Chemical element, symbol Fm and atomic number 100
Fermium
is a
synthetic chemical element
; it has
symbol
Fm
and
atomic number
100. It is an
actinide
and the heaviest element that can be formed by
neutron
bombardment of lighter elements, and hence the last element that can be prepared in macroscopic quantities, although pure fermium metal has not yet been prepared.
[4]
A total of 20 isotopes are known, with
257
Fm being the longest-lived with a half-life of 100.5 days.
It was discovered in the debris of the
first
hydrogen bomb
explosion in 1952, and named after
Enrico Fermi
, one of the pioneers of
nuclear physics
. Its chemistry is typical for the late actinides, with a preponderance of the +3
oxidation state
but also an accessible +2 oxidation state. Owing to the small amounts of produced fermium and all of its isotopes having relatively short half-lives, there are currently no uses for it outside basic scientific research.
Discovery
[
edit
]
 Fermium was first observed in the fallout from the
Ivy Mike
nuclear test.
Fermium was first observed in the fallout from the
Ivy Mike
nuclear test.
 The element was named after
Enrico Fermi
.
The element was named after
Enrico Fermi
.
 The element was discovered by a team headed by
Albert Ghiorso
.
The element was discovered by a team headed by
Albert Ghiorso
.
Fermium was first discovered in the fallout from the '
Ivy Mike
' nuclear test (1 November 1952), the first successful test of a hydrogen bomb.
[5]
[6]
[7]
Initial examination of the debris from the explosion had shown the production of a new isotope of
plutonium
,
244
94
Pu
: this could only have formed by the absorption of six
neutrons
by a
uranium-238
nucleus followed by two
β
?
decays
. At the time, the absorption of neutrons by a heavy nucleus was thought to be a rare process, but the identification of
244
94
Pu
raised the possibility that still more neutrons could have been absorbed by the uranium nuclei, leading to new elements.
[7]
Element 99 (
einsteinium
) was quickly discovered on filter papers which had been flown through the cloud from the explosion (the same sampling technique that had been used to discover
244
94
Pu
).
[7]
It was then identified in December 1952 by
Albert Ghiorso
and co-workers at the
University of California at Berkeley
.
[5]
[6]
[7]
They discovered the isotope
253
Es (
half-life
20.5 days) that was made by the
capture
of 15
neutrons
by
uranium-238
nuclei ? which then underwent seven successive
beta decays
:
![{\displaystyle {\ce {^{238}_{92}U ->[+ 15 {\ce {n}}][7 \beta^-] ^{253}_{99}Es}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/38f0bd3ea3e7092c2e8d58a9cb7b20044a2680e3) | | (
1
)
|
Some
238
U atoms, however, could capture another amount of neutrons (most likely, 16 or 17).
The discovery of fermium (
Z
= 100) required more material, as the yield was expected to be at least an order of magnitude lower than that of element 99, and so contaminated coral from the
Enewetak atoll
(where the test had taken place) was shipped to the
University of California Radiation Laboratory
in
Berkeley, California
, for processing and analysis. About two months after the test, a new component was isolated emitting high-energy
α-particles
(7.1 MeV) with a
half-life
of about a day. With such a short half-life, it could only arise from the β
?
decay of an isotope of einsteinium, and so had to be an isotope of the new element 100: it was quickly identified as
255
Fm (
t
= 20.07(7) hours
).
[7]
The discovery of the new elements, and the new data on neutron capture, was initially kept secret on the orders of the U.S. military until 1955 due to
Cold War
tensions.
[7]
[8]
[9]
Nevertheless, the Berkeley team was able to prepare elements 99 and 100 by civilian means, through the neutron bombardment of
plutonium-239
, and published this work in 1954 with the disclaimer that it was not the first studies that had been carried out on the elements.
[10]
[11]
The "Ivy Mike" studies were declassified and published in 1955.
[8]
The Berkeley team had been worried that another group might discover lighter isotopes of element 100 through ion-bombardment techniques before they could publish their classified research,
[7]
and this proved to be the case. A group at the Nobel Institute for Physics in Stockholm independently discovered the element, producing an
isotope
later confirmed to be
250
Fm (
t
1/2
= 30 minutes) by bombarding a
238
92
U
target with
oxygen-16
ions, and published their work in May 1954.
