Chromium(III) chloride

Anhydrous
|

Hexahydrate
|
| Names
|
IUPAC name
- Chromium(III) chloride
- Chromium trichloride
|
| Other names
Chromic chloride
|
| Identifiers
|
|
|
|
|
|
|
| ChEBI
|
|
| ChEMBL
|
|
| ChemSpider
|
|
| DrugBank
|
|
| ECHA InfoCard
|
100.030.023

|
|
|
1890 130477 532690
|
|
|
|
| RTECS number
|
|
| UNII
|
|
|
|
|
InChI=1S/3ClH.Cr/h3*1H;/q;;;+3/p-3
 Y Y
Key: QSWDMMVNRMROPK-UHFFFAOYSA-K
 Y Y
InChI=1/3ClH.Cr/h3*1H;/q;;;+2/p-3
Key: HUQISNLCWMVGCG-DFZHHIFOAJ
InChI=1/3ClH.Cr/h3*1H;/q;;;+3/p-3
Key: QSWDMMVNRMROPK-DFZHHIFOAF
|
|
|
| Properties
|
|
|
CrCl
3
|
| Molar mass
|
158.36 g/mol (anhydrous)
266.45 g/mol (hexahydrate)
[1]
|
| Appearance
|
Purple (anhydrous), dark green (hexahydrate)
|
| Density
|
2.87 g/cm
3
(anhydrous)
1.760 g/cm
3
(hexahydrate)
|
| Melting point
|
1,152 °C (2,106 °F; 1,425 K) (anhydrous)
81 °C (hexahydrate)
[2]
|
| Boiling point
|
1,300 °C (2,370 °F; 1,570 K) decomposes
|
|
|
slightly soluble (anhydrous)
585 g/L (hexahydrate)
|
| Solubility
|
insoluble in
ethanol
insoluble in
ether
,
acetone
|
| Acidity
(p
K
a
)
|
2.4 (0.2M solution)
|
|
|
+6890.0·10
?6
cm
3
/mol
|
| Structure
|
|
|
YCl
3
structure
|
|
|
Octahedral
|
| Hazards
|
| GHS
labelling
:
|
|
|
  
|
|
|
Danger
|
|
|
H302
,
H314
,
H411
|
|
|
P260
,
P264
,
P270
,
P273
,
P280
,
P301+P312
,
P301+P330+P331
,
P303+P361+P353
,
P304+P340
,
P305+P351+P338
,
P310
,
P321
,
P330
,
P363
,
P391
,
P405
,
P501
|
| NFPA 704
(fire diamond)
|
|
| Flash point
|
Non-flammable
|
| Lethal dose
or concentration (LD, LC):
|
|
|
1870 mg/kg (oral, rat)
[4]
|
| NIOSH
(US health exposure limits):
|
|
|
TWA 1 mg/m
3
[3]
|
|
|
TWA 0.5 mg/m
3
[3]
|
|
|
250 mg/m
3
[3]
|
| Safety data sheet
(SDS)
|
ICSC 1316 (anhydrous)
ICSC 1532 (hexahydrate)
|
| Related compounds
|
|
|
|
|
|
|
Related compounds
|
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25 °C [77 °F], 100 kPa).
|
Chemical compound
Chromium(III) chloride
(also called
chromic chloride
) is an
inorganic chemical compound
with the
chemical formula
Cr
Cl
3
. It forms several
hydrates
with the formula
CrCl
3
·
n
H
2
O
, among which are hydrates where
n
can be 5 (chromium(III) chloride pentahydrate
CrCl
3
·5H
2
O
) or 6 (chromium(III) chloride hexahydrate
CrCl
3
·6H
2
O
). The
anhydrous
compound with the formula
CrCl
3
are violet
crystals
, while the most common form of the chromium(III) chloride are the dark green crystals of hexahydrate,
CrCl
3
·6H
2
O
. Chromium chlorides find use as
catalysts
and as precursors to dyes for wool.
