"CoCl₂" redirects here. For the compound with molecular formula COCl₂, see
Phosgene
.
Cobalt(II) chloride
 Anhydrous
|
 Hexahydrate
|
|

Structure of anhydrous compound
|
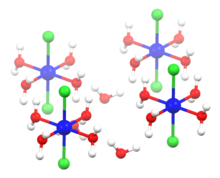
Structure of hexahydrate
|
| Names
|
| IUPAC name
Cobalt(II) chloride
|
Other names
Cobaltous chloride
Cobalt dichloride
Muriate of cobalt
[1]
|
| Identifiers
|
|
|
|
|
|
|
| ChEBI
|
|
| ChemSpider
|
|
| ECHA InfoCard
|
100.028.718

|
| EC Number
|
|
|
|
|
| RTECS number
|
|
| UNII
|
|
| UN number
|
3288
|
|
|
|
InChI=1S/2ClH.Co/h2*1H;/q;;+2/p-2
 Y Y
Key: GVPFVAHMJGGAJG-UHFFFAOYSA-L
 Y Y
InChI=1/2ClH.Co/h2*1H;/q;;+2/p-2
Key: GVPFVAHMJGGAJG-NUQVWONBAU
|
|
|
| Properties
|
|
|
CoCl
2
|
| Molar mass
|
129.839 g/mol (anhydrous)
165.87 g/mol (dihydrate)
237.93 g/mol (hexahydrate)
|
| Appearance
|
blue crystals (anhydrous)
violet-blue (dihydrate)
rose red crystals (hexahydrate)
|
| Density
|
3.356 g/cm
3
(anhydrous)
2.477 g/cm
3
(dihydrate)
1.924 g/cm
3
(hexahydrate)
|
| Melting point
|
726 °C (1,339 °F; 999 K) ±2 (anhydrous)
[2]
140 °C (monohydrate)
100 °C (dihydrate)
86 °C (hexahydrate)
|
| Boiling point
|
1,049 °C (1,920 °F; 1,322 K)
|
|
|
43.6 g/100 mL (0 °C)
45 g/100 mL (7 °C)
52.9 g/100 mL (20 °C)
105 g/100 mL (96 °C)
|
| Solubility
|
38.5 g/100 mL (methanol)
8.6 g/100 mL (acetone)
soluble in
ethanol
,
pyridine
,
glycerol
|
|
|
+12,660·10
?6
cm
3
/mol
|
| Structure
|
|
|
CdCl
2
structure
|
|
|
hexagonal (anhydrous)
monoclinic (dihydrate)
Octahedral
(hexahydrate)
|
| Hazards
|
| GHS
labelling
:
|
|
|
  
|
| NFPA 704
(fire diamond)
|
|
| Flash point
|
Non-flammable
|
| Lethal dose
or concentration (LD, LC):
|
|
|
80 mg/kg (rat, oral)
|
| Safety data sheet
(SDS)
|
ICSC 0783
|
| Related compounds
|
|
|
Cobalt(II) fluoride
Cobalt(II) bromide
Cobalt(II) iodide
|
|
|
Rhodium(III) chloride
Iridium(III) chloride
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25 °C [77 °F], 100 kPa).
|
Chemical compound
Cobalt(II) chloride
is an
inorganic compound
, a salt of
cobalt
and
chlorine
, with the formula
CoCl
2
. The compound forms several
hydrates
CoCl
2
·
n
H
2
O
, for
n
= 1, 2, 6, and 9. Claims of the formation of tri- and tetrahydrates have not been confirmed.
[4]
The anhydrous form is a blue crystalline solid; the dihydrate is purple and the hexahydrate is pink. Commercial samples are usually the hexahydrate, which is one of the most commonly used cobalt salts in the lab.
[5]
Properties
[
edit
]
Anhydrous
[
edit
]
At room temperature, anhydrous cobalt chloride has the
cadmium chloride
structure (
CdCl
2
) (R
3
m) in which the cobalt(II) ions are octahedrally coordinated. At about 706 °C (20 degrees below the melting point), the
coordination
is believed to change to tetrahedral.
[2]
The vapor pressure has been reported as 7.6
mmHg
at the melting point.
