapping the electrostatic force field of single molecules from high-resolution scanning probe images"
.
Nature Communications
.
7
: 11560.
Bibcode
:
2016NatCo...711560H
.
doi
:
10.1038/ncomms11560
.
PMC
4894979
.
PMID
27230940
.
- ^
a
b
IUPAC
,
Compendium of Chemical Terminology
, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
Molecule
".
doi
:
10.1351/goldbook.M04002
- ^
Ebbin, Darrell D. (1990).
General Chemistry
(3rd ed.). Boston:
Houghton Mifflin Co.
ISBN
978-0-395-43302-7
.
- ^
Brown, T.L.; Kenneth C. Kemp; Theodore L. Brown; Harold Eugene LeMay; Bruce Edward Bursten (2003).
Chemistry ? the Central Science
(9th ed.). New Jersey:
Prentice Hall
.
ISBN
978-0-13-066997-1
.
- ^
Chang, Raymond (1998).
Chemistry
(6th ed.). New York:
McGraw Hill
.
ISBN
978-0-07-115221-1
.
- ^
Zumdahl, Steven S. (1997).
Chemistry
(4th ed.). Boston: Houghton Mifflin.
ISBN
978-0-669-41794-4
.
- ^
Chandra, Sulekh (2005).
Comprehensive Inorganic Chemistry
. New Age Publishers.
ISBN
978-81-224-1512-4
.
- ^
"Molecule"
.
Encyclopædia Britannica
. 22 January 2016.
Archived
from the original on 3 May 2020
. Retrieved
23 February
2016
.
- ^
Harper, Douglas.
"molecule"
.
Online Etymology Dictionary
. Retrieved
22 February
2016
.
- ^
"molecule"
.
Merriam-Webster
.
Archived
from the original on 24 February 2021
. Retrieved
22 February
2016
.
- ^
Molecule Definition
Archived
13 October 2014 at the
Wayback Machine
(
Frostburg State University
)
- ^
Ley, Willy (June 1966).
"The Re-Designed Solar System"
. For Your Information.
Galaxy Science Fiction
. pp. 94?106.
- ^
Avogadro, Amedeo (1811).
"Masses of the Elementary Molecules of Bodies"
.
Journal de Physique
.
73
: 58?76.
Archived
from the original on 12 May 2019
. Retrieved
25 August
2022
.
- ^
Seymour H. Mauskopf (1969). "The Atomic Structural Theories of Ampere and Gaudin: Molecular Speculation and Avogadro's Hypothesis".
Isis
.
60
(1): 61?74.
doi
:
10.1086/350449
.
JSTOR
229022
.
S2CID
143759556
.
- ^
Perrin, Jean, B. (1926).
Discontinuous Structure of Matter
Archived
29 May 2019 at the
Wayback Machine
, Nobel Lecture, 11 December.
- ^
Heitler, Walter; London, Fritz (1927). "Wechselwirkung neutraler Atome und homoopolare Bindung nach der Quantenmechanik".
Zeitschrift fur Physik
.
44
(6?7): 455?472.
Bibcode
:
1927ZPhy...44..455H
.
doi
:
10.1007/BF01397394
.
S2CID
119739102
.
- ^
Pauling, Linus (1931). "The nature of the chemical bond. Application of results obtained from the quantum mechanics and from a theory of paramagnetic susceptibility to the structure of molecules".
J. Am. Chem. Soc
.
53
(4): 1367?1400.
doi
:
10.1021/ja01355a027
.
- ^
Harry, B. Gray.
Chemical Bonds: An Introduction to Atomic and Molecular Structure
(PDF)
. pp. 210?211.
Archived
(PDF)
from the original on 31 March 2021
. Retrieved
22 November
2021
.
- ^
"How many gold atoms make gold metal?"
.
phys.org
.
Archived
from the original on 30 October 2020
. Retrieved
22 November
2021
.
- ^
Campbell, Neil A.; Brad Williamson; Robin J. Heyden (2006).
Biology: Exploring Life
. Boston:
Pearson Prentice Hall
.
ISBN
978-0-13-250882-7
.
Archived
from the original on 2 November 2014
. Retrieved
5 February
2012
.
- ^
Campbell, Flake C. (2008).
Elements of Metallurgy and Engineering Alloys
.
ASM International
.
ISBN
978-1-61503-058-3
.
Archived
from the original on 31 March 2021
. Retrieved
27 October
2020
.
- ^
Roger L. DeKock; Harry B. Gray; Harry B. Gray (1989).
Chemical structure and bonding
. University Science Books. p. 199.
ISBN
978-0-935702-61-3
.
Archived
from the original on 31 March 2021
. Retrieved
27 October
2020
.
- ^
Chang RL; Deen WM; Robertson CR; Brenner BM (1975).
"Permselectivity of the glomerular capillary wall: III. Restricted transport of polyanions"
.
Kidney Int
.
8
(4): 212?218.
doi
:
10.1038/ki.1975.104
.
PMID
1202253
.
- ^
Chang RL; Ueki IF; Troy JL; Deen WM; Robertson CR; Brenner BM (1975).
"Permselectivity of the glomerular capillary wall to macromolecules. II. Experimental studies in rats using neutral dextran"
.
Biophys. J
.
15
(9): 887?906.
Bibcode
:
1975BpJ....15..887C
.
doi
:
10.1016/S0006-3495(75)85863-2
.
PMC
1334749
.
PMID
1182263
.
- ^
Wink, Donald J.; Fetzer-Gislason, Sharon; McNicholas, Sheila (2003).
The Practice of Chemistry
. Macmillan.
ISBN
978-0-7167-4871-7
.
Archived
from the original on 10 April 2022
. Retrieved
27 October
2020
.
- ^
"ChemTeam: Empirical Formula"
.
www.chemteam.info
.
Archived
from the original on 19 January 2021
. Retrieved
16 April
2017
.
- ^
Hirsch, Brandon E.; Lee, Semin; Qiao, Bo; Chen, Chun-Hsing; McDonald, Kevin P.; Tait, Steven L.; Flood, Amar H. (2014).
"Anion-induced dimerization of 5-fold symmetric cyanostars in 3D crystalline solids and 2D self-assembled crystals"
.
Chemical Communications
.
50
(69): 9827?30.
doi
:
10.1039/C4CC03725A
.
PMID
25080328
.
S2CID
12439952
.
Archived
from the original on 31 March 2021
. Retrieved
20 April
2018
.
- ^
Zoldan, V. C.; Faccio, R; Pasa, A.A. (2015).
"N and p type character of single molecule diodes"
.
Scientific Reports
.
5
: 8350.
Bibcode
:
2015NatSR...5E8350Z
.
doi
:
10.1038/srep08350
.
PMC
4322354
.
PMID
25666850
.
- ^
IUPAC
,
Compendium of Chemical Terminology
, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
Spectroscopy
".
doi
:
10.1351/goldbook.S05848
- ^
Anderson JB (May 2004).
"Comment on "An exact quantum Monte Carlo calculation of the helium-helium intermolecular potential" [J. Chem. Phys. 115, 4546 (2001)]"
.
J Chem Phys
.
120
(20): 9886?7.
Bibcode
:
2004JChPh.120.9886A
.
doi
:
10.1063/1.1704638
.
PMID
15268005
.
External links