Group of natural pigments found in most organisms
| Melanin
|
|---|
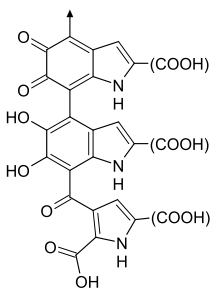 One possible structure of Eumelanin
|
| Material type
| Heterogeneous biopolymer
|
|---|
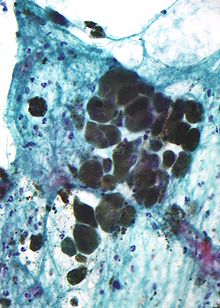 Micrograph of Melanin pigment (light refracting granular material?center of image) in a pigmented
melanoma
.
Micrograph of Melanin pigment (light refracting granular material?center of image) in a pigmented
melanoma
.
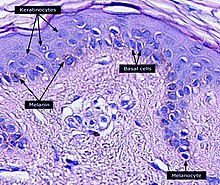 Micrograph of the epidermis, with melanin labeled at left.
Micrograph of the epidermis, with melanin labeled at left.
Melanin
(
; from
Ancient Greek
μ?λα?
(
melas
)
'black, dark') is a family of
biomolecules
organized as
oligomers
or
polymers
, which among other functions provide the
pigments
of many
organisms
.
[1]
Melanin pigments are produced in a specialized group of cells known as
melanocytes
.
There are five basic types of melanin:
eumelanin
,
pheomelanin
,
neuromelanin
,
allomelanin
and
pyomelanin
.
[2]
Eumelanin is produced through a multistage chemical process known as
melanogenesis
, where the
oxidation
of the
amino acid
tyrosine
is followed by
polymerization
. Eumelanin is the most common type. Pheomelanin, which is produced when melanocytes are malfunctioning due to derivation of the gene to its recessive format, is a
cysteine
-derivative that contains poly
benzothiazine
portions that are largely responsible for the
red
or
yellow
tint given to some skin or hair colors. Neuromelanin is found in the brain. Research has been undertaken to investigate its efficacy in treating neurodegenerative disorders such as
Parkinson's
.
[3]
Allomelanin and pyomelanin are two types of nitrogen-free melanin.
In the human skin, melanogenesis is initiated by exposure to
UV radiation
, causing the skin to darken. Eumelanin is an effective absorbent of light; the pigment is able to dissipate over 99.9% of absorbed UV radiation.
[4]
Because of this property, eumelanin is thought to protect skin cells from UVA and UVB radiation damage, reducing the risk of folate depletion and dermal degradation. Exposure to UV radiation is associated with increased risk of
malignant melanoma
, a cancer of melanocytes (melanin cells). Studies have shown a lower incidence for skin cancer in individuals with more concentrated melanin, i.e. darker
skin tone
.
[5]
Melanin types
[
edit
]
Eumelanin
[
edit
]
 Part of the structural formula of eumelanin. "(COOH)" can be COOH or H, or (more rarely) other
substituents
. The arrow denotes where the polymer continues.
Part of the structural formula of eumelanin. "(COOH)" can be COOH or H, or (more rarely) other
substituents
. The arrow denotes where the polymer continues.
Eumelanin has two forms linked to
5,6-dihydroxyindole
(DHI) and
5,6-dihydroxyindole-2-carboxylic acid
(DHICA). DHI-derived eumelanin is dark brown or black and insoluble, and DHICA -derived eumelanin which is lighter and soluble in alkali. Both eumelanins arise from the oxidation of tyrosine in specialized organelles called
melanosomes
. This reaction is catalyzed by the enzyme
tyrosinase
. The initial product,
dopaquinone
can transform into either 5,6-dihydroxyindole (DHI) or 5,6-dihydroxyindole-2-carboxylic acid (DHICA). DHI and DHICA are oxidized and then polymerize to form the two eumelanins.
[6]
In natural conditions, DHI and DHICA often co-polymerize, resulting in a range of eumelanin polymers. These polymers contribute to the variety of melanin components in human skin and hair, ranging from light yellow/red pheomelanin to light brown DHICA-enriched eumelanin and dark brown or black DHI-enriched eumelanin. These final polymers differ in solubility and color.
[6]
Analysis of highly pigmented (
Fitzpatrick type
V and VI) skin finds that DHI-eumelanin comprises the largest portion, approximately 60?70%, followed by DHICA-eumelanin at 25?35%, and pheomelanin only 2?8%. Notably, while an enrichment of DHI-eumelanin occurs in during
sun tanning
, it is accompanied by a decrease in DHICA-eumelanin and pheomelanin.
[6]
A small amount of black eumelanin in the absence of other pigments causes grey hair. A small amount of eumelanin in the absence of other pigments causes blond hair.
[7]
Eumelanin is present in the skin and hair, etc.
Pheomelanin
[
edit
]
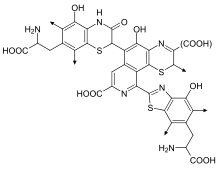 Part of the structural formula of pheomelanin. "(COOH)" can be COOH or H, or (more rarely) other
substituents
. The arrows denote where the polymer continues.
Part of the structural formula of pheomelanin. "(COOH)" can be COOH or H, or (more rarely) other
substituents
. The arrows denote where the polymer continues.
Pheomelanins (or phaeomelanins) impart a range of yellowish to reddish colors.
[8]
Pheomelanins are particularly concentrated in the lips, nipples, glans of the penis, and vagina.
[9]
When a small amount of eumelanin in hair (which would otherwise cause blond hair) is mixed with pheomelanin, the result is orange hair, which is typically called
"red" or "ginger" hair
. Pheomelanin is also present in the skin, and redheads consequently often have a more pinkish hue to their skin as well. Exposure of the skin to ultraviolet light increases pheomelanin content, as it does for eumelanin; but rather than absorbing light, pheomelanin within the hair and skin reflect yellow to red light, which may increase damage from UV radiation exposure.
[10]
Pheomelanin production is highly dependent on
cysteine
availability, which is transported into the melanosome, reacting with dopaquinone to form cys-dopa. Cys-dopa then undergoes several transformations before forming pheomelanin.
[6]
In chemical terms, pheomelanins differ from eumelanins in that the oligomer structure incorporates
benzothiazine
and
benzothiazole
units that are produced,
[11]
instead of DHI and
DHICA
, when the amino acid
L-cysteine
is present.
