Any chemical compound having two acyl groups bonded to the same oxygen atom
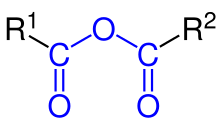 Generic example of an anhydride of two carboxylic acids
R
1
COOH
and
R
2
COOH
, with the blue marked carboxylic acid anhydride functional group, where R stands for any group (typically
hydrogen
or
organyl
).
Generic example of an anhydride of two carboxylic acids
R
1
COOH
and
R
2
COOH
, with the blue marked carboxylic acid anhydride functional group, where R stands for any group (typically
hydrogen
or
organyl
).
An
organic acid anhydride
is an
acid anhydride
that is also an
organic compound
. An acid anhydride is a compound that has two
acyl groups
bonded
to the same
oxygen
atom.
[1]
A common type of organic acid anhydride is a
carboxylic anhydride
, where the parent acid is a
carboxylic acid
, the formula of the anhydride being (RC(O))
2
O. Symmetrical acid anhydrides of this type are named by replacing the word
acid
in the name of the parent carboxylic acid by the word
anhydride
.
[2]
Thus, (CH
3
CO)
2
O is called
acetic anhydride.
Mixed
(or
unsymmetrical
) acid anhydrides, such as
acetic formic anhydride
(see below), are known, whereby reaction occurs between two different carboxylic acids. Nomenclature of unsymmetrical acid anhydrides list the names of both of the reacted carboxylic acids before the word "anhydride" (for example, the dehydration reaction between benzoic acid and propanoic acid would yield "benzoic propanoic anhydride").
[3]
One or both acyl groups of an acid anhydride may also be derived from another type of
organic acid
, such as
sulfonic acid
or a
phosphonic acid
. One of the acyl groups of an acid anhydride can be derived from an
inorganic acid
such as
phosphoric acid
. The mixed anhydride
1,3-bisphosphoglyceric acid
, an intermediate in the formation of ATP via
glycolysis
,
[4]
is the mixed anhydride of
3-phosphoglyceric acid
and
phosphoric acid
.
Acidic oxides
are also classified as acid anhydrides.
Nomenclature
[
edit
]
The nomenclature of organic acid anhydrides is derived from the names of the constituent carboxylic acids. In symmetrical acid anhydrides, only the prefix of the original carboxylic acid is used and the suffix "anhydride" is added. For most unsymmetrical acid anhydrides - also called mixed anhydrides- the prefixes from both acids reacted are listed before the suffix, e.g., benzoic propanoic anhydride.
[5]
Preparation
[
edit
]
Organic acid anhydrides are prepared in industry by diverse means.
Acetic anhydride
is mainly produced by the
carbonylation
of
methyl acetate
.
[6]
Maleic anhydride
is produced by the oxidation of
benzene
or
butane
. Laboratory routes emphasize the dehydration of the corresponding acids. The conditions vary from acid to acid, but
phosphorus pentoxide
is a common
dehydrating agent
:
- 2
CH
3
COOH
+ P
4
O
10
→
CH
3
C(O)OC(O)CH
3
+ "P
4
O
9
(OH)
2
"
Acid chlorides
are also effective precursors:
[7]
- CH
3
C(O)Cl +
HCO
2
Na
→ HCO
2
COCH
3
+ NaCl
Mixed anhydrides containing the acetyl group are prepared from
ketene
:
- RCO
2
H + H
2
C=C=O → RCO
2
C(O)CH
3
Reactions
[
edit
]
Acid anhydrides are a source of reactive acyl groups, and their reactions and uses resemble those of
acyl halides
. In reactions with protic substrates, the reactions afford equal amounts of the acylated product and the carboxylic acid:
- RC(O)OC(O)R + HY → RC(O)Y + RCO
2
H
for HY = HOR (alcohols), HNR'
2
(ammonia, primary, secondary amines), aromatic ring (see
Friedel-Crafts acylation
).