[12]
Nevertheless, the priority of the Berkeley team was generally recognized, and with it the prerogative to name the new element in honour of
Enrico Fermi
, the developer of the first artificial self-sustained nuclear reactor. Fermi was still alive when the name was proposed, but had died by the time it became official.
[13]
Isotopes
[
edit
]
 Decay pathway of fermium-257
Decay pathway of fermium-257
There are 20 isotopes of fermium listed in N
UBASE
2016,
[14]
with atomic weights of 241 to 260,
[b]
of which
257
Fm is the longest-lived with a
half-life
of 100.5 days.
253
Fm has a half-life of 3 days, while
251
Fm of 5.3 h,
252
Fm of 25.4 h,
254
Fm of 3.2 h,
255
>Fm of 20.1 h, and
256
Fm of 2.6 hours. All the remaining ones have half-lives ranging from 30 minutes to less than a millisecond.
[15]
The neutron capture product of fermium-257,
258
Fm, undergoes
spontaneous fission
with a half-life of just 370(14) microseconds;
259
Fm and
260
Fm also undergo spontaneous fission (
t
1/2
= 1.5(3) s and 4 ms respectively).
[15]
This means that neutron capture cannot be used to create
nuclides
with a
mass number
greater than 257, unless carried out in a nuclear explosion. As
257
Fm
alpha decays
to
253
Cf, and no known fermium isotopes undergo
beta minus decay
to the next element,
mendelevium
, fermium is also the last element that can be synthesized by neutron-capture.
[4]
[16]
[17]
Because of this impediment in forming heavier isotopes, these short-lived isotopes
258?260
Fm constitute the "fermium gap."
[18]
Production
[
edit
]
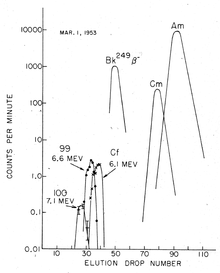 Elution
: chromatographic separation of Fm(100), Es(99), Cf, Bk, Cm and Am
Elution
: chromatographic separation of Fm(100), Es(99), Cf, Bk, Cm and Am
Fermium is produced by the bombardment of lighter
actinides
with
neutrons
in a nuclear reactor. Fermium-257 is the heaviest isotope that is obtained via neutron capture, and can only be produced in picogram quantities.
[c]
[19]
The major source is the 85 MW
High Flux Isotope Reactor
(HFIR) at the
Oak Ridge National Laboratory
in
Tennessee
, USA, which is dedicated to the production of transcurium (
Z
> 96) elements.
[20]
Lower mass fermium isotopes are available in greater quantities, though these isotopes (
254
Fm and
255
Fm) are comparatively short-lived. In a "typical processing campaign" at Oak Ridge, tens of grams of
curium
are irradiated to produce decigram quantities of
californium
, milligram quantities of
berkelium
and
einsteinium
, and picogram quantities of fermium.
[21]
However, nanogram
[22]
quantities of fermium can be prepared for specific experiments. The quantities of fermium produced in 20?200 kiloton thermonuclear explosions is believed to be of the order of milligrams, although it is mixed in with a huge quantity of debris; 4.0 picograms of
257
Fm was recovered from 10 kilograms of debris from the "
Hutch
" test (16 July 1969).
[23]
The Hutch experiment produced an estimated total of 250 micrograms of
257
Fm.
After production, the fermium must be separated from other actinides and from
lanthanide
fission products. This is usually achieved by
ion-exchange chromatography
, with the standard process using a cation exchanger such as Dowex 50 or T
EVA
eluted with a solution of ammonium α-hydroxyisobutyrate.
[4]
[24]
Smaller cations form more stable complexes with the α-hydroxyisobutyrate anion, and so are preferentially eluted from the column.
[4]
A rapid
fractional crystallization
method has also been described.
[4]
[25]
Although the most stable isotope of fermium is
257
Fm, with a
half-life
of 100.5 days, most studies are conducted on
255
Fm (
t
1/2
= 20.07(7) hours), since this isotope can be easily isolated as required as the decay product of
255
Es (
t
1/2
= 39.8(12) days).