[6]
Solutions
[
edit
]
Cobalt chloride is fairly
soluble
in water. Under atmospheric pressure, the
mass concentration
of a
saturated solution
of
CoCl
2
in water is about 54% at the boiling point, 120.2 °C; 48% at 51.25 °C; 35% at 25 °C; 33% at 0 °C; and 29% at ?27.8 °C.
[4]
Diluted
aqueous
solutions of
CoCl
2
contain the species
[Co(H
2
O)
6
]
2+
, besides
chloride
ions. Concentrated solutions are red at room temperature but become blue at higher temperatures.
[7]
Hydrates
[
edit
]
 Subunit of CoCl
2
(H
2
O)
2
lattice.
Subunit of CoCl
2
(H
2
O)
2
lattice.
The crystal unit of the solid hexahydrate
CoCl
2
?6
H
2
O
contains the neutral molecule
trans
-
CoCl
2
(H
2
O)
4
and two molecules of
water of crystallization
.
[8]
This species dissolves readily in water and
alcohol
.
The anhydrous salt is
hygroscopic
and the hexahydrate is
deliquescent
.
[
citation needed
]
The dihydrate, CoCl
2
(H
2
O)
2
, is a
coordination polymer
. Each Co center is coordinated to four doubly
bridging chloride ligands
. The octahedron is completed by a pair of mutually trans
aquo ligands
.
[9]
Preparation
[
edit
]
Cobalt chloride can be prepared in aqueous solution from
cobalt(II) hydroxide
or
cobalt(II) carbonate
and
hydrochloric acid
:
- CoCO
3
+ 2 HCl
(aq)
→
CoCl
2
(aq)
+
CO
2
+
H
2
O
- Co(OH)
2
+ 2 HCl
(aq)
→
CoCl
2
(aq)
+ 2
H
2
O
The solid dihydrate and hexahydrate can be obtained by evaporation. Cooling saturated aqueous solutions yields the dihydrate between 120.2 °C and 51.25 °C, and the hexahydrate below 51.25 °C. Water ice, rather than cobalt chloride, will crystallize from solutions with concentration below 29%. The monohydrate and the anhydrous forms can be obtained by cooling solutions only under high pressure, above 206 °C and 335 °C, respectively.
[4]
The anhydrous compound can be prepared by heating the hydrates.
[10]
On rapid heating or in a closed container, each of the 6-, 2-, and 1- hydrates partially melts into a mixture of the next lower hydrate and a saturated solution?at 51.25 °C, 206 °C, and 335 °C, respectively.
[4]
On slow heating in an open container, so that the water vapor pressure over the solid is practically zero, water evaporates out of each of the solid 6-, 2-, and 1- hydrates, leaving the next lower hydrate, at about 40°C, 89°C, and 125°C, respectively. If the partial pressure of the water vapor is in equilibrium with the solid, as in a confined but not pressurized contained, the decomposition occurs at about 115°C, 145°C, and 195°C, respectively.
[4]
Dehydration can also be effected with
trimethylsilyl chloride
:
[11]
- CoCl
2
?6
H
2
O
+ 12
(CH
3
)
3
SiCl
→
CoCl
2
+ 6
[(CH
3
)
3
SiCl]
2
O
+ 12 HCl
The anhydrous compound can be purified by sublimation in vacuum.
[2]
Reactions
[
edit
]
In the laboratory, cobalt(II) chloride serves as a common precursor to other cobalt compounds. Generally, diluted aqueous solutions of the salt behave like other cobalt(II) salts since these solutions consist of the
[Co(H
2
O)
6
]
2+
ion regardless of the anion. For example, such solutions give a precipitate of
cobalt sulfide
CoS
upon treatment with
hydrogen sulfide
H
2
S
.