Neuromelanin
[
edit
]
Neuromelanin (NM) is an insoluble polymer pigment produced in specific populations of
catecholaminergic neurons
in the brain. Humans have the largest amount of NM, which is present in lesser amounts in other primates, and totally absent in many other species.
[12]
The biological function remains unknown, although human NM has been shown to efficiently bind
transition metals
such as iron, as well as other potentially toxic molecules. Therefore, it may play crucial roles in
apoptosis
and the related
Parkinson's disease
.
[13]
Other forms of melanins
[
edit
]
Up until the 1960s, melanin was classified into eumelanin and pheomelanin. However in 1955 a melanin associated with nerve cells was discovered, neuromelanin. In 1972 a water-soluble form, pyomelanin was discovered. In 1976, allomelanin, the fifth form of the melanins was found in nature.
[2]
Selenomelanin
[
edit
]
It is possible to enrich melanin with
selenium
instead of
sulphur
. This selenium analogue of pheomelanin has been successfully synthesized through chemical and biosynthetic routes using selenocystine as a feedstock.
[14]
Due selenium's higher atomic number, the obtained selenomelanin can be expected to provide better protection against ionising radiation as compared to the other known forms of melanin. This protection has been demonstrated with radiation experiments on human cells and bacteria, opening up the possibility of applications in space travel.
[15]
Trichochromes
[
edit
]
Trichochromes (formerly called trichosiderins) are pigments produced from the same
metabolic pathway
as the eumelanins and pheomelanins, but unlike those molecules they have low molecular weight. They occur in some red human hair.
[16]
Humans
[
edit
]
 Albinism
occurs when melanocytes produce little melanin. This albino girl is from
Papua New Guinea
.
Albinism
occurs when melanocytes produce little melanin. This albino girl is from
Papua New Guinea
.
In humans, melanin is the primary determinant of
skin color
. It is also found in hair, the pigmented tissue underlying the
iris
of the eye, and the
stria vascularis
of the
inner ear
. In the brain, tissues with melanin include the
medulla
and pigment-bearing neurons within areas of the
brainstem
, such as the
locus coeruleus
. It also occurs in the
zona reticularis
of the
adrenal gland
.
[17]
The melanin in the skin is produced by
melanocytes
, which are found in the
basal layer
of the
epidermis
. Although, in general, human beings possess a similar concentration of melanocytes in their skin, the melanocytes in some individuals and ethnic groups produce variable amounts of melanin. Some humans have very little or no melanin synthesis in their bodies, a condition known as
albinism
.
[18]
Because melanin is an aggregate of smaller component molecules, there are many different types of melanin with different proportions and bonding patterns of these component molecules. Both pheomelanin and eumelanin are found in human skin and hair, but eumelanin is the most abundant melanin in humans, as well as the form most likely to be deficient in albinism.
[19]
Other organisms
[
edit
]
Melanins have very diverse roles and functions in various organisms. A form of melanin makes up the ink used by many
cephalopods
(see
cephalopod ink
) as a defense mechanism against predators. Melanins also protect microorganisms, such as bacteria and fungi, against stresses that involve cell damage such as
UV radiation
from the sun and
reactive oxygen species
. Melanin also protects against damage from high temperatures, chemical stresses (such as
heavy metals
and
oxidizing agents
), and biochemical threats (such as host defenses against invading microbes).
[20]
Therefore, in many pathogenic microbes (for example, in
Cryptococcus neoformans
, a fungus) melanins appear to play important roles in
virulence
and
pathogenicity
by protecting the microbe against
immune
responses of its
host
. In invertebrates, a major aspect of the innate immune defense system against invading pathogens involves melanin. Within minutes after infection, the microbe is encapsulated within melanin (melanization), and the generation of free radical byproducts during the formation of this capsule is thought to aid in killing them.
[21]
Some types of fungi, called
radiotrophic fungi
, appear to be able to use melanin as a
photosynthetic pigment
that enables them to capture
gamma rays
[22]
and harness this energy for growth.
[23]
In
fish
, melanin occurs not only in the skin but also in internal organs such as eyes. Most fish species use eumelanin,
[24]
[25]
but
Stegastes apicalis
and
Cyprinus carpio
use pheomelanin instead.
[26]
[27]
The darker
feathers
of birds owe their color to melanin and are less readily degraded by bacteria than unpigmented ones or those containing
carotenoid
pigments.
[28]
Feathers that contain melanin are also 39% more resistant to abrasion than those that do not because melanin granules help fill the space between the
keratin
strands that form feathers.
[29]
[30]
Pheomelanin synthesis in birds implies the consumption of cysteine, a semi?essential amino acid that is necessary for the synthesis of the antioxidant glutathione (GSH) but that may be toxic if in excess in the diet. Indeed, many carnivorous birds, which have a high protein content in their diet, exhibit pheomelanin?based coloration.
[31]
Melanin is also important in
mammalian
pigmentation.
[32]
The coat pattern of mammals is determined by the
agouti gene
which regulates the distribution of melanin.
[33]
[34]
The mechanisms of the gene have been extensively studied in mice to provide an insight into the diversity of mammalian coat patterns.
[35]
Melanin in
arthropods
has been observed to be deposited in layers thus producing a
Bragg reflector
of alternating refractive index. When the scale of this pattern matches the wavelength of visible light,
structural coloration
arises: giving a number of species an
iridescent
color.
[36]
[37]
Arachnids
are one of the few groups in which melanin has not been easily detected, though researchers found data suggesting spiders do in fact produce melanin.
[38]
Some moth species, including the
wood tiger moth
, convert resources to melanin to enhance their thermoregulation. As the wood tiger moth has populations over a large range of latitudes, it has been observed that more northern populations showed higher rates of melanization. In both yellow and white male phenotypes of the wood tiger moth, individuals with more melanin had a heightened ability to trap heat but an increased predation rate due to a weaker and less effective
aposematic
signal.
[39]
Melanin protects
Drosophila
flies and
mice
against DNA damage from non-UV radiation.
[40]
Important studies in Drosophila models include Hopwood
et al.
, 1985.
[40]
Much of our understanding of the
radioprotective
effects of melanin against
gamma radiation
come from the laboratories and research groups of Irma Mosse.
[41]
[42]
[43]
[44]
[45]
[46]
[47]
: 1151
Mosse began in
radiobiology
in the Soviet era, was increasingly supported by government funding in the wake of the discovery of
radiotrophic
microbes in
Chernobyl
, and as of 2022
[update]
continues under the
Belarusian Institute of Genetics and Cytology
.
[46]
Her most significant contribution is Mosse
et al.
