Dimethylformamide

|
 Ball and stick model of dimethylformamide Ball and stick model of dimethylformamide
|
 Spacefill model of dimethylformamide Spacefill model of dimethylformamide
|
|
| Names
|
Preferred IUPAC name
N
,
N
-Dimethylformamide
[2]
|
Other names
Dimethylformamide
N
,
N
-Dimethylmethanamide
[1]
DMF
|
| Identifiers
|
|
|
|
|
|
|
| 3DMet
|
|
|
|
605365
|
| ChEBI
|
|
| ChEMBL
|
|
| ChemSpider
|
|
| DrugBank
|
|
| ECHA InfoCard
|
100.000.617

|
| EC Number
|
|
| KEGG
|
|
| MeSH
|
Dimethylformamide
|
|
|
|
| RTECS number
|
|
| UNII
|
|
| UN number
|
2265
|
|
|
|
InChI=1S/C3H7NO/c1-4(2)3-5/h3H,1-2H3
 Y Y
Key: ZMXDDKWLCZADIW-UHFFFAOYSA-N
 Y Y
|
|
|
| Properties
|
|
|
C
3
H
7
N
O
|
| Molar mass
|
73.095
g·mol
?1
|
| Appearance
|
Colourless liquid
|
| Odor
|
Odorless, fishy if impure
|
| Density
|
0.948 g/mL
|
| Melting point
|
?61 °C (?78 °F; 212 K)
|
| Boiling point
|
153 °C (307 °F; 426 K)
|
|
|
Miscible
|
| log
P
|
?0.829
|
| Vapor pressure
|
516 Pa
|
| Acidity
(p
K
a
)
|
?0.3 (for the conjugate acid) (H
2
O)
[3]
|
| UV-vis
(λ
max
)
|
270 nm
|
| Absorbance
|
1.00
|
|
|
1.4305 (at 20 °C)
|
| Viscosity
|
0.92 mPa·s (at 20 °C)
|
| Structure
|
|
|
3.86 D
|
| Thermochemistry
|
|
|
146.05 J/(K·mol)
|
|
|
?239.4?±?1.2 kJ/mol
|
|
|
?1.9416?±?0.0012 MJ/mol
|
| Hazards
|
| GHS
labelling
:
|
|
|
  
|
|
|
Danger
|
|
|
H226
,
H312
,
H319
,
H332
,
H360
|
|
|
P280
,
P305+P351+P338
,
P308+P313
|
| NFPA 704
(fire diamond)
|
|
| Flash point
|
58 °C (136 °F; 331 K)
|
|
|
445 °C (833 °F; 718 K)
|
| Explosive limits
|
2.2?15.2%
|
|
|
30 mg/m
3
(TWA)
|
| Lethal dose
or concentration (LD, LC):
|
|
|
- 1.5 g/kg
(rabbit, dermal)
- 2.8 g/kg
(rat, oral)
- 3.7 g/kg
(mouse, oral)
- 3.5 g/kg
(rat, oral)
|
|
|
3092 ppm (mouse, 2
h
)
[5]
|
|
|
5000 ppm (rat, 6 h)
[5]
|
| NIOSH
(US health exposure limits):
|
|
|
TWA 10 ppm (30 mg/m
3
) [skin]
[4]
|
|
|
TWA 10 ppm (30 mg/m
3
) [skin]
[4]
|
|
|
500 ppm
[4]
|
| Related compounds
|
Related alkanamides
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25 °C [77 °F], 100 kPa).
|
Chemical compound
Dimethylformamide
is an
organic compound
with the
chemical formula
H
C
O
N
(CH
3
)
2
. Its structure is
HC(=O)?N(
?CH
3
)
2
. Commonly abbreviated as DMF (although this initialism is sometimes used for
dimethylfuran
, or
dimethyl fumarate
), this colourless liquid is
miscible
with
water
and the majority of organic liquids. DMF is a common
solvent
for
chemical reactions
. Dimethylformamide is odorless, but
technical-grade
or degraded samples often have a fishy smell due to impurity of
dimethylamine
. Dimethylamine degradation impurities can be removed by
sparging
samples with an inert gas such as
argon
or by
sonicating
the samples under reduced pressure. As its name indicates, it is structurally related to
formamide
, having two
methyl groups
in the place of the two hydrogens. DMF is a
polar
(
hydrophilic
)
aprotic solvent
with a high
boiling point
. It facilitates reactions that follow polar mechanisms, such as
S
N
2
reactions.
Structure and properties
[
edit
]
As for most
amides
, the spectroscopic evidence indicates partial double bond character for the C?N and C?O bonds. Thus, the
infrared spectrum
shows a C=O stretching frequency at only 1675 cm
?1
, whereas a ketone would absorb near 1700 cm
?1
.