[
citation needed
]
Complexed chlorides
[
edit
]
The hexahydrate and the anhydrous salt are weak
Lewis acids
. The
adducts
are usually either
octahedral
or
tetrahedral
. It forms an
octahedral complex with pyridine
(
C
5
H
5
N
):
[12]
- CoCl
2
·6
H
2
O
+ 4
C
5
H
5
N
→
CoCl
2
(C
5
H
5
N)
4
+ 6
H
2
O
With
triphenylphosphine
(
P(C
6
H
5
)
3
), a tetrahedral complex results:
- CoCl
2
·6
H
2
O
+ 2
P(C
6
H
5
)
3
→
CoCl
2
[P(C
6
H
5
)
3
]
2
+ 6
H
2
O
Salts of the anionic complex CoCl
4
2?
can be prepared using tetraethylammonium chloride:
[13]
- CoCl
2
+ 2 [(C
2
H
5
)
4
N]Cl → [(C
2
H
5
)
4
N)]
2
[CoCl
4
]
The tetrachlorocobaltate ion [CoCl
4
]
2?
is the blue ion that forms upon addition of
hydrochloric acid
to aqueous solutions of hydrated cobalt chloride, which are pink.
Reduction
[
edit
]
 The structure of a cobalt(IV)
coordination complex
with the
norbornyl
anion
The structure of a cobalt(IV)
coordination complex
with the
norbornyl
anion
Reaction of the anhydrous compound with
sodium cyclopentadienide
gives
cobaltocene
Co(C
5
H
5
)
2
. This 19-electron species is a good reducing agent, being readily oxidised to the yellow
18-electron
cobaltocenium cation
[Co(C
5
H
5
)
2
]
+
.
Oxidation to cobalt(III)
[
edit
]
Compounds of cobalt in the +3 oxidation state exist, such as
cobalt(III) fluoride
CoF
3
,
nitrate
Co(NO
3
)
3
, and
sulfate
Co
2
(SO
4
)
3
; however,
cobalt(III) chloride
CoCl
3
is not stable in normal conditions, and would decompose immediately into
CoCl
2
and
chlorine
.
[14]
On the other hand, cobalt(III) chlorides can be obtained if the cobalt is bound also to other ligands of greater Lewis basicity than chloride, such as
amines
. For example, in the presence of
ammonia
, cobalt(II) chloride is readily
oxidised
by atmospheric
oxygen
to
hexamminecobalt(III) chloride
:
- 4
CoCl
2
·6
H
2
O
+ 4
NH
4
Cl + 20
NH
3
+
O
2
→ 4
[Co(NH
3
)
6
]Cl
3
+ 26
H
2
O
Similar reactions occur with other
amines
. These reactions are often performed in the presence of
charcoal
as a catalyst, or with
hydrogen peroxide
H
2
O
2
substituted for atmospheric oxygen. Other highly basic ligands, including
carbonate
,
acetylacetonate
, and
oxalate
, induce the formation of Co(III) derivatives. Simple carboxylates and halides do not.
[
citation needed
]
Unlike Co(II)
complexes
, Co(III) complexes are very slow to exchange
ligands
, so they are said to be
kinetically inert
. The German chemist
Alfred Werner
was awarded the
Nobel prize
in 1913 for his studies on a series of these cobalt(III) compounds, work that led to an understanding of the structures of such
coordination compounds
.
[
citation needed
]
Oxidation to cobalt(IV)
[
edit
]
Reaction of 1-norbornyllithium with the
CoCl
2
·THF in pentane produces the brown, thermally stable
tetrakis(1-norbornyl)cobalt(IV)
[15]
[16]
? a rare example of a stable transition metal/saturated alkane compound,
[5]
different products are obtained in other solvents.
[17]
Moisture indication
[
edit
]
 The deep blue colour of this moisture indicating silica gel is due to cobalt chloride. When hydrated the colour changes to a light pink/purple.
The deep blue colour of this moisture indicating silica gel is due to cobalt chloride. When hydrated the colour changes to a light pink/purple.
Cobalt chloride is a common visual moisture indicator due to its distinct colour change when hydrated. The colour change is from some shade of blue when dry, to a pink when hydrated, although the shade of colour depends on the substrate and concentration. It is impregnated into paper to make test strips for detecting moisture in solutions, or more slowly, in air/gas.
Desiccants
such as
silica gel
can incorporate cobalt chloride to indicate when it is "spent" (i.e. hydrated).
[18]
Health issues
[
edit
]
Cobalt is essential for most higher forms of life, but more than a few milligrams each day is harmful. Although poisonings have rarely resulted from cobalt compounds, their chronic ingestion has caused serious health problems at doses far less than the lethal dose. In 1966, the addition of cobalt compounds to stabilize
beer foam
in Canada led to a peculiar form of toxin-induced
cardiomyopathy
, which came to be known as
beer drinker's cardiomyopathy
.