, 2000 on mice
[41]
[42]
[43]
[44]
[45]
[46]
[47]
: 1151
but also includes Mosse
et al.
, 1994,
[45]
Mosse
et al.
, 1997,
[45]
Mosse
et al.
, 1998,
[44]
Mosse
et al.
, 2001,
[45]
Mosse
et al.
, 2002,
[44]
[45]
Mosse
et al.
, 2006,
[44]
[45]
Mosse
et al.
, 2007
[45]
and Mosse
et al.
, 2008.
[45]
Plants
[
edit
]
 Chemical structure of indole-5,6-quinone
Chemical structure of indole-5,6-quinone
Melanin produced by plants are sometimes referred to as 'catechol melanins' as they can yield
catechol
on alkali fusion. It is commonly seen in the
enzymatic browning
of fruits such as bananas. Chestnut shell melanin can be used as an antioxidant and coloring agent.
[48]
Biosynthesis involves the oxidation of
indole-5,6-quinone
by the tyrosinase type
polyphenol oxidase
from
tyrosine
and
catecholamines
leading to the formation of catechol melanin. Despite this many plants contain compounds which inhibit the production of melanins.
[49]
Interpretation as a single monomer
[
edit
]
It is now understood that melanins do not have a single structure or stoichiometry.
[
citation needed
]
Nonetheless, chemical databases such as PubChem include structural and empirical formulae; typically
3,8-Dimethyl-2,7-dihydrobenzo[1,2,3-
cd
:4,5,6-
c
′
d
′]diindole-4,5,9,10-tetrone
. This can be thought of as a single monomer that accounts for the measured elemental composition and some properties of melanin, but is unlikely to be found in nature.
[50]
Solano
[50]
claims that this misleading trend stems from a report of an empirical formula in 1948,
[51]
but provides no other historical detail.
3,8-Dimethyl-2,7-dihydrobenzo[1,2,3-
cd
:4,5,6-
c
′
d
′]diindole-4,5,9,10-tetrone
![3,8-Dimethyl-2,7-dihydrobenzo[1,2,3-cd:4,5,6-c′d′]diindole-4,5,9,10-tetrone](//upload.wikimedia.org/wikipedia/commons/thumb/2/20/Melanin.svg/240px-Melanin.svg.png)
|
![3,8-Dimethyl-2,7-dihydrobenzo[1,2,3-cd:4,5,6-c′d′]diindole-4,5,9,10-tetrone ball and stick model](//upload.wikimedia.org/wikipedia/commons/thumb/7/7b/Melanin_ball_and_stick.png/240px-Melanin_ball_and_stick.png)
|
| Names
|
Preferred IUPAC name
3,8-Dimethyl-2,7-dihydrobenzo[1,2,3-
cd
:4,5,6-
c
′
d
′]diindole-4,5,9,10-tetrone
|
| Identifiers
|
|
|
|
| ChemSpider
|
|
|
|
|
| Properties
|
|
|
C
18
H
10
N
2
O
4
|
| Molar mass
|
318.288
g·mol
?1
|
| Density
|
1.6 to 1.8 g/cm
3
|
| Melting point
|
< ?20 °C (?4 °F; 253 K)
|
| Boiling point
|
450 to 550 °C (842 to 1,022 °F; 723 to 823 K)
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25 °C [77 °F], 100 kPa).
|
Chemical compound
Biosynthetic pathways
[
edit
]
The first step of the biosynthetic pathway for both eumelanins and pheomelanins is
catalysed
by
tyrosinase
.
[52]
- Tyrosine
→
DOPA
→
dopaquinone
Dopaquinone can combine with
cysteine
by two pathways to benzothiazines and pheomelanins
- dopaquinone + cysteine → 5-S-cysteinyldopa → benzothiazine intermediate → pheomelanin
- dopaquinone + cysteine → 2-S-cysteinyldopa → benzothiazine intermediate → pheomelanin
Also, dopaquinone can be converted to
leucodopachrome
and follow two more pathways to the eumelanins
- dopaquinone → leucodopachrome →
dopachrome
→ 5,6-dihydroxyindole-2-carboxylic acid → quinone → eumelanin
- dopaquinone → leucodopachrome → dopachrome → 5,6-dihydroxyindole →
quinone
→ eumelanin
Detailed metabolic pathways can be found in the
KEGG
database (see
External links
).
-
L-tyrosine
-
L-DOPA
-
L-dopaquinone
-
L-leucodopachrome
-
L-dopachrome
Microscopic appearance
[
edit
]
Melanin is brown, non-refractile, and finely granular with individual granules having a diameter of less than 800 nanometers. This differentiates melanin from common
blood breakdown pigments
, which are larger, chunky, and refractile, and range in color from green to yellow or red-brown. In heavily pigmented lesions, dense aggregates of melanin can obscure histologic detail. A dilute solution of
potassium permanganate
is an effective melanin bleach.
[53]
Genetic disorders and disease states
[
edit
]
There are approximately nine types of
oculocutaneous albinism
, which is mostly an autosomal recessive disorder. Certain ethnicities have higher incidences of different forms. For example, the most common type, called oculocutaneous albinism type 2 (OCA2), is especially frequent among people of black African descent and white Europeans. People with OCA2 usually have fair skin, but are often not as pale as OCA1. They (OCA2 or OCA1? see comments in History) have pale blonde to golden, strawberry blonde, or even brown hair, and most commonly blue eyes. 98.7?100% of modern Europeans are carriers of the derived allele
SLC24A5
, a known cause of nonsyndromic oculocutaneous albinism. It is an autosomal recessive disorder characterized by a
congenital
reduction or absence of melanin pigment in the skin, hair, and eyes. The estimated frequency of OCA2 among African-Americans is 1 in 10,000, which contrasts with a frequency of 1 in 36,000 in white Americans.
[54]
In some African nations, the frequency of the disorder is even higher, ranging from 1 in 2,000 to 1 in 5,000.
[55]
Another form of Albinism, the "yellow oculocutaneous albinism", appears to be more prevalent among the
Amish
, who are of primarily Swiss and German ancestry. People with this IB variant of the disorder commonly have white hair and skin at birth, but rapidly develop normal skin pigmentation in infancy.
[55]
Ocular albinism affects not only eye pigmentation but visual acuity, as well. People with albinism typically test poorly, within the 20/60 to 20/400 range. In addition, two forms of albinism, with approximately 1 in 2,700 most prevalent among people of Puerto Rican origin, are associated with mortality beyond melanoma-related deaths.