Structure
[
edit
]
Anhydrous chromium(III) chloride adopts the
YCl
3
structure,
[6]
with
Cr
3+
occupying one third of the
octahedral
interstices in alternating layers of a pseudo-
cubic close packed
lattice of
Cl
?
ions. The absence of cations in alternate layers leads to weak bonding between adjacent layers. For this reason, crystals of
CrCl
3
cleave
easily along the planes between layers, which results in the flaky (
micaceous
) appearance of samples of chromium(III) chloride.
[7]
[8]
The anhydrous
CrCl
3
is exfoliable down to the monolayer limit.
[6]
If pressurized to 9.9
GPa
it goes under a phase transition.
[9]
-
Space-filling model
of cubic close packing of chloride ions in the crystal structure of
CrCl
3
-
-
Stacking of layers
Chromium(III) chloride hydrates
[
edit
]
The hydrated chromium(III) chlorides display the somewhat unusual property of existing in a number of distinct chemical forms (isomers), which differ in terms of the number of chloride anions that are coordinated to Cr(III) and the
water of crystallization
. The different forms exist both as solids and in
aqueous solutions
. Several members are known of the series of
[CrCl
3?
q
(H
2
O)
n
]
q
+
. The common hexahydrate can be more precisely described as
[CrCl
2
(H
2
O)
4
]Cl·2H
2
O
. It consists of the cation
trans
-
[CrCl
2
(H
2
O)
4
]
+
and additional molecules of water and a chloride anion in the lattice.
[10]
Two other hydrates are known, pale green
[CrCl(H
2
O)
5
]Cl
2
·H
2
O
and violet
[Cr(H
2
O)
6
]Cl
3
. Similar
hydration isomerism
is seen with other chromium(III) compounds.
[
citation needed
]
Preparation
[
edit
]
Anhydrous chromium(III) chloride may be prepared by
chlorination
of
chromium
metal directly, or indirectly by
carbothermic
chlorination of
chromium(III) oxide
at 650?800 °C
[11]
[12]
- Cr
2
O
3
+ 3 C + 3 Cl
2
→ 2 CrCl
3
+ 3 CO
The hydrated chlorides are prepared by treatment of chromate with hydrochloric acid and aqueous methanol.
Reactions
[
edit
]
Slow reaction rates are common with chromium(III) complexes. The low reactivity of the d
3
Cr
3+
ion can be explained using
crystal field theory
. One way of opening
CrCl
3
up to substitution in solution is to reduce even a trace amount to
CrCl
2
, for example using
zinc
in
hydrochloric acid
. This chromium(II) compound undergoes substitution easily, and it can exchange electrons with
CrCl
3
via a
chloride
bridge, allowing all of the
CrCl
3
to react quickly. With the presence of some chromium(II), solid
CrCl
3
dissolves rapidly in water. Similarly,
ligand
substitution reactions of solutions of
[CrCl
2
(H
2
O)
4
]
+
are accelerated by chromium(II) catalysts.
With molten
alkali metal
chlorides
such as
potassium chloride
,
CrCl
3
gives salts of the type
M
3
[CrCl
6
]
and
K
3
[Cr
2
Cl
9
]
, which is also octahedral but where the two chromiums are linked via three chloride bridges.