[4]
Synthesis in nuclear explosions
[
edit
]
The analysis of the debris at the 10-
megaton
Ivy Mike
nuclear test was a part of long-term project, one of the goals of which was studying the efficiency of production of transuranium elements in high-power nuclear explosions. The motivation for these experiments was as follows: synthesis of such elements from uranium requires multiple neutron capture. The probability of such events increases with the neutron flux, and nuclear explosions are the most powerful neutron sources, providing densities on the order 10
23
neutrons/cm
2
within a microsecond, i.e. about 10
29
neutrons/(cm
2
·s). For comparison, the flux of the HFIR reactor is 5
×
10
15
neutrons/(cm
2
·s). A dedicated laboratory was set up right at
Enewetak Atoll
for preliminary analysis of debris, as some isotopes could have decayed by the time the debris samples reached the U.S. The laboratory was receiving samples for analysis, as soon as possible, from airplanes equipped with paper filters which flew over the atoll after the tests. Whereas it was hoped to discover new chemical elements heavier than fermium, those were not found after a series of megaton explosions conducted between 1954 and 1956 at the atoll.
[26]
 Estimated yield of transuranium elements in the U.S. nuclear tests Hutch and Cyclamen.
[27]
Estimated yield of transuranium elements in the U.S. nuclear tests Hutch and Cyclamen.
[27]
The atmospheric results were supplemented by the underground test data accumulated in the 1960s at the
Nevada Test Site
, as it was hoped that powerful explosions conducted in confined space might result in improved yields and heavier isotopes. Apart from traditional uranium charges, combinations of uranium with americium and thorium have been tried, as well as a mixed plutonium-neptunium charge. They were less successful in terms of yield, which was attributed to stronger losses of heavy isotopes due to enhanced fission rates in heavy-element charges. Isolation of the products was found to be rather problematic, as the explosions were spreading debris through melting and vaporizing rocks under the great depth of 300?600 meters, and drilling to such depth in order to extract the products was both slow and inefficient in terms of collected volumes.
[26]
[27]
Among the nine underground tests, which were carried between 1962 and 1969 and codenamed Anacostia (5.2
kilotons
, 1962), Kennebec (<5 kilotons, 1963), Par (38 kilotons, 1964), Barbel (<20 kilotons, 1964), Tweed (<20 kilotons, 1965), Cyclamen (13 kilotons, 1966), Kankakee (20-200 kilotons, 1966), Vulcan (25 kilotons, 1966) and Hutch (20-200 kilotons, 1969),
[28]
the last one was most powerful and had the highest yield of transuranium elements. In the dependence on the atomic mass number, the yield showed a saw-tooth behavior with the lower values for odd isotopes, due to their higher fission rates.
[27]
The major practical problem of the entire proposal, however, was collecting the radioactive debris dispersed by the powerful blast. Aircraft filters adsorbed only about 4
×
10
?14
of the total amount and collection of tons of corals at Enewetak Atoll increased this fraction by only two orders of magnitude. Extraction of about 500 kilograms of underground rocks 60 days after the Hutch explosion recovered only about 10
?7
of the total charge. The amount of transuranium elements in this 500-kg batch was only 30 times higher than in a 0.4 kg rock picked up 7 days after the test. This observation demonstrated the highly nonlinear dependence of the transuranium elements yield on the amount of retrieved radioactive rock.
[29]
In order to accelerate sample collection after the explosion, shafts were drilled at the site not after but before the test, so that the explosion would expel radioactive material from the epicenter, through the shafts, to collecting volumes near the surface. This method was tried in the Anacostia and Kennebec tests and instantly provided hundreds of kilograms of material, but with actinide concentrations 3 times lower than in samples obtained after drilling; whereas such a method could have been efficient in scientific studies of short-lived isotopes, it could not improve the overall collection efficiency of the produced actinides.
[30]
Though no new elements (apart from einsteinium and fermium) could be detected in the nuclear test debris, and the total yields of transuranium elements were disappointingly low, these tests did provide significantly higher amounts of rare heavy isotopes than previously available in laboratories. For example, 6
×
10
9
atoms of
257
Fm could be recovered after the Hutch detonation. They were then used in the studies of thermal-neutron induced fission of
257
Fm and in discovery of a new fermium isotope
258
Fm. Also, the rare isotope
250
Cm was synthesized in large quantities, which is very difficult to produce in nuclear reactors from its progenitor
249
Cm; the half-life of
249
Cm (64 minutes) is much too short for months-long reactor irradiations, but is very "long" on the explosion timescale.