[6]
DMF is a classic example of a
fluxional molecule
.
[7]

The ambient temperature
1
H NMR spectrum
shows two methyl signals, indicative of hindered rotation about the (O)C?N bond.
[6]
At temperatures near 100 °C, the 500 MHz NMR spectrum of this compound shows only one signal for the methyl groups.
DMF is miscible with water.
[8]
The vapour pressure at 20 °C is 3.5 hPa.
[9]
A
Henry's law
constant of 7.47 × 10
?5
hPa·m
3
/mol can be deduced from an experimentally determined equilibrium constant at 25 °C.
[10]
The
partition coefficient
log
P
OW
is measured to ?0.85.
[11]
Since the density of DMF (0.95 g·cm
−3
at 20 °C
[8]
) is similar to that of water, significant flotation or stratification in surface waters in case of accidental losses is not expected.
 Left: two resonance structures of DMF. Right: illustration highlighting delocalization.
Left: two resonance structures of DMF. Right: illustration highlighting delocalization.
Reactions
[
edit
]
DMF is hydrolyzed by strong acids and bases, especially at elevated temperatures. With
sodium hydroxide
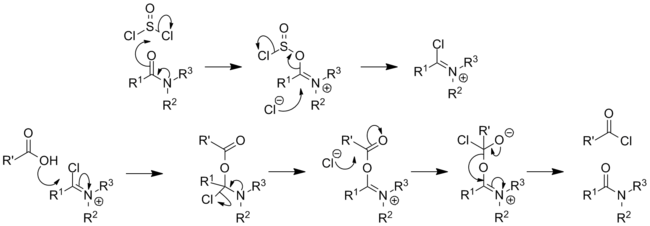
, DMF converts to formate and dimethylamine. DMF undergoes
decarbonylation
near its boiling point to give dimethylamine. Distillation is therefore conducted under reduced pressure at lower temperatures.
[12]
In one of its main uses in
organic synthesis
, DMF is a reagent in the
Vilsmeier?Haack reaction
, which is used to formylate aromatic compounds.
[13]
[14]
The process involves initial conversion of DMF to a chloroiminium ion, [(CH
3
)
2
N=CH(Cl)]
+
, known as a
Vilsmeier reagent
,
[15]
which attacks arenes.
Organolithium compounds
and
Grignard reagents
react with DMF to give
aldehydes
after hydrolysis in a reaction called
Bouveault aldehyde synthesis
.
[16]
Dimethylformamide forms 1:1
adducts
with a variety of Lewis acids such as the soft acid
I
2
, and the hard acid
phenol
. It is classified as a
hard Lewis base
and its
ECW model
base parameters are E
B
= 2.19 and C
B
= 1.31.
[17]
Its relative donor strength toward a series of acids, versus other Lewis bases, can be illustrated by
C-B plots
.
[18]
[19]
Production
[
edit
]
DMF was first prepared in 1893 by the French chemist Albert Verley (8 January 1867 ? 27 November 1959), by distilling a mixture of dimethylamine hydrochloride and potassium formate.
[20]
DMF is prepared by combining
methyl formate
and
dimethylamine
or by reaction of dimethylamine with
carbon monoxide
.
[21]
Although currently impractical, DMF can be prepared from
supercritical carbon dioxide
using
ruthenium
-based catalysts.
[22]
Applications
[
edit
]
The primary use of DMF is as a solvent with low evaporation rate. DMF is used in the production of
acrylic fibers
and
plastics
. It is also used as a solvent in
peptide coupling
for pharmaceuticals, in the development and production of
pesticides
, and in the manufacture of
adhesives
, synthetic
leathers
, fibers, films, and surface coatings.
[8]
- DMF penetrates most
plastics
and makes them swell. Because of this property DMF is suitable for
solid phase peptide synthesis
and as a component of
paint strippers
.
- DMF is used as a solvent to recover
olefins
such as
1,3-butadiene
via
extractive distillation
.
- It is used in the manufacturing of solvent dyes as an important raw material. It is consumed during reaction.
- Pure
acetylene
gas cannot be compressed and stored without the danger of explosion. Industrial acetylene is safely compressed in the presence of dimethylformamide, which forms a safe, concentrated solution. The casing is also filled with
agamassan
, which renders it safe to transport and use.
As a cheap and common reagent, DMF has many uses in a research laboratory.
- DMF is effective at separating and suspending
carbon nanotubes
, and is recommended by the
NIST
for use in
near infrared spectroscopy
of such.
[29]
- DMF can be utilized as a standard in proton NMR spectroscopy allowing for a quantitative determination of an unknown compound.
- In the synthesis of organometallic compounds, it is used as a source of
carbon monoxide
ligands.
- DMF is a common solvent used in
electrospinning
.