The hexahydrate can also be dehydrated with
thionyl chloride
:
[13]
- CrCl
3
·6H
2
O + 6 SOCl
2
→ CrCl
3
+ 6 SO
2
+ 12
HCl
Complexes with organic ligands
[
edit
]
CrCl
3
is a
Lewis acid
, classified as "hard" according to the
Hard-Soft Acid-Base theory
. It forms a variety of adducts of the type
[CrCl
3
L
3
]
q
+
, where L is a
Lewis base
. For example, it reacts with
pyridine
(
C
5
H
5
N
) to form the
pyridine complex
:
- CrCl
3
+ 3 C
5
H
5
N → CrCl
3
(C
5
H
5
N)
3
Treatment with
trimethylsilylchloride
in
THF
gives the anhydrous THF complex:
[14]
- CrCl
3
·6H
2
O + 12 (CH
3
)
3
SiCl → CrCl
3
(THF)
3
+ 6 ((CH
3
)
3
Si)
2
O + 12 HCl
Precursor to organochromium complexes
[
edit
]
Chromium(III) chloride is used as the precursor to many
organochromium compounds
, for example
bis(benzene)chromium
, an analogue of
ferrocene
:
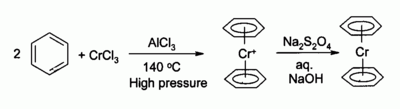
Phosphine complexes derived from
CrCl
3
catalyse the trimerization of
ethylene
to
1-hexene
.
[15]
[16]
Use in organic synthesis
[
edit
]
One niche use of
CrCl
3
in
organic synthesis
is for the
in situ
preparation of
chromium(II) chloride
, a reagent for the reduction of
alkyl halides
and for the synthesis of (
E
)-alkenyl halides. The reaction is usually performed using two
moles
of
CrCl
3
per mole of
lithium aluminium hydride
, although if aqueous acidic conditions are appropriate
zinc
and
hydrochloric acid
may be sufficient.

Chromium(III) chloride has also been used as a Lewis acid in organic reactions, for example to catalyse the nitroso
Diels-Alder reaction
.
[17]
Dyestuffs
[
edit
]
A number of chromium-containing dyes are used commercially for wool. Typical dyes are
triarylmethanes
consisting of ortho-hydroxylbenzoic acid derivatives.
[18]
Precautions
[
edit
]
Although
trivalent
chromium is far less poisonous than
hexavalent
, chromium salts are generally considered toxic.
References
[
edit
]
- ^
"Chromium(III) chloride sublimation, 99 10025-73-7"
.
- ^
"Chromium(III) chloride hexahydrate Technipur™ | Sigma-Aldrich"
. Retrieved
2022-08-16
.
- ^
a
b
c
NIOSH Pocket Guide to Chemical Hazards.
"#0141"
.
National Institute for Occupational Safety and Health
(NIOSH).
- ^
"Chromium(III) compounds [as Cr(III)]"
.
Immediately Dangerous to Life or Health Concentrations (IDLH)
.
National Institute for Occupational Safety and Health
(NIOSH).
- ^
Cameo Chemicals MSDS
- ^
a
b
Kazim, S; Ali, M; Palleschi, S; D’Olimpio, G; Mastrippolito, D; Politano, A; Gunnella, R; Di Cicco, A; Renzelli, M; Moccia, G; Cacioppo, O A; Alfonsetti, R; Strychalska-Nowak, J; Klimczuk, T; J Cava, R (2020-07-06).
"Mechanical exfoliation and layer number identification of single crystal monoclinic CrCl
3
"
.
Nanotechnology
.
31
(39): 395706.
doi
:
10.1088/1361-6528/ab7de6
.
hdl
:
11581/438597
.
ISSN
0957-4484
.
- ^
Greenwood, Norman N.
; Earnshaw, Alan (1997).
Chemistry of the Elements
(2nd ed.).
Butterworth-Heinemann
. p. 1020.
ISBN
978-0-08-037941-8
.
- ^
A. F. Wells,
Structural Inorganic Chemistry
, 5th ed., Oxford University Press, Oxford, UK, 1984.
- ^
Meiling Hong (2022). "Pressure-Induced Structural Phase Transition and Metallization of CrCl3 under Different Hydrostatic Environments up to 50.0 GPa".
Inorg. Chem
.
61
(12): 4852?4864.
doi
:
10.1021/acs.inorgchem.1c03486
.
PMID
35289613
.
S2CID
247452267
.