[31]
Natural occurrence
[
edit
]
Because of the short half-life of all known isotopes of fermium, any
primordial
fermium, that is fermium present on Earth during its formation, has decayed by now. Synthesis of fermium from naturally occurring uranium and thorium in the Earth's crust requires multiple neutron captures, which is extremely unlikely. Therefore, most fermium is produced on Earth in laboratories, high-power nuclear reactors, or in
nuclear tests
, and is present for only a few months afterward. The
transuranic elements
americium
to fermium did occur naturally in the
natural nuclear fission reactor
at
Oklo
, but no longer do so.
[32]
Chemistry
[
edit
]
 A fermium-
ytterbium
alloy used for measuring the
enthalpy of vaporization
of fermium metal
A fermium-
ytterbium
alloy used for measuring the
enthalpy of vaporization
of fermium metal
The chemistry of fermium has only been studied in solution using tracer techniques, and no solid compounds have been prepared. Under normal conditions, fermium exists in solution as the Fm
3+
ion, which has a
hydration number
of 16.9 and an
acid dissociation constant
of 1.6
×
10
?4
(p
K
a
= 3.8).
[33]
[34]
Fm
3+
forms complexes with a wide variety of organic ligands with
hard
donor atoms such as oxygen, and these complexes are usually more stable than those of the preceding actinides.
[4]
It also forms anionic complexes with ligands such as
chloride
or
nitrate
and, again, these complexes appear to be more stable than those formed by
einsteinium
or
californium
.
[35]
It is believed that the bonding in the complexes of the later actinides is mostly
ionic
in character: the Fm
3+
ion is expected to be smaller than the preceding An
3+
ions because of the higher
effective nuclear charge
of fermium, and hence fermium would be expected to form shorter and stronger metal?ligand bonds.
[4]
Fermium(III) can be fairly easily reduced to fermium(II),
[36]
for example with
samarium(II) chloride
, with which fermium(II) coprecipitates.
[37]
[38]
In the precipitate, the compound fermium(II) chloride (FmCl
2
) was produced, though it was not purified or studied in isolation.
[39]
The
electrode potential
has been estimated to be similar to that of the
ytterbium
(III)/(II) couple, or about ?1.15 V with respect to the
standard hydrogen electrode
,
[40]
a value which agrees with theoretical calculations.
[41]
The Fm
2+
/Fm
0
couple has an electrode potential of ?2.37(10) V based on
polarographic
measurements.
[42]
Toxicity
[
edit
]
Though few people come in contact with fermium, the
International Commission on Radiological Protection
has set annual exposure limits for the two most stable isotopes. For fermium-253, the ingestion limit was set at 10
7
becquerels
(1 Bq equals one decay per second), and the inhalation limit at 10
5
Bq; for fermium-257, at 10
5
Bq and 4,000 Bq respectively.
[43]
Notes and references
[
edit
]
Notes
[
edit
]
- ^
The density is calculated from the predicted metallic radius (Silva 2006, p. 1628) and the predicted close-packed crystal structure (Fournier 1976).
- ^
The discovery of
260
Fm is considered "unproven" in N
UBASE
2003.
[15]
- ^
All isotopes of elements Z > 100 can only be produced by accelerator-based nuclear reactions with charged particles and can be obtained only in tracer quantities (e.g., 1 million atoms for Md (
Z
= 101) per hour of irradiation (see Silva 2006).
References
[
edit
]
- ^
a
b
Fournier, Jean-Marc (1976). "Bonding and the electronic structure of the actinide metals".
Journal of Physics and Chemistry of Solids
.
37
(2): 235?244.
Bibcode
:
1976JPCS...37..235F
.
doi
:
10.1016/0022-3697(76)90167-0
.
- ^
Sato, Tetsuya K.; Asai, Masato; Borschevsky, Anastasia; Beerwerth, Randolf; Kaneya, Yusuke; Makii, Hiroyuki; Mitsukai, Akina; Nagame, Yuichiro; Osa, Akihiko; Toyoshima, Atsushi; Tsukada, Kazuki; Sakama, Minoru; Takeda, Shinsaku; Ooe, Kazuhiro; Sato, Daisuke; Shigekawa, Yudai; Ichikawa, Shin-ichi; Dullmann, Christoph E.; Grund, Jessica; Renisch, Dennis; Kratz, Jens V.; Schadel, Matthias; Eliav, Ephraim; Kaldor, Uzi; Fritzsche, Stephan; Stora, Thierry (25 October 2018). "First Ionization Potentials of Fm, Md, No, and Lr: Verification of Filling-Up of 5f Electrons and Confirmation of the Actinide Series".
Journal of the American Chemical Society
.