Acid anhydrides tend to be less
electrophilic
than
acyl chlorides
, and only one acyl group is transferred per molecule of acid anhydride, which leads to a lower
atom efficiency
. The low cost, however, of
acetic anhydride
makes it a common choice for
acetylation
reactions.
Applications and occurrence of acid anhydrides
[
edit
]
Acetic anhydride
is a major industrial chemical widely used for preparing acetate esters, e.g.
cellulose acetate
.
Maleic anhydride
is the precursor to various
resins
by
copolymerization
with
styrene
. Maleic anhydride is a dienophile in the
Diels-Alder reaction
.
[8]
Dianhydrides, molecules containing two acid anhydride functions, are used to synthesize
polyimides
and sometimes
polyesters
[9]
and
polyamides
.
[10]
Examples of dianhydrides: pyromellitic dianhydride (PMDA), 3,3’, 4,4’ - oxydiphtalic dianhydride (ODPA),
3,3’, 4,4’-benzophenone tetracarboxylic dianhydride (BTDA)
, 4,4’-diphtalic (hexafluoroisopropylidene) anhydride (6FDA),
benzoquinonetetracarboxylic dianhydride
,
ethylenetetracarboxylic dianhydride
.
Polyanhydrides
are a class of
polymers
characterized by anhydride bonds that connect
repeat units
of the polymer
backbone chain
.
Biological occurrence
[
edit
]
Natural products containing acid anhydrides have been isolated from animals, bacteria and fungi.
[11]
[12]
[13]
Examples include
cantharidin
from species of blister beetle, including the
Spanish fly
,
Lytta vesicatoria
, and
tautomycin
, from the bacterium
Streptomyces spiroverticillatus
. The maleidride family of fungal secondary metabolites, which possess a wide range of antibiotic and antifungal activity, are
alicyclic compounds
with maleic anhydride functional groups.
[14]
A number of proteins in prokaryotes
[15]
and eukaryotes
[16]
undergo spontaneous cleavage between the amino acid residues
aspartic acid
and
proline
via an acid anhydride intermediate. In some cases, the anhydride may then react with nucleophiles of other cellular components, such as at the surface of the bacterium
Neisseria meningitidis
or on proteins localized nearby.
[17]
Analogues
[
edit
]
Nitrogen
[
edit
]
 A general linear imide functional group
A general linear imide functional group
Imides
are structurally related analogues, where the bridging oxygen is replaced by nitrogen. They are similarly formed by the condensation of dicarboxylic acids with ammonia. The replacement of all oxygen atoms with nitrogen gives
imidines
, these are a rare functional group which are very prone to hydrolysis.
Sulfur
[
edit
]
Sulfur
can replace oxygen, either in the carbonyl group or in the bridge. In the former case, the name of the acyl group is enclosed in parentheses to avoid ambiguity in the name,
[2]
e.g., (thioacetic) anhydride (CH
3
C(S)OC(S)CH
3
). When two acyl groups are attached to the same sulfur atom, the resulting compound is called a thioanhydride,
[2]
e.g., acetic thioanhydride ((CH
3
C(O))
2
S).
See also
[
edit
]
References
[
edit
]
- ^
IUPAC
,
Compendium of Chemical Terminology
, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "
acid anhydrides
".
doi
:
10.1351/goldbook.A00072
- ^
a
b
c
Panico, R.; Powell, W. H.; Richer, J. C., eds. (1993).
"Recommendation R-5.7.7"
.
A Guide to IUPAC Nomenclature of Organic Compounds
.
IUPAC
/
Blackwell Science
. pp. 123?25.
ISBN
0-632-03488-2
.
- ^
"Nomenclature of Anhydrides"
. 8 November 2013.
- ^
Nelson, D. L.; Cox, M. M. "Lehninger, Principles of Biochemistry" 3rd Ed. Worth Publishing: New York, 2000.
ISBN
1-57259-153-6
.