The connection between albinism and
deafness
is well known, though poorly understood. In his 1859 treatise
On the Origin of Species
,
Charles Darwin
observed that "cats which are entirely white and have blue eyes are generally deaf".
[56]
In humans, hypopigmentation and deafness occur together in the rare
Waardenburg's syndrome
, predominantly observed among the
Hopi
in North America.
[57]
The incidence of albinism in Hopi Indians has been estimated as approximately 1 in 200 individuals. Similar patterns of albinism and deafness have been found in other mammals, including dogs and rodents. However, a lack of melanin
per se
does not appear to be directly responsible for deafness associated with hypopigmentation, as most individuals lacking the enzymes required to synthesize melanin have normal auditory function.
[58]
Instead, the absence of
melanocytes
in the stria vascularis of the inner ear results in
cochlear
impairment,
[59]
though the reasons for this are not fully understood.
In Parkinson's disease, a disorder that affects
neuromotor
functioning, there is decreased neuromelanin in the substantia nigra and locus coeruleus as a consequence of specific dropping out of dopaminergic and noradrenergic pigmented neurons. This results in diminished
dopamine
and
norepinephrine
synthesis. While no correlation between race and the level of neuromelanin in the substantia nigra has been reported, the significantly lower incidence of Parkinson's in blacks than in whites has "prompt[ed] some to suggest that cutaneous melanin might somehow serve to protect the neuromelanin in substantia nigra from external toxins."
[60]
In addition to melanin deficiency, the molecular weight of the melanin polymer may be decreased by various factors such as oxidative stress, exposure to light, perturbation in its association with melanosomal
matrix proteins
, changes in
pH
, or in local concentrations of metal ions. A decreased molecular weight or a decrease in the degree of polymerization of
ocular melanin
has been proposed to turn the normally anti-oxidant polymer into a
pro-oxidant
. In its pro-oxidant state, melanin has been suggested to be involved in the causation and progression of
macular degeneration
and
melanoma
.
[61]
Rasagiline
, an important monotherapy drug in Parkinson's disease, has melanin binding properties, and melanoma tumor reducing properties.
[62]
Higher eumelanin levels also can be a disadvantage, however, beyond a higher disposition toward vitamin D deficiency. Dark skin is a complicating factor in the laser removal of
port-wine stains
. Effective in treating white skin, in general, lasers are less successful in removing port-wine stains in people of Asian or African descent. Higher concentrations of melanin in darker-skinned individuals simply diffuse and absorb the laser radiation, inhibiting light absorption by the targeted tissue. In a similar manner, melanin can complicate laser treatment of other dermatological conditions in people with darker skin.
Freckles
and
moles
are formed where there is a localized concentration of melanin in the skin. They are highly associated with pale skin.
Nicotine
has an affinity for melanin-containing tissues because of its precursor function in melanin synthesis or its irreversible binding of melanin. This has been suggested to underlie the increased
nicotine dependence
and lower
smoking cessation
rates in darker pigmented individuals.
[63]
Human adaptations
[
edit
]
Physiology
[
edit
]
Melanocytes insert granules of melanin into specialized cellular
vesicles
called
melanosomes
. These are then transferred into the
keratinocyte
cells of the human
epidermis
. The melanosomes in each recipient cell accumulate atop the
cell nucleus
, where they protect the nuclear
DNA
from mutations caused by the
ionizing radiation
of the sun's
ultraviolet
rays. In general, people whose ancestors lived for long periods in the regions of the globe near the
equator
have larger quantities of eumelanin in their skins. This makes their skins brown or black and protects them against high levels of exposure to the sun, which more frequently result in
melanomas
in lighter-skinned people.
[64]
Not all the effects of pigmentation are advantageous. Pigmentation increases the heat load in hot climates, and dark-skinned people absorb 30% more heat from sunlight than do very light-skinned people, although this factor may be offset by more profuse sweating. In cold climates dark skin entails more heat loss by radiation. Pigmentation also hinders synthesis of
vitamin D
. Since pigmentation appears to be not entirely advantageous to life in the tropics, other hypotheses about its biological significance have been advanced; for example a secondary phenomenon induced by adaptation to parasites and tropical diseases.
[65]
Evolutionary origins
[
edit
]
Early humans
evolved dark skin color, as an adaptation to a loss of body hair that increased the effects of UV radiation. Before the development of hairlessness, early humans might have had light skin underneath their fur, similar to that found in other
primates
.
[66]
Anatomically modern humans
evolved in Africa between 200,000 and 100,000 years ago,
[67]
and then populated the rest of the world through migration between 80,000 and 50,000 years ago, in some areas
interbreeding
with certain
archaic human
species (
Neanderthals
,
Denisovans
, and possibly others).
[68]
The first modern humans had darker skin as the indigenous people of Africa today. Following migration and settlement in Asia and Europe, the selective pressure dark UV-radiation protecting skin decreased where radiation from the sun was less intense. This resulted in the current range of human skin color. Of the two common gene variants known to be associated with pale human skin,
Mc1r
does not appear to have undergone positive selection,
[69]
while
SLC24A5
has undergone positive selection.
[70]
Effects
[
edit
]
As with peoples having migrated northward, those with light skin migrating toward the equator acclimatize to the much stronger solar radiation. Nature selects for less melanin when ultraviolet radiation is weak. Most people's skin darkens when exposed to UV light, giving them more protection when it is needed. This is the physiological purpose of
sun tanning
. Dark-skinned people, who produce more skin-protecting eumelanin, have a greater protection against
sunburn
and the development of melanoma, a potentially deadly form of skin cancer, as well as other health problems related to exposure to strong
solar radiation
, including the
photodegradation
of certain
vitamins
such as
riboflavins
,
carotenoids
,
tocopherol
, and
folate
.
[71]
Melanin in the eyes, in the
iris
and
choroid
, helps protect from
ultraviolet
and
high-frequency visible light
; people with
blue, green, and grey eyes
are more at risk of sun-related eye problems. Furthermore, the ocular lens yellows with age, providing added protection. However, the lens also becomes more rigid with age, losing most of its
accommodation
?the ability to change shape to focus from far to near?a detriment due probably to
protein
crosslinking caused by UV exposure.
Recent research suggests that melanin may serve a protective role other than photoprotection.
[72]
Melanin is able to effectively
chelate
metal ions through its carboxylate and phenolic hydroxyl groups, often much more efficiently than the powerful chelating ligand ethylenediaminetetraacetate (EDTA). Thus, it may serve to sequester potentially toxic metal ions, protecting the rest of the cell. This hypothesis is supported by the fact that the loss of neuromelanin, observed in Parkinson's disease, is accompanied by an increase in iron levels in the brain.