[19]
[20]
[21]
Furthermore, cobalt(II) chloride is suspected of causing
cancer
(i.e., possibly
carcinogenic
,
IARC Group 2B
) as per the
International Agency for Research on Cancer
(IARC) Monographs.
[22]
In 2005?06, cobalt chloride was the eighth-most-prevalent
allergen
in
patch tests
(8.4%).
[23]
Other uses
[
edit
]
- Invisible ink
: when suspended in solution, cobalt(II) chloride can be made to appear invisible on a surface; when that same surface is subsequently exposed to significant heat (such as from a handheld
heat gun
or lighter) the ink reversibly changes to blue.
[24]
- Cobalt chloride is an established chemical inducer of hypoxia-like responses such as
erythropoiesis
.
[
citation needed
]
Cobalt supplementation is not banned and therefore would not be detected by current anti-doping testing.
[25]
Cobalt chloride is a banned substance under the Australian Thoroughbred Racing Board.
[26]
- Cobalt chloride is one method used to induce pulmonary arterial hypertension in animals for research and evaluation of treatment efficacy.
References
[
edit
]
- ^
"Cobalt muriate, CAS Number: 7646-79-9"
.
www.chemindustry.com
. Archived from
the original
on 28 May 2019
. Retrieved
19 April
2018
.
- ^
a
b
c
Wojakowska, A.; Krzy?ak, E.; Pli?ska, S. (2007). "Melting and high-temperature solid state transitions in cobalt(II) halides".
Journal of Thermal Analysis and Calorimetry
.
88
(2): 525?530.
doi
:
10.1007/s10973-006-8000-9
.
- ^
Santa Cruz Biotechnology: Cobalt(II) chloride
- ^
a
b
c
d
e
M. T. Saugier, M. Noailly, R. Cohen-Adad, F. Paulik, and J. Paulik (1977): "Equilibres solide ? liquide ? vapeur du systeme binaire
CoCl
2
-
H
2
O
"
Journal of Thermal Analysis
, volume 11, issue 1, pages 87?100.
doi
:
10.1007/BF02104087
Note: the lowest point of fig.6 is inconsistent with fig.7; probably should be at -27.8 C instead of 0 C.
- ^
a
b
Greenwood, Norman N.
; Earnshaw, Alan (1997).
Chemistry of the Elements
(2nd ed.).
Butterworth-Heinemann
.
ISBN
978-0-08-037941-8
.
- ^
Yuzo Saeki, Ryoko Matsuzaki, Naomi Aoyama (1977): "The vapor pressure of cobalt dichloride".
Journal of the Less Common Metals
, volume 55, issue 2, pages 289-291.
doi
:
10.1016/0022-5088(77)90204-1
- ^
The Merck Index
, 7th edition, Merck & Co, Rahway, New Jersey, USA, 1960.
- ^
Wells, A. F. (1984),
Structural Inorganic Chemistry
(5th ed.), Oxford: Clarendon Press,
ISBN
0-19-855370-6
- ^
Morosin, B.; Graeber, E. J. (1965). "Crystal structures of manganese(II) and iron(II) chloride dihydrate".
Journal of Chemical Physics
.
42
(3): 898?901.
Bibcode
:
1965JChPh..42..898M
.
doi
:
10.1063/1.1696078
.
- ^
John Dallas Donaldson, Detmar Beyersmann, "Cobalt and Cobalt Compounds" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005.
doi
:
10.1002/14356007.a07_281.pub2
- ^
Philip Boudjouk; Jeung-Ho So (2007). "Solvated and Unsolvated Anhydrous Metal Chlorides from Metal Chloride Hydrates".
Inorganic Syntheses
. Vol. 29. pp. 108?111.
doi
:
10.1002/9780470132609.ch26
.
ISBN
9780470132609
.
- ^
Long, Gary J.; Clarke, Peter J. (1978). "Crystal and Molecular Structures of
trans
-Tetrakis(pyridine)dichloroiron(II), -Nickel(II), and -Cobalt(II) and
trans
-Tetrakis(pyridine)dichloroiron(II) Monohydrate".