- ^
Ian G. Dance, Hans C. Freeman "The Crystal Structure of Dichlorotetraaquochromium(III) Chloride Dihydrate: Primary and Secondary Metal Ion Hydration" Inorganic Chemistry 1965, volume 4, 1555?1561.
doi
:
10.1021/ic50033a006
- ^
D. Nicholls,
Complexes and First-Row Transition Elements
, Macmillan Press, London, 1973.
- ^
Brauer, Georg (1965) [1962].
Handbuch Der Praparativen Anorganischen Chemie
[
Handbook of Preparative Inorganic Chemistry
] (in German). Vol. 2. Stuttgart; New York, New York: Ferdinand Enke Verlag; Academic Press, Inc. p. 1340.
ISBN
978-0-32316129-9
. Retrieved
2014-01-10
.
- ^
Pray, A. P. (1990). "Anhydrous Metal Chlorides".
Inorganic Syntheses
. Inorganic Syntheses. Vol. 28. pp. 321?2.
doi
:
10.1002/9780470132401.ch36
.
ISBN
9780470132401
.
- ^
Boudjouk, Philip; So, Jeung-Ho (1992). "Solvated and Unsolvated Anhydrous Metal Chlorides from Metal Chloride Hydrates".
Inorganic Syntheses
. Inorganic Syntheses. Vol. 29. pp. 108?111.
doi
:
10.1002/9780470132609.ch26
.
ISBN
9780470132609
.
- ^
John T. Dixon, Mike J. Green, Fiona M. Hess, David H. Morgan "Advances in selective ethylene trimerisation ? a critical overview" Journal of Organometallic Chemistry 2004, Volume 689, pp 3641-3668.
doi
:
10.1016/j.jorganchem.2004.06.008
- ^
Feng Zheng, Akella Sivaramakrishna, John R. Moss "Thermal studies on metallacycloalkanes" Coordination Chemistry Reviews 2007, Volume 251, 2056-2071.
doi
:
10.1016/j.ccr.2007.04.008
- ^
Calvet, G.; Dussaussois, M.; Blanchard, N.; Kouklovsky, C. (2004). "Lewis Acid-Promoted Hetero Diels-Alder Cycloaddition of α-Acetoxynitroso Dienophiles".
Organic Letters
.
6
(14): 2449?2451.
doi
:
10.1021/ol0491336
.
PMID
15228301
.
- ^
Thomas Gessner and Udo Mayer "Triarylmethane and Diarylmethane Dyes" in
Ullmann's Encyclopedia of Industrial Chemistry
2002,
Wiley-VCH
, Weinheim.
doi
:
10.1002/14356007.a27_179
Further reading
[
edit
]
- Handbook of Chemistry and Physics
, 71st edition, CRC Press, Ann Arbor, Michigan, 1990.
- The Merck Index
, 7th edition, Merck & Co, Rahway, New Jersey, USA, 1960.
- J. March,
Advanced Organic Chemistry
, 4th ed., p. 723, Wiley, New York, 1992.
- K. Takai, in
Handbook of Reagents for Organic Synthesis, Volume 1: Reagents, Auxiliaries and Catalysts for C-C Bond Formation
, (R. M. Coates, S. E. Denmark, eds.), pp. 206?211, Wiley, New York, 1999.
External links
[
edit
]
|
|---|
| Chromium(0)
| | Organochromium(0) compounds
| |
|---|
|
|---|
| Chromium(I)
| | Organochromium(I) compounds
| |
|---|
|
|---|
| Chromium(II)
| | Organochromium(II) compounds
| |
|---|
|
|---|
| Chromium(II, III)
| |
|---|
| Chromium(III)
| |
|---|
| Chromium(IV)
| |
|---|
| Chromium(V)
| |
|---|
| Chromium(VI)
| |
|---|
| Polyatomic ion
| |
|---|
Salts and covalent derivatives of the
chloride
ion
|
|---|
|