Physical properties and technological applications
[
edit
]
Evidence exists for a highly cross-linked
heteropolymer
bound
covalently
to matrix scaffolding
melanoproteins
.
[73]
It has been proposed that the ability of melanin to act as an
antioxidant
is directly proportional to its degree of polymerization or
molecular weight
.
[74]
Suboptimal conditions for the effective polymerization of melanin
monomers
may lead to formation of pro-oxidant melanin with lower-molecular-weight, implicated in the causation and progression of
macular degeneration
and
melanoma
.
[75]
Signaling pathways
that
upregulate
melanization in the
retinal pigment epithelium
(RPE) also may be implicated in the
downregulation
of
rod
outer segment
phagocytosis
by the RPE. This phenomenon has been attributed in part to
foveal
sparing in
macular degeneration
.
[76]
Role in melanoma metastasis
[
edit
]
Heavily pigmented melanoma cells have a
Young's modulus
of about 4.93 kPa, compared to non-pigmented cells, with a value of 0.98 kPa.
[77]
The
elasticity
of melanoma cells is crucial to metastasis and growth; non-pigmented tumors were larger than pigmented tumors, and spread far more easily. Pigmented and non-pigmented cells are both present in melanoma
tumors
, so that they can both be
drug-resistant
and metastatic.
[77]
See also
[
edit
]
References
[
edit
]
- ^
Casadevall, Arturo (2018). "Melanin triggers antifungal defences".
Nature
.
555
(7696): 319?320.
Bibcode
:
2018Natur.555..319C
.
doi
:
10.1038/d41586-018-02370-x
.
ISSN
0028-0836
.
PMID
29542711
.
S2CID
3832753
.
- ^
a
b
Cao, Wei; Zhou, Xuhao; McCallum, Naneki C.; Hu, Ziying; Ni, Qing Zhe; Kapoor, Utkarsh; Heil, Christian M.; Cay, Kristine S.; Zand, Tara; Mantanona, Alex J.; Jayaraman, Arthi (9 February 2021).
"Unraveling the Structure and Function of Melanin through Synthesis"
.
Journal of the American Chemical Society
.
143
(7): 2622?2637.
doi
:
10.1021/jacs.0c12322
.
hdl
:
1854/LU-8699336
.
ISSN
0002-7863
.
PMID
33560127
.
S2CID
231872855
.
- ^
Haining, Robert L.; Achat-Mendes, Cindy (March 2017).
"Neuromelanin, one of the most overlooked molecules in modern medicine, is not a spectator"
.
Neural Regeneration Research
.
12
(3): 372?375.
doi
:
10.4103/1673-5374.202928
.
PMC
5399705
.
PMID
28469642
.
- ^
Meredith P, Riesz J (2004). "Radiative relaxation quantum yields for synthetic eumelanin".
Photochemistry and Photobiology
.
79
(2): 211?6.
arXiv
:
cond-mat/0312277
.
doi
:
10.1111/j.1751-1097.2004.tb00012.x
.
PMID
15068035
.
S2CID
222101966
.
- ^
Brenner M, Hearing VJ (2008).
"The protective role of melanin against UV damage in human skin"
.
Photochemistry and Photobiology
.
84
(3): 539?49.
doi
:
10.1111/j.1751-1097.2007.00226.x
.
PMC
2671032
.
PMID
18435612
.
- ^
a
b
c
d
Alaluf, Simon; Heath, Alan; Carter, Nik; Atkins, Derek; Mahalingam, Harish; Barrett, Karen; Kolb, Ria; Smit, Nico (2001). "Variation in Melanin Content and Composition in Type V and VI Photoexposed and Photoprotected Human Skin: The Dominant Role of DHI".
Pigment Cell Research
.
14
(5): 337?347.
doi
:
10.1034/j.1600-0749.2001.140505.x
.
ISSN
0893-5785
.
PMID
11601655
.
- ^
Ito, S.; Wakamatsu, K. (December 2011). "Diversity of human hair pigmentation as studied by chemical analysis of eumelanin and pheomelanin".
Journal of the European Academy of Dermatology and Venereology
.
25
(12): 1369?1380.
doi
:
10.1111/j.1468-3083.2011.04278.x
.
ISSN
1468-3083
.
PMID
22077870
.
S2CID
5121042
.
- ^
Slominski A, Tobin DJ, Shibahara S, Wortsman J (October 2004). "Melanin pigmentation in mammalian skin and its hormonal regulation".
Physiological Reviews
.
84
(4): 1155?228.
doi
:
10.1152/physrev.00044.2003
.
PMID
15383650
.
S2CID
21168932
.
- ^
"pheomelanin"
.
MetaCyc Metabolic Pathway Database
. 2010.
[
full citation needed
]
- ^
Thody, A. J.; Higgins, E. M.; Wakamatsu, K.; Ito, S.; Burchill, S. A.; Marks, J. M. (August 1991).
"Pheomelanin as well as eumelanin is present in human epidermis"
.
The Journal of Investigative Dermatology
.
97
(2): 340?344.
doi
:
10.1111/1523-1747.ep12480680
.
PMID
2071942
.
- ^
Greco G, Panzella L, Verotta L, d'Ischia M, Napolitano A (April 2011). "Uncovering the structure of human red hair pheomelanin: benzothiazolylthiazinodihydroisoquinolines as key building blocks".
Journal of Natural Products
.
74
(4): 675?82.
doi
:
10.1021/np100740n
.
PMID
21341762
.
- ^
Fedorow H, Tribl F, Halliday G, Gerlach M, Riederer P, Double KL (2005). "Neuromelanin in human dopamine neurons: comparison with peripheral melanins and relevance to Parkinson's disease".
Prog Neurobiol
.
75
(2): 109?124.
doi
:
10.1016/j.pneurobio.2005.02.001
.
PMID
15784302
.
S2CID
503902
.
- ^
Double KL (2006). "Functional effects of neuromelanin and synthetic melanin in model systems".
J Neural Transm
.
113
(6): 751?756.
doi
:
10.1007/s00702-006-0450-5
.
PMID
16755379
.
S2CID
23096297
.
- ^
Wei Cao; et al. (2020). "Selenomelanin: An Abiotic Selenium Analogue of Pheomelanin".
Journal of the American Chemical Society
.
142
(29): 12802?12810.
doi
:
10.1021/jacs.0c05573
.
PMID
32638590
.
S2CID
220413025
.
- ^
Mark Heiden (8 July 2020).