- DMF is commonly used in the solvothermal synthesis of
metal?organic frameworks
.
- DMF-
d
7
in the presence of a catalytic amount of
potassium
tert
-butoxide
under microwave heating is a reagent for deuteration of polyaromatic hydrocarbons.
Safety
[
edit
]
Dimethylformamide vapor exposure has shown reduced alcohol tolerance and skin irritation in some cases.
[30]
On 20 June 2018, the
Danish Environmental Protective Agency
published an article about DMF's use in
squishies
. The density of the compound in the toy resulted in all squishies being removed from the Danish market. All squishies were recommended to be thrown out as household waste.
[31]
Toxicity
[
edit
]
The acute
LD50
(oral, rats and mice) is 2.2?7.55 g/kg.
[8]
Hazards of DMF have been examined.
[32]
References
[
edit
]
- ^
N
,
N
-Dimethylmethanamide
, NIST web thermo tables
- ^
Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book)
. Cambridge:
The Royal Society of Chemistry
. 2014. pp. 841, 844.
doi
:
10.1039/9781849733069-FP001
.
ISBN
978-0-85404-182-4
.
The traditional name 'formamide' is retained for HCO-NH
2
and is the preferred IUPAC name. Substitution is permitted on the ?NH
2
group.
- ^
"Hazardous Substances Data Bank (HSDB) - N,N-DIMETHYLFORMAMIDE"
.
- ^
a
b
c
NIOSH Pocket Guide to Chemical Hazards.
"#0226"
.
National Institute for Occupational Safety and Health
(NIOSH).
- ^
a
b
"Dimethylformamide"
.
Immediately Dangerous to Life or Health Concentrations (IDLH)
.
National Institute for Occupational Safety and Health
(NIOSH).
- ^
a
b
"Dimethylformamide"
.
Spectral Database for Organic Compounds
. Japan: AIST
. Retrieved
2012-06-28
.
[
permanent dead link
]
- ^
H. S. Gutowsky; C. H. Holm (1956). "Rate Processes and Nuclear Magnetic Resonance Spectra. II. Hindered Internal Rotation of Amides".
J. Chem. Phys
.
25
(6): 1228?1234.
Bibcode
:
1956JChPh..25.1228G
.
doi
:
10.1063/1.1743184
.
- ^
a
b
c
d
Bipp, H.; Kieczka, H. "Formamides".
Ullmann's Encyclopedia of Industrial Chemistry
. Weinheim: Wiley-VCH.
doi
:
10.1002/14356007.a12_001.pub2
.
ISBN
978-3527306732
.
- ^
IPCS (International Programme on Chemical Safety) (1991). Environmental Health Criteria 114 "Dimethylformamide" United Nations Environment Programme, International Labour Organisation, World Health Organization; 1?124.
- ^
Taft, R. W.; Abraham, M. H.; Doherty, R. M.; Kamlet, M. J. (1985). "The molecular properties governing solubilities of organic nonelectrolytes in water".
Nature
.
313
(6001): 384?386.
Bibcode
:
1985Natur.313..384T
.
doi
:
10.1038/313384a0
.
S2CID
36740734
.
- ^
(BASF AG, department of analytical, unpublished data, J-No. 124659/08, 27.11.1987)
- ^
Comins, Daniel L.; Joseph, Sajan P. (2001). "N,N-Dimethylformamide".
N
,
N
-Dimethylformamide
.
Encyclopedia of Reagents for Organic Synthesis
.
John Wiley & Sons
.
doi
:
10.1002/047084289x.rd335
.
ISBN
9780470842898
.
- ^
a
b
Vilsmeier, Anton
; Haack, Albrecht (1927). "Uber die Einwirkung von Halogenphosphor auf Alkyl-formanilide. Eine neue Methode zur Darstellung sekundarer und tertiarer
p
-Alkylamino-benzaldehyde" [On the reaction of phosphorus halides with alkyl formanilides. A new method for the preparation of secondary and tertiary
p
-alkylamino-benzaldehyde].
Ber. Dtsch. Chem. Ges. A/B
(in German).
60
(1): 119?122.
doi
:
10.1002/cber.19270600118
.
- ^
a
b
Meth-Cohn, Otto; Stanforth, Stephen P. (1993). "The Vilsmeier-Haack Reaction". In
Trost, Barry M.
;
Heathcock, Clayton H.
(eds.).
Additions to CX π-Bonds, Part 2
. Comprehensive Organic Synthesis: Selectivity, Strategy and Efficiency in Modern Organic Chemistry. Vol. 2.
Elsevier
. pp. 777?794.
doi
:
10.1016/B978-0-08-052349-1.00049-4
.
ISBN
9780080405933
.
- ^
Jones, Gurnos; Stanforth, Stephen P. (2000). "The Vilsmeier Reaction of Non-Aromatic Compounds".