140
(44): 14609?14613.
doi
:
10.1021/jacs.8b09068
.
- ^
Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021).
"The NUBASE2020 evaluation of nuclear properties"
(PDF)
.
Chinese Physics C
.
45
(3): 030001.
doi
:
10.1088/1674-1137/abddae
.
- ^
a
b
c
d
e
f
g
h
Silva, Robert J. (2006).
"Fermium, Mendelevium, Nobelium, and Lawrencium"
(PDF)
. In Morss, Lester R.; Edelstein, Norman M.; Fuger, Jean (eds.).
The Chemistry of the Actinide and Transactinide Elements
. Vol. 3 (3rd ed.). Dordrecht: Springer. pp. 1621?1651.
doi
:
10.1007/1-4020-3598-5_13
.
ISBN
978-1-4020-3555-5
. Archived from
the original
(PDF)
on 17 July 2010.
- ^
a
b
"Einsteinium"
. Archived from
the original
on 26 October 2007
. Retrieved
7 December
2007
.
- ^
a
b
Fermium ? National Research Council Canada
Archived
2010-12-25 at the
Wayback Machine
. Retrieved 2 December 2007
- ^
a
b
c
d
e
f
g
Ghiorso, Albert
(2003).
"Einsteinium and Fermium"
.
Chemical and Engineering News
.
81
(36): 174?175.
doi
:
10.1021/cen-v081n036.p174
.
- ^
a
b
Ghiorso, A.; Thompson, S.; Higgins, G.; Seaborg, Glenn T.; Studier, M.; Fields, P.; Fried, S.; Diamond, H.; et al. (1955).
"New Elements Einsteinium and Fermium, Atomic Numbers 99 and 100"
(PDF)
.
Phys. Rev
.
99
(3): 1048?1049.
Bibcode
:
1955PhRv...99.1048G
.
doi
:
10.1103/PhysRev.99.1048
.
- ^
Fields, P. R.; Studier, M. H.; Diamond, H.; Mech, J. F.; Inghram, M. G. Pyle, G. L.; Stevens, C. M.; Fried, S.; Manning, W. M. (Argonne National Laboratory, Lemont, Illinois); Ghiorso, A.; Thompson, S. G.; Higgins, G. H.; Seaborg, G. T. (University of California, Berkeley, California): "Transplutonium Elements in Thermonuclear Test Debris", in:
Fields, P.; Studier, M.; Diamond, H.; Mech, J.; Inghram, M.; Pyle, G.; Stevens, C.; Fried, S.; Manning, W.; Ghiorso, A.; Thompson, S.; Higgins, G.; Seaborg, G. (1956). "Transplutonium Elements in Thermonuclear Test Debris".
Physical Review
.
102
(1): 180.
Bibcode
:
1956PhRv..102..180F
.
doi
:
10.1103/PhysRev.102.180
.
- ^
Thompson, S. G.;
Ghiorso, A.
; Harvey, B. G.; Choppin, G. R. (1954).
"Transcurium Isotopes Produced in the Neutron Irradiation of Plutonium"
(PDF)
.
Physical Review
.
93
(4): 908.
Bibcode
:
1954PhRv...93..908T
.
doi
:
10.1103/PhysRev.93.908
.
- ^
Choppin, G. R.; Thompson, S. G.;
Ghiorso, A.
; Harvey, B. G. (1954).
"Nuclear Properties of Some Isotopes of Californium, Elements 99 and 100"
.
Physical Review
.
94
(4): 1080?1081.
Bibcode
:
1954PhRv...94.1080C
.
doi
:
10.1103/PhysRev.94.1080
.
- ^
Atterling, Hugo; Forsling, Wilhelm; Holm, Lennart W.; Melander, Lars; Astrom, Bjorn (1954). "Element 100 Produced by Means of Cyclotron-Accelerated Oxygen Ions".
Physical Review
.
95
(2): 585?586.
Bibcode
:
1954PhRv...95..585A
.
doi
:
10.1103/PhysRev.95.585.2
.
- ^
Hoffman, D. C.
;
Ghiorso, A.
; Seaborg, G. T. (2000).
The Transuranium People: The Inside Story
.
World Scientific
. pp. 187?189.
ISBN
978-1-78-326244-1
.
- ^
Audi, G.; Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S. (2017).
"The NUBASE2016 evaluation of nuclear properties"
(PDF)
.
Chinese Physics C
.