"New biomaterial could shield against harmful radiation"
. Northwestern University
. Retrieved
29 January
2023
.
- ^
Prota, G.; Searle, A. G. (1978).
"Biochemical sites of gene action for melanogenesis in mammals"
.
Annales de Genetique et de Selection Animale
.
10
(1): 1?8.
doi
:
10.1186/1297-9686-10-1-1
.
PMC
2757330
.
PMID
22896083
.
- ^
Solano, F. (2014).
"Melanins: Skin Pigments and Much More?Types, Structural Models, Biological Functions, and Formation Routes"
.
New Journal of Science
.
2014
: 1?28.
doi
:
10.1155/2014/498276
.
- ^
Cichorek, Mirosława; Wachulska, Małgorzata; Stasiewicz, Aneta; Tymi?ska, Agata (20 February 2013).
"Skin melanocytes: biology and development"
.
Advances in Dermatology and Allergology
.
30
(1): 30?41.
doi
:
10.5114/pdia.2013.33376
.
PMC
3834696
.
PMID
24278043
.
- ^
"oculocutaneous albinism"
.
Genetics Home Reference
. Retrieved
25 September
2017
.
- ^
Hamilton AJ, Gomez BL (March 2002).
"Melanins in fungal pathogens"
.
Journal of Medical Microbiology
.
51
(3): 189?91.
doi
:
10.1099/0022-1317-51-3-189
.
PMID
11871612
.
- ^
Cerenius L, Soderhall K (April 2004). "The prophenoloxidase-activating system in invertebrates".
Immunological Reviews
.
198
: 116?26.
doi
:
10.1111/j.0105-2896.2004.00116.x
.
PMID
15199959
.
S2CID
10614298
.
- ^
Castelvecchi, Davide (26 May 2007). "Dark Power: Pigment seems to put radiation to good use".
Science News
.
171
(21): 325.
doi
:
10.1002/scin.2007.5591712106
.
- ^
Dadachova E, Bryan RA, Huang X, et al. (2007).
"Ionizing radiation changes the electronic properties of melanin and enhances the growth of melanized fungi"
.
PLOS ONE
.
2
(5): e457.
Bibcode
:
2007PLoSO...2..457D
.
doi
:
10.1371/journal.pone.0000457
.
PMC
1866175
.
PMID
17520016
.
- ^
Skold HN, Aspengren S, Cheney KL, Wallin M (2016). "Chapter Four - Fish Chromatophores?From Molecular Motors to Animal Behavior".
International Review of Cell and Molecular Biology
.
321
: 171?219.
doi
:
10.1016/bs.ircmb.2015.09.005
.
PMID
26811288
.
- ^
Kottler, Verena A.; Kunstner, Axel; Schartl, Manfred (May 2015).
"Pheomelanin in fish?"
.
Pigment Cell & Melanoma Research
.
28
(3): 355?356.
doi
:
10.1111/pcmr.12359
.
ISSN
1755-1471
.
PMID
25660115
.
S2CID
8877527
.
- ^
Xu, Peng; Zhang, Xiaofeng; Wang, Xumin; Li, Jiongtang; Liu, Guiming; Kuang, Youyi; Xu, Jian; Zheng, Xianhu; Ren, Lufeng; Wang, Guoliang; Zhang, Yan; Huo, Linhe; Zhao, Zixia; Cao, Dingchen; Lu, Cuiyun (November 2014).
"Genome sequence and genetic diversity of the common carp, Cyprinus carpio"
.
Nature Genetics
.
46
(11): 1212?1219.
doi
:
10.1038/ng.3098
.
ISSN
1061-4036
.
PMID
25240282
.
- ^
Mouchet SR, Cortesi F, Bokic B, Lazovic V, Vukusic P, Marshall NJ, Kolaric B (1 November 2023).
"Morphological and Optical Modification of Melanosomes in Fish Integuments upon Oxidation"
.
Optics
.
4
(4): 563?562.
doi
:
10.3390/opt4040041
.
- ^
Gunderson, Alex R.; Frame, Alicia M.; Swaddle, John P.; Forsyth, Mark H. (1 September 2008). "Resistance of melanized feathers to bacterial degradation: is it really so black and white?".
Journal of Avian Biology
.
39
(5): 539?545.
doi
:
10.1111/j.0908-8857.2008.04413.x
.
- ^
Bonser, Richard H. C. (1995).
"Melanin and the Abrasion Resistance of Feathers"
.
Condor
.
97
(2): 590?591.
doi
:
10.2307/1369048
.
JSTOR
1369048
.
- ^
Galvan, Ismael; Solano, Francisco (8 April 2016).
"Bird Integumentary Melanins: Biosynthesis, Forms, Function and Evolution"
.
International Journal of Molecular Sciences
.
17
(4): 520.
doi
:
10.3390/ijms17040520
.
PMC
4848976
.
PMID
27070583
.
- ^
Rodriguez-Martinez, Sol; Galvan, Ismael (2020). "Juvenile pheomelanin-based plumage coloration has evolved more frequently in carnivorous species".
Ibis
.
162
(1): 238?244.
doi
:
10.1111/ibi.12770
.
hdl
:
10261/207451
.
ISSN
1474-919X
.
S2CID
202018215
.
- ^
Jimbow, K; Quevedo WC, Jr; Fitzpatrick, TB; Szabo, G (July 1976).
"Some aspects of melanin biology: 1950?1975"
.
The Journal of Investigative Dermatology
.
67
(1): 72?89.
doi
:
10.1111/1523-1747.ep12512500
.
PMID
819593
.
- ^
Meneely, Philip (2014).
Genetic Analysis: Genes, Genomes, and Networks in Eukaryotes
. Oxford University Press.
ISBN
9780199681266
.
- ^
Griffiths, Anthony JF; Miller, Jeffrey H.; Suzuki, David T.; Lewontin, Richard C.; Gelbart, William M. (2000).
"Gene interaction in coat color of mammals"
.
NCBI
.
[
permanent dead link
]
- ^
Millar, S. E.; Miller, M. W.; Stevens, M. E.; Barsh, G. S. (October 1995). "Expression and transgenic studies of the mouse agouti gene provide insight into the mechanisms by which mammalian coat color patterns are generated".
Development
.
121
(10): 3223?3232.
doi
:
10.1242/dev.121.10.3223
.
PMID
7588057
.
- ^
Neville, A. C. (2012).
Biology of the Arthropod Cuticle
. Springer Science & Business Media.
ISBN
9783642809101
.