Org. React.
56
(2): 355?686.
doi
:
10.1002/0471264180.or056.02
.
- ^
Wang, Zerong (2009).
Comprehensive organic name reactions and reagents
. Hoboken, N.J.: John Wiley. pp. 490?492.
ISBN
9780471704508
.
- ^
Vogel G. C.; Drago, R. S. (1996). "The ECW Model".
Journal of Chemical Education
.
73
(8): 701?707.
Bibcode
:
1996JChEd..73..701V
.
doi
:
10.1021/ed073p701
.
- ^
Laurence, C. and Gal, J-F. Lewis Basicity and Affinity Scales, Data and Measurement, (Wiley 2010) pp 50-51 ISBN 978-0-470-74957-9
- ^
Cramer, R. E.; Bopp, T. T. (1977). "Graphical display of the enthalpies of adduct formation for Lewis acids and bases".
Journal of Chemical Education
.
54
: 612?613.
doi
:
10.1021/ed054p612
.
The plots shown in this paper used older parameters. Improved E&C parameters are listed in
ECW model
.
- ^
Verley, A. (1893).
"Sur la preparation des amides en general"
[On the preparation of amides in general].
Bulletin de la Societe Chimique de Paris
. 3rd series (in French).
9
: 690?692.
On p. 692, Verley states that DMF is prepared by a procedure analogous to that for the preparation of dimethylacetamide (see p. 691), which would be by distilling dimethylamine hydrochloride and
potassium formate
.
- ^
Weissermel, K.; Arpe, H.-J. (2003).
Industrial Organic Chemistry: Important Raw Materials and Intermediates
. Wiley-VCH. pp. 45?46.
ISBN
3-527-30578-5
.
- ^
Walter Leitner; Philip G. Jessop (1999).
Chemical synthesis using supercritical fluids
. Wiley-VCH. pp. 408?.
ISBN
978-3-527-29605-7
. Retrieved
27 June
2011
.
- ^
Bouveault, Louis
(1904).
"Modes de formation et de preparation des aldehydes saturees de la serie grasse"
[Methods of preparation of saturated aldehydes of the aliphatic series].
Bulletin de la Societe Chimique de Paris
. 3rd series (in French).
31
: 1306?1322.
- ^
Bouveault, Louis
(1904).
"Nouvelle methode generale synthetique de preparation des aldehydes"
[Novel general synthetic method for preparing aldehydes].
Bulletin de la Societe Chimique de Paris
. 3rd series (in French).
31
: 1322?1327.
- ^
Li, Jie Jack (2014).
"Bouveault aldehyde synthesis"
.
Name Reactions: A Collection of Detailed Mechanisms and Synthetic Applications
(5th ed.).
Springer Science & Business Media
. pp. 72?73.
ISBN
978-3-319-03979-4
.
- ^
Oestreich, Martin, ed. (2009).
The Mizoroki–Heck Reaction
.
John Wiley & Sons
.
ISBN
9780470716069
.
- ^
Clayden, J. (2001).
Organic Chemistry
. Oxford: Oxford University Press. pp.
276?296
.
ISBN
0-19-850346-6
.
- ^
Ansell, M. F. in "The Chemistry of Acyl Halides"; S. Patai, Ed.; John Wiley and Sons: London, 1972; pp 35?68.
- ^
Haddon, R.; Itkis, M. (March 2008).
"3. Near-Infrared (NIR) Spectroscopy"
(pdf)
. In Freiman, S.; Hooker, S.; Migler; K.; Arepalli, S. (eds.).
Publication 960-19 Measurement Issues in Single Wall Carbon Nanotubes
. NIST. p. 20
. Retrieved
2012-06-28
.
- ^
Lyle, W. H.; Spence, T. W.; McKinneley, W. M.; Duckers, K. (1979).
"Dimethylformamide and alcohol intolerance"
.
British Journal of Industrial Medicine
.
36
(1): 63?66.
doi
:
10.1136/oem.36.1.63
.
PMC
1008494
.
PMID
444443
.
- ^
Magnus Løfstedt.
"Skumlegetøj afgiver farlige kemikalier (in English- Squishies giving dangerous chemicals)"
. Archived from
the original
on 2021-09-03
. Retrieved
2019-06-13
.
- ^
Redlich, C.; Beckett, W. S.; Sparer, J.; Barwick, K. W.; Riely, C. A.; Miller, H.; Sigal, S. L.; Shalat, S. L.; Cullen, M. R. (1988). "Liver disease associated with occupational exposure to the solvent dimethylformamide".
Annals of Internal Medicine
.
108
(5): 680?686.
doi
:
10.7326/0003-4819-108-5-680
.
PMID
3358569
.
External links
[
edit
]