41
(3): 030001.
Bibcode
:
2017ChPhC..41c0001A
.
doi
:
10.1088/1674-1137/41/3/030001
.
- ^
a
b
c
Audi, Georges; Bersillon, Olivier; Blachot, Jean;
Wapstra, Aaldert Hendrik
(2003),
"The N
UBASE
evaluation of nuclear and decay properties"
,
Nuclear Physics A
,
729
: 3?128,
Bibcode
:
2003NuPhA.729....3A
,
doi
:
10.1016/j.nuclphysa.2003.11.001
- ^
Greenwood, Norman N.
; Earnshaw, Alan (1984).
Chemistry of the Elements
. Oxford:
Pergamon Press
. p. 1262.
ISBN
978-0-08-022057-4
.
- ^
Sonzogni, Alejandro.
"Interactive Chart of Nuclides"
. National Nuclear Data Center: Brookhaven National Laboratory. Archived from
the original
on 21 June 2018
. Retrieved
6 June
2008
.
- ^
Zagrebaev, Valeriy; Karpov, Alexander; Greiner, Walter (2013).
"Future of superheavy element research: Which nuclei could be synthesized within the next few years?"
(PDF)
.
Journal of Physics
.
420
(12001): 11.
arXiv
:
1207.5700
.
Bibcode
:
2013JPhCS.420a2001Z
.
doi
:
10.1088/1742-6596/420/1/012001
.
S2CID
55434734
.
- ^
Luig, Heribert; Keller, Cornelius; Wolf, Walter; Shani, Jashovam; Miska, Horst; Zyball, Alfred; Gerve, Andreas; Balaban, Alexandru T.; Kellerer, Albrecht M.; Griebel, Jurgen (2000). "Radionuclides".
Ullmann's Encyclopedia of Industrial Chemistry
.
doi
:
10.1002/14356007.a22_499
.
ISBN
978-3527306732
.
- ^
"High Flux Isotope Reactor"
. Oak Ridge National Laboratory
. Retrieved
23 September
2010
.
- ^
Porter, C. E.; Riley, F. D. Jr.; Vandergrift, R. D.; Felker, L. K. (1997).
"Fermium Purification Using Teva Resin Extraction Chromatography"
.
Sep. Sci. Technol
.
32
(1?4): 83?92.
doi
:
10.1080/01496399708003188
.
- ^
Sewtz, M.; Backe, H.; Dretzke, A.; Kube, G.; Lauth, W.; Schwamb, P.; Eberhardt, K.; Gruning, C.; Thorle, P.; Trautmann, N.; Kunz, P.; Lassen, J.; Passler, G.; Dong, C.; Fritzsche, S.; Haire, R. (2003). "First Observation of Atomic Levels for the Element Fermium (
Z
=100)".
Phys. Rev. Lett
.
90
(16): 163002.
Bibcode
:
2003PhRvL..90p3002S
.
doi
:
10.1103/PhysRevLett.90.163002
.
PMID
12731975
.
S2CID
16234935
.
- ^
Hoff, R. W.; Hulet, E. K. (1970). "Engineering with Nuclear Explosives".
2
: 1283?1294.
- ^
Choppin, G. R.; Harvey, B. G.; Thompson, S. G. (1956).
"A new eluant for the separation of the actinide elements"
(PDF)
.
J. Inorg. Nucl. Chem
.
2
(1): 66?68.
doi
:
10.1016/0022-1902(56)80105-X
.
- ^
Mikheev, N. B.; Kamenskaya, A. N.; Konovalova, N. A.; Rumer, I. A.; Kulyukhin, S. A. (1983). "High-speed method for the separation of fermium from actinides and lanthanides".
Radiokhimiya
.
25
(2): 158?161.
- ^
a
b
Seaborg, p. 39
- ^
a
b
c
Seaborg, p. 40
- ^
United States Nuclear Tests July 1945 through September 1992
Archived
June 15, 2010, at the
Wayback Machine
, DOE/NV--209-REV 15, December 2000
- ^
Seaborg, p. 43
- ^
Seaborg, p. 44
- ^
Seaborg, p. 47
- ^
Emsley, John (2011).
Nature's Building Blocks: An A-Z Guide to the Elements
(New ed.). New York, NY: Oxford University Press.
ISBN
978-0-19-960563-7
.
- ^
Lundqvist, Robert; Hulet, E. K.; Baisden, T. A.; Nasakkala, Elina; Wahlberg, Olof (1981).