- ^
Mouchet, Sebastien R; Deparis, Olivier (2021),
Natural Photonics and Bioinspiration
(1st ed.), Artech House,
ISBN
978-163-081-797-8
- ^
Hsiung, B.-K.; Blackledge, T. A.; Shawkey, M. D. (2015).
"Spiders do have melanin after all"
.
Journal of Experimental Biology
.
218
(22): 3632?3635.
doi
:
10.1242/jeb.128801
.
PMID
26449977
.
- ^
Hegna, Robert H.; Nokelainen, Ossi; Hegna, Jonathan R.; Mappes, Johanna (2013).
"To quiver or to shiver: increased melanization benefits thermoregulation, but reduces warning signal efficacy in the wood tiger moth"
.
Proc. R. Soc. B
.
280
(1755): 20122812.
doi
:
10.1098/rspb.2012.2812
.
PMC
3574392
.
PMID
23363631
.
- ^
a
b
Mosse, Irma B.; Dubovic, Boris V.; Plotnikova, Svetlana I.; Kostrova, Ludmila N.; Molophei, Vadim; Subbot, Svetlana T.; Maksimenya, Inna P. (20?25 May 2001). Obelic, B.; Ranogajev-Komor, M.; Miljanic, S.; Krajcar Bronic, I. (eds.).
Melanin is Effective Radioprotector against Chronic Irradiation and Low Radiation Doses
. IRPA Regional Congress on Radiation Protection in Central Europe: Radiation Protection and Health.
INIS
.
Dubrovnik
(Croatia):
Croatian Radiation Protection Association
. p. 35 (of 268).
- ^
a
b
Gessler, N. N.; Egorova, A. S.; Belozerskaya, T. A. (2014).
"Melanin pigments of fungi under extreme environmental conditions (Review)"
.
Applied Biochemistry and Microbiology
.
50
(2).
Pleiades Publishing
: 105?113.
doi
:
10.1134/s0003683814020094
.
ISSN
0003-6838
.
PMID
25272728
.
S2CID
8570835
.
- ^
a
b
Nenoi, M; Wang, B; Vares, G (12 June 2014).
"In vivo radioadaptive response"
.
Toxicology
.
34
(3).
Sage
: 272?283.
doi
:
10.1177/0960327114537537
.
ISSN
0960-3271
.
PMC
4442823
.
PMID
24925363
.
- ^
a
b
Liu, Heng; Yang, Youyuan; Liu, Yu; Pan, Jingjing; Wang, Junqing; Man, Fengyuan; Zhang, Weiguo; Liu, Gang (7 February 2020).
"Melanin-Like Nanomaterials for Advanced Biomedical Applications: A Versatile Platform with Extraordinary Promise"
.
Advanced Science
.
7
(7).
Wiley-VCH
: 1903129.
doi
:
10.1002/advs.201903129
.
ISSN
2198-3844
.
PMC
7141020
.
PMID
32274309
.
- ^
a
b
c
d
e
Mosse, Irma B. (2012). "Genetic effects of ionizing radiation ? some questions with no answers".
Journal of Environmental Radioactivity
.
112
.
Elsevier
: 70?75.
Bibcode
:
2012JEnvR.112...70M
.
doi
:
10.1016/j.jenvrad.2012.05.009
.
ISSN
0265-931X
.
PMID
22683898
.
- ^
a
b
c
d
e
f
g
h
i
Mosse, Irma; Kilchevsky, Alexander; Nikolova, Nevena; Zhelev, Nikolai (14 December 2016).
"Some problems and errors in cytogenetic biodosimetry"
.
Biotechnology & Biotechnological Equipment
.
31
(3).
Taylor & Francis
: 460?468.
doi
:
10.1080/13102818.2016.1259018
.
ISSN
1310-2818
.
S2CID
59398089
.
- ^
a
b
c
Mosse, Irma (18 January 2022). "Radiobiology in my life ? Irma Mosse".
International Journal of Radiation Biology
.
98
(3: Women in Radiobiology).
Taylor & Francis
: 474?478.
doi
:
10.1080/09553002.2022.2026517
.
ISSN
0955-3002
.
PMID
34994663
.
S2CID
245823003
.
- ^
a
b
Dadachova, Ekaterina; Casadevall, Arturo (2011). Horikoshi, K?ki (ed.).
Extremophiles handbook
. Tokyo New York City:
Springer
. pp. xxix+1247.
ISBN
978-4-431-53898-1
.
OCLC
700199222
.
ISBN
978-4-431-53897-4
.
OCLC
711778164
.
- ^
Yao, Zeng-Yu; Qi, Jian-Hua (22 April 2016).
"Comparison of Antioxidant Activities of Melanin Fractions from Chestnut Shell"
.
Molecules
.
21
(4): 487.
doi
:
10.3390/molecules21040487
.
PMC
6273334
.
PMID
27110763
.
- ^
Kim, Y.-J.; Uyama, H. (15 May 2005).
"Tyrosinase inhibitors from natural and synthetic sources: structure, inhibition mechanism and perspective for the future"
.
Cellular and Molecular Life Sciences
.
62
(15): 1707?1723.
doi
:
10.1007/s00018-005-5054-y
.
PMC
11139184
.
PMID
15968468
.
S2CID
8280251
.
- ^
a
b
Solano, F. (2014).
"Melanins: Skin Pigments and Much More?Types, Structural Models, Biological Functions, and Formation Routes"
.
New Journal of Science
.
2014
(498276): 1?28.
doi
:
10.1155/2014/498276
.
- ^
Mason, H. S. (1948).
"The chemistry of melanin. Mechanism of the oxidation of dihydroxyphenylalanine by tyrosinase"
.
Journal of Biological Chemistry
.
172
(1): 83?99.
doi
:
10.1016/S0021-9258(18)35614-X
.
PMID
18920770
.
- ^
Zaidi, Kamal Uddin; Ali, Ayesha S.; Ali, Sharique A.; Naaz, Ishrat (2014).
"Microbial Tyrosinases: Promising Enzymes for Pharmaceutical, Food Bioprocessing, and Environmental Industry"
.
Biochemistry Research International
.
2014
: 1?16 (see Fig. 3).
doi
:
10.1155/2014/854687
.
PMC
4033337
.
PMID
24895537
.
- ^
"Melanin"
.
pubchem.ncbi.nlm.nih.gov
. Retrieved
25 September
2017
.
- ^
"Oculocutaneous Albinism"
. Archived from
the original
on 23 December 2008.
