Coordination complex of a metal and cyclopentadienyl groups
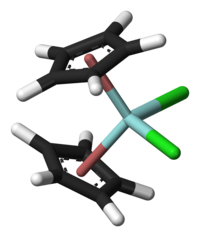 Zirconocene dichloride
, a
cyclopentadienyl complex
Zirconocene dichloride
, a
cyclopentadienyl complex
A
cyclopentadienyl complex
is a
coordination complex
of a
metal
and
cyclopentadienyl groups
(
C
5
H
?
5
, abbreviated as
Cp
?
). Cyclopentadienyl ligands almost invariably bind to metals as a
pentahapto
(
η
5
-) bonding mode. The metal?cyclopentadienyl interaction is typically drawn as a single line from the metal center to the center of the Cp ring.
[1]
[2]
Examples
[
edit
]
Bis
cyclopentadienyl complexes are called
metallocenes
. A famous example of this type of complex is
ferrocene
(FeCp
2
), which has many analogues for other metals, such as
chromocene
(CrCp
2
),
cobaltocene
(CoCp
2
), and
nickelocene
(NiCp
2
). When the Cp rings are mutually parallel the compound is known as a
sandwich
complex
. This area of
organometallic chemistry
was first developed in the 1950s. Bent metallocenes are represented by compounds of the type [MCp
2
L
x
]. Some are catalysts for
ethylene
polymerization
.
[3]
Metallocenes are often thermally stable, and find use as
catalysts
in various types of reactions.
Mixed-ligand Cp complexes containing Cp ligand and one or more other ligands. They are more numerous. One widely studied example is the
Fp dimer
, (Cp
2
Fe
2
(CO)
4
). Monometallic compounds featuring only one Cp ring are often known as
half sandwich compounds
or as piano stool compounds, one example being
methylcyclopentadienylmanganese tricarbonyl
(CpMn(CO)
3
).
Bonding modes
[
edit
]
All 5 carbon atoms of a Cp ligand are bound to the metal in the vast majority of M?Cp complexes. This bonding mode is called
η
5
-coordination
. The M?Cp bonding arises from overlap of the five π molecular orbitals of the Cp ligand with the s, p, and d orbitals on the metal. These complexes are referred to as π-complexes. Almost all of the
transition metals
employ this coordination mode.
[1]
In relatively rare cases, Cp binds to metals via only one carbon center. These types of interactions are described as σ-complexes because they only have a
σ bond
between the metal and the cyclopentadienyl group. Typical examples of this type of complex are group 14 metal complexes such as CpSiMe
3
. An example of both is (Cp
2
Fe(CO)
2
). It is probable that
η
1
-Cp complexes are intermediates in the formation of
η
5
-Cp complexes.
Still rarer, the Cp unit can bond to the metal via three carbons. In these
η
3
-Cp complexes, the bonding resembles that in
allyl ligands
. Such complexes, sometimes called "slipped Cp complexes", are invoked as intermediates in
ring slipping reactions
.
Moreover, inverse sandwich compounds with the "metal?Cp?metal" structures are known.
[4]
Synthesis of Cp complexes
[
edit
]
The compounds are generally prepared by
salt metathesis reactions
of alkali-metal cyclopentadienyl compounds with transition metal chlorides. Sodium cyclopentadienide (NaCp) and
lithium cyclopentadienide
are commonly used.
Trimethylsilylcyclopentadiene
cyclopentadienylthallium
(CpTl) are alternative sources.
[1]
For the preparation of some particularly robust complexes, e.g. nickelocene,
cyclopentadiene
is employed in the presence of a conventional
base
such as KOH. When only a single Cp ligand is installed, the other
ligands
typically carbonyl, halogen, alkyl, and hydride.
Most Cp complexes are prepared by substitution of preformed Cp complexes by replacement of halide, CO, and other simple ligands.
Variations of Cp complexes
[
edit
]
Ansa Cp ligands
[
edit
]
A pair of cyclopentadienyl ligands can be covalently linked giving rise to so-call ansa metallocenes. The angle between the two Cp rings is fixed. Rotation of the rings about the metal-centroid axis is stopped as well. A related class of derivatives give rise to the
constrained geometry complexes
. In these cases, a Cp ligand as linked to a non-Cp ligand. Such complexes have been commercialized for the production of polypropylene.
Bulky Cp ligands
[
edit
]
Pentamethylcyclopentadiene
gives rise to pentamethylcyclopentadienyl (Cp*) complexes. These ligands are more basic and more lipophilic. Replacing methyl groups with larger substituents results in cyclopentadienes that are so encumbered that pentaalkyl derivatives are no longer possible. Well-studied ligands of this type include C
5
R
4
H
?
(R = iso-Pr) and 1,2,4-C
5
R
3
H
2
?
(R =
tert
-Bu).
Constrained geometry complexes
[
edit
]
Constrained geometry complexes are related to ansa-metallocenes except that one ligand is not Cp-related.
Applications
[
edit
]
Cp metal complexes are mainly used as stoichiometric reagents in chemical research. Ferrocenium reagents are oxidants. Cobaltocene is a strong, soluble reductant.
Derivatives of
Cp
2
TiCl
2
and
Cp
2
ZrCl
2
are the basis of some reagents in
organic synthesis
. Upon treatment with
aluminoxane
, these dihalides give catalysts for
olefin polymerization
. Such species are called
Kaminsky-type catalysts
.
References
[
edit
]
- ^
a
b
c
Elschenbroich, C. "Organometallics" (2006) Wiley-VCH: Weinheim.
ISBN
978-3-527-29390-2
- ^
Yamamoto, A. (1986).
Organotransition Metal Chemistry: Fundamental Concepts and Applications
. New York, NY: Wiley-Interscience. p. 105.
[
ISBN missing
]
- ^
Crabtree, R. H. (2001).
The Organometallic Chemistry of the Transition Metals
(3rd ed.). New York, NY: John Wiley & Sons.
[
ISBN missing
]
- ^
Yu, Chao; Liang, Jiefeng; Deng, Chong; Lefevre, Guillaume; Cantat, Thibault;
Diaconescu, Paula L.
; Huang, Wenliang (2020).
"Arene-Bridged Dithorium Complexes: Inverse Sandwiches Supported by a δ Bonding Interaction"
(PDF)
.
Journal of the American Chemical Society
.
142
(51): 21292?21297.
doi
:
10.1021/jacs.0c11215
.
ISSN
0002-7863
.
PMID
33315367
.
S2CID
229180362
.
Further reading
[
edit
]
- Shriver, D.; Atkins, P. W. (1999).
Inorganic Chemistry
. New York, NY: W. H. Freeman.
[
ISBN missing
]
- King, R. B.; Bisnette, M. B. (1967). "Organometallic chemistry of the transition metals XXI. Some π-pentamethylcyclopentadienyl derivatives of various transition metals".
J. Organomet. Chem
.
8
(2): 287?297.
doi
:
10.1016/S0022-328X(00)91042-8
.
[Initial examples of the synthesis of Cp*-metal complexes]
|
|---|
| H donors:
| |
|---|
| B donors:
| |
|---|
| C donors:
| |
|---|
| Si donors:
| |
|---|
| N donors:
| |
|---|
| P donors:
| |
|---|
| O donors:
| |
|---|
| S donors:
| |
|---|
| Halide donors:
| |
|---|
|
|---|
| Principles
| |
|---|
| Reactions
| |
|---|
| Types of compounds
| |
|---|
| Applications
| |
|---|
| Related branches of
chemistry
| |
|---|