"Electromigration Method in Tracer Studies of Complex Chemistry. II. Hydrated Radii and Hydration Numbers of Trivalent Actinides"
.
Acta Chemica Scandinavica A
.
35
: 653?661.
doi
:
10.3891/acta.chem.scand.35a-0653
.
- ^
Hussonnois, H.; Hubert, S.; Aubin, L.;
Guillaumont, R.
; Boussieres, G. (1972).
Radiochem. Radioanal. Lett
.
10
: 231?238.
- ^
Thompson, S. G.; Harvey, B. G.; Choppin, G. R.;
Seaborg, G. T.
(1954).
"Chemical Properties of Elements 99 and 100"
.
J. Am. Chem. Soc
.
76
(24): 6229?6236.
doi
:
10.1021/ja01653a004
.
- ^
Maly, Jaromir (1967). "The amalgamation behaviour of heavy elements 1. Observation of anomalous preference in formation of amalgams of californium, einsteinium, and fermium".
Inorg. Nucl. Chem. Lett
.
3
(9): 373?381.
doi
:
10.1016/0020-1650(67)80046-1
.
- ^
Mikheev, N. B.; Spitsyn, V. I.; Kamenskaya, A. N.; Gvozdec, B. A.; Druin, V. A.; Rumer, I. A.; Dyachkova, R. A.; Rozenkevitch, N. A.; Auerman, L. N. (1972). "Reduction of fermium to divalent state in chloride aqueous ethanolic solutions".
Inorg. Nucl. Chem. Lett
.
8
(11): 929?936.
doi
:
10.1016/0020-1650(72)80202-2
.
- ^
Hulet, E. K.; Lougheed, R. W.; Baisden, P. A.; Landrum, J. H.; Wild, J. F.; Lundqvist, R. F. (1979). "Non-observance of monovalent Md".
J. Inorg. Nucl. Chem
.
41
(12): 1743?1747.
doi
:
10.1016/0022-1902(79)80116-5
.
- ^
Dictionary of Inorganic Compounds
. Vol. 3 (1 ed.). Chapman & Hall. 1992. p. 2873.
ISBN
0412301202
.
- ^
Mikheev, N. B.; Spitsyn, V. I.; Kamenskaya, A. N.; Konovalova, N. A.; Rumer, I. A.; Auerman, L. N.; Podorozhnyi, A. M. (1977). "Determination of oxidation potential of the pair Fm
2+
/Fm
3+
".
Inorg. Nucl. Chem. Lett
.
13
(12): 651?656.
doi
:
10.1016/0020-1650(77)80074-3
.
- ^
Nugent, L. J. (1975).
MTP Int. Rev. Sci.: Inorg. Chem
.
7
: 195?219.
- ^
Samhoun, K.; David, F.; Hahn, R. L.; O'Kelley, G. D.; Tarrant, J. R.; Hobart, D. E. (1979). "Electrochemical study of mendelevium in aqueous solution: No evidence for monovalent ions".
J. Inorg. Nucl. Chem
.
41
(12): 1749?1754.
doi
:
10.1016/0022-1902(79)80117-7
.
- ^
Koch, Lothar (2000). "Transuranium Elements".
Transuranium Elements, in Ullmann's Encyclopedia of Industrial Chemistry
. Wiley.
doi
:
10.1002/14356007.a27_167
.
ISBN
978-3527306732
.
Further reading
[
edit
]
- Robert J. Silva:
Fermium, Mendelevium, Nobelium, and Lawrencium
, in: Lester R. Morss, Norman M. Edelstein, Jean Fuger (Hrsg.):
The Chemistry of the Actinide and Transactinide Elements
, Springer, Dordrecht 2006;
ISBN
1-4020-3555-1
, p. 1621?1651;
doi
:
10.1007/1-4020-3598-5_13
.
- Seaborg, Glenn T.
(ed.) (1978)
Proceedings of the Symposium Commemorating the 25th Anniversary of Elements 99 and 100
, 23 January 1978, Report LBL-7701
- Gmelins Handbuch der anorganischen Chemie
, System Nr. 71, Transurane: Teil A 1 II, p. 19?20; Teil A 2, p. 47; Teil B 1, p. 84.
External links
[
edit
]
Wikimedia Commons has media related to
Fermium
.
Look up
fermium
in Wiktionary, the free dictionary.