Inorganic Chemistry
.
17
(6): 1394?1401.
doi
:
10.1021/ic50184a002
.
- ^
Gill, N. S. & Taylor, F. B. (1967). "Tetrahalo Complexes of Dipositive Metals in the First Transition Series".
Inorganic Syntheses
. Vol. 9. pp. 136?142.
doi
:
10.1002/9780470132401.ch37
.
ISBN
9780470132401
.
- ^
Handbook of Chemistry and Physics
, 71st edition, CRC Press, Ann Arbor, Michigan, 1990.
- ^
Barton K. Bower & Howard G. Tennent (1972). "Transition metal bicyclo[2.2.1]hept-1-yls".
J. Am. Chem. Soc.
94
(7): 2512?2514.
doi
:
10.1021/ja00762a056
.
- ^
Erin K. Byrne; Darrin S. Richeson & Klaus H. Theopold (1986). "Tetrakis(1-norbornyl)cobalt, a low spin tetrahedral complex of a first row transition metal".
J. Chem. Soc., Chem. Commun.
(19): 1491?1492.
doi
:
10.1039/C39860001491
.
- ^
Erin K. Byrne; Klaus H. Theopold (1989). "Synthesis, characterization, and electron-transfer reactivity of norbornyl complexes of cobalt in unusually high oxidation states".
J. Am. Chem. Soc.
111
(11): 3887?3896.
doi
:
10.1021/ja00193a021
.
- ^
Solomon, M. E. (February 1945).
"The use of cobalt salts as indicators of humidity and moisture"
.
Annals of Applied Biology
.
32
(1): 75?85.
doi
:
10.1111/j.1744-7348.1945.tb06763.x
.
ISSN
0003-4746
.
- ^
Morin Y; T?tu A; Mercier G (1969). "Quebec beer-drinkers' cardiomyopathy: Clinical and hemodynamic aspects".
Annals of the New York Academy of Sciences
.
156
(1): 566?576.
Bibcode
:
1969NYASA.156..566M
.
doi
:
10.1111/j.1749-6632.1969.tb16751.x
.
PMID
5291148
.
S2CID
7422045
.
- ^
Barceloux, Donald G. & Barceloux, Donald (1999). "Cobalt".
Clinical Toxicology
.
37
(2): 201?216.
doi
:
10.1081/CLT-100102420
.
PMID
10382556
.
- ^
11.1.5 The unusual type of myocardiopathy recognized in 1965 and 1966 in Quebec (Canada), Minneapolis (Minnesota), Leuven (Belgium), and Omaha (Nebraska) was associated with episodes of acute heart failure (e/g/, 50 deaths among 112 beer drinkers).
- ^
[PDF
- ^
Zug KA, Warshaw EM, Fowler JF Jr, Maibach HI, Belsito DL, Pratt MD, Sasseville D, Storrs FJ, Taylor JS, Mathias CG, Deleo VA, Rietschel RL, Marks J. Patch-test results of the North American Contact Dermatitis Group 2005?2006. Dermatitis. 2009 May?Jun;20(3):149-60.
- ^
"Making invisible ink"
.
Royal Society of Chemistry
. Retrieved
7 April
2023
.
- ^
Lippi G, Franchini M, Guidi GC (November 2005).
"Cobalt chloride administration in athletes: a new perspective in blood doping?"
.
Br J Sports Med
.
39
(11): 872?873.
doi
:
10.1136/bjsm.2005.019232
.
PMC
1725077
.
PMID
16244201
.
- ^
Bartley, Patrick (6 February 2015).
"Cobalt crisis turns the eyes of the world onto Australian racing"
.
The Sydney Morning Herald
.
External links
[
edit
]
|
|---|
| Cobalt(I)
| |
|---|
| Cobalt(II)
| |
|---|
| Cobalt(0, III)
| |
|---|
| Cobalt(II, III)
| |
|---|
| Cobalt(III)
| |
|---|
| Cobalt(III,IV)
| |
|---|
| Cobalt(IV)
| |
|---|
| Cobalt(V)
| |
|---|
Salts and covalent derivatives of the
chloride
ion
|
|---|
|