- ^
a
b
Peracha, Mohammed O.; Cosgrove, Frances M.; Garcia-Valenzuela, Enrique; Eliott, Dean (5 November 2015). Roy, Sr, Hampton; Talavera, Francisco; Rowsey, J. James (eds.).
"Ocular Manifestations of Albinism: Background, Pathophysiology, Epidemiology"
.
Medscape
. Additional contributions from Kilbourn Gordon, III.
WebMD
.
Archived
from the original on 28 March 2017
. Retrieved
8 September
2022
– via
eMedicine
.
- ^
"Causes of Variability"
. Archived from
the original
on 23 September 2006
. Retrieved
18 September
2006
.
- ^
EntrezGene
300700
- ^
EntrezGene
606933
- ^
Cable J, Huszar D, Jaenisch R, Steel KP (February 1994). "Effects of mutations at the W locus (c-kit) on inner ear pigmentation and function in the mouse".
Pigment Cell Research
.
7
(1): 17?32.
doi
:
10.1111/j.1600-0749.1994.tb00015.x
.
PMID
7521050
.
- ^
"Lewy Body Disease"
. Archived from
the original
on 21 July 2009.
- ^
Meyskens FL, Farmer P, Fruehauf JP (June 2001).
"Redox regulation in human melanocytes and melanoma"
(PDF)
.
Pigment Cell Research
.
14
(3): 148?54.
doi
:
10.1034/j.1600-0749.2001.140303.x
.
PMID
11434561
.
- ^
Meier-Davis SR, Dines K, Arjmand FM, et al. (December 2012). "Comparison of oral and transdermal administration of rasagiline mesylate on human melanoma tumor growth in vivo".
Cutaneous and Ocular Toxicology
.
31
(4): 312?7.
doi
:
10.3109/15569527.2012.676119
.
PMID
22515841
.
S2CID
30344869
.
- ^
King G, Yerger VB, Whembolua GL, Bendel RB, Kittles R, Moolchan ET (June 2009). "Link between facultative melanin and tobacco use among African Americans".
Pharmacology Biochemistry and Behavior
.
92
(4): 589?96.
doi
:
10.1016/j.pbb.2009.02.011
.
PMID
19268687
.
S2CID
3070838
.
- ^
"Human Skin Color Variation"
.
The Smithsonian Institution's Human Origins Program
. 20 June 2012
. Retrieved
24 August
2019
.
- ^
Berth-Jones, J. (2010), "Constitutive pigmentation, human pigmentation and the response to sun exposure", in Tony Burns; Stephen Breathnach; Neil Cox; Christopher Griffiths (eds.),
Rook's Textbook of Dermatology
, vol. 3 (8th ed.), Wiley-Blackwell, p. 58.9,
ISBN
978-1-4051-6169-5
- ^
Wade, Nicholas (19 August 2003).
"Why Humans and Their Fur Parted Ways"
.
The New York Times
.
ISSN
0362-4331
. Retrieved
24 August
2019
.
- ^
Tishkoff SA, Reed FA, Friedlaender FR, et al. (May 2009).
"The genetic structure and history of Africans and African Americans"
.
Science
.
324
(5930): 1035?44.
Bibcode
:
2009Sci...324.1035T
.
doi
:
10.1126/science.1172257
.
PMC
2947357
.
PMID
19407144
.
- ^
"A Single Migration From Africa Populated the World, Studies Find"
.
The New York Times
. 22 September 2016.
- ^
Harding RM, Healy E, Ray AJ, et al. (April 2000).
"Evidence for variable selective pressures at MC1R"
.
American Journal of Human Genetics
.
66
(4): 1351?61.
doi
:
10.1086/302863
.
PMC
1288200
.
PMID
10733465
.
- ^
Lamason RL, Mohideen MA, Mest JR, et al. (December 2005). "SLC24A5, a putative cation exchanger, affects pigmentation in zebrafish and humans".
Science
.
310
(5755): 1782?6.
Bibcode
:
2005Sci...310.1782L
.
doi
:
10.1126/science.1116238
.
PMID
16357253
.
S2CID
2245002
.
- ^
Jablonski, Nina G.; Chaplin, George (11 May 2010).
"Human skin pigmentation as an adaptation to UV radiation"
.
Proceedings of the National Academy of Sciences
.
107
(Supplement 2): 8962?8968.
Bibcode
:
2010PNAS..107.8962J
.
doi
:
10.1073/pnas.0914628107
.
PMC
3024016
.
PMID
20445093
.
- ^
Liu Y, Hong L, Kempf VR, Wakamatsu K, Ito S, Simon JD (June 2004). "Ion-exchange and adsorption of Fe(III) by Sepia melanin".
Pigment Cell Research
.
17
(3): 262?9.
doi
:
10.1111/j.1600-0749.2004.00140.x
.
PMID
15140071
.
- ^
Donatien PD, Orlow SJ (August 1995). "Interaction of melanosomal proteins with melanin".
European Journal of Biochemistry
.
232
(1): 159?64.
doi
:
10.1111/j.1432-1033.1995.tb20794.x
.
PMID
7556145
.
- ^
Sarangarajan R, Apte SP (2005). "Melanin aggregation and polymerization: possible implications in age-related macular degeneration".
Ophthalmic Research
.
37
(3): 136?41.
doi
:
10.1159/000085533
.
PMID
15867475
.
S2CID
27499198
.
- ^
Meyskens FL, Farmer PJ, Anton-Culver H (April 2004).
"Etiologic pathogenesis of melanoma: a unifying hypothesis for the missing attributable risk"
(PDF)
.
Clinical Cancer Research
.
10
(8): 2581?3.
doi
:
10.1158/1078-0432.ccr-03-0638
.
PMID
15102657
.
S2CID
26079375
.
- ^
Sarangarajan R, Apte SP (2005). "Melanization and phagocytosis: implications for age related macular degeneration".
Molecular Vision
.
11
: 482?90.
PMID
16030499
.
- ^
a
b
Sarna, Michal; Krzykawska-Serda, Martyna; Jakubowska, Monika; Zadlo, Andrzej; Urbanska, Krystyna (26 June 2019).
"Melanin presence inhibits melanoma cell spread in mice in a unique mechanical fashion"
.
Scientific Reports
.
9
(1): 9280.
Bibcode
:
2019NatSR...9.9280S
.
doi
:
10.1038/s41598-019-45643-9
.
ISSN
2045-2322
.
PMC
6594928
.
PMID
31243305
.
External links
[
edit
]
Look up
melanin
in Wiktionary, the free dictionary.
Look up
leuco form
in Wiktionary, the free dictionary.