- ^
"Nomenclature of Anhydrides"
. 8 November 2013.
- ^
Zoeller, J. R.; Agreda, V. H.; Cook, S. L.; Lafferty, N. L.; Polichnowski, S. W.; Pond, D. M. "Eastman Chemical Company Acetic Anhydride Process" Catalysis Today (1992), volume 13, pp.73-91.
doi
:
10.1016/0920-5861(92)80188-S
- ^
Lewis I. Krimen (1988).
"Acetic Formic Anhydride"
.
Organic Syntheses
;
Collected Volumes
, vol. 6, p. 8
.
- ^
Heimo Held, Alfred Rengstl, Dieter Mayer "Acetic Anhydride and Mixed Fatty Acid Anhydrides" Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim.
doi
:
10.1002/14356007.a01_065
- ^
Chiang, Wen-Yen; Chiang, Wen-Chang (1988-05-05).
"Condensation polymerization of multifunctional monomers and properties of related polyester resins. II. Thermal properties of polyester?imide varnishes"
.
Journal of Applied Polymer Science
.
35
(6): 1433?1439.
doi
:
10.1002/app.1988.070350603
.
- ^
Faghihi, Khalil; Ashouri, Mostafa; Hajibeygi, Mohsen (2013-10-25).
"High Temperature and Organosoluble Poly(amide-imide)s Based on 1,4-Bis[4-aminophenoxy]butane and Aromatic Diacids by Direct Polycondensation: Synthesis and Properties"
.
High Temperature Materials and Processes
.
32
(5): 451?458.
doi
:
10.1515/htmp-2012-0164
.
ISSN
2191-0324
.
- ^
Saleem, Muhammad; Hussain, Hidayat; Ahmed, Ishtiaq; Draeger, Siegfried; Schulz, Barbara; Meier, Kathrin; Steinert, Michael; Pescitelli, Gennaro; Kurtan, Tibor; Florke, Ulrich; Krohn, Karsten (February 2011). "Viburspiran, an Antifungal Member of the Octadride Class of Maleic Anhydride Natural Products".
European Journal of Organic Chemistry
.
2011
(4): 808?812.
doi
:
10.1002/ejoc.201001324
.
- ^
Han, Chunguang; Furukawa, Hiroyuki; Tomura, Tomohiko; Fudou, Ryosuke; Kaida, Kenichi; Choi, Bong-Keun; Imokawa, Genji; Ojika, Makoto (24 April 2015). "Bioactive Maleic Anhydrides and Related Diacids from the Aquatic Hyphomycete Tricladium castaneicola".
Journal of Natural Products
.
78
(4): 639?644.
doi
:
10.1021/np500773s
.
PMID
25875311
.
- ^
Heard, David M.; Tayler, Emyr R.; Cox, Russell J.; Simpson, Thomas J.; Willis, Christine L. (3 January 2020).
"Structural and synthetic studies on maleic anhydride and related diacid natural products"
(PDF)
.
Tetrahedron
.
76
(1): 130717.
doi
:
10.1016/j.tet.2019.130717
.
hdl
:
1983/53998d06-9017-4cfb-822b-c6453348000a
.
S2CID
209714625
.
- ^
Chen, Xiaolong; Zheng, Yuguo; Shen, Yinchu (May 2007). "Natural Products with Maleic Anhydride Structure: Nonadrides, Tautomycin, Chaetomellic Anhydride, and Other Compounds".
Chemical Reviews
.
107
(5): 1777?1830.
doi
:
10.1021/cr050029r
.
PMID
17439289
.
- ^
Kuban, Vojtech (2020).
"Structural Basis of Ca 2+-Dependent Self-Processing Activity of Repeat-in-Toxin Proteins"
.
mBio
.
11
(2): e00226-20.
doi
:
10.1128/mBio.00226-20
.
PMC
7078468
.
PMID
32184239
.
- ^
Bell, Christian (2013).
"Structure of the repulsive guidance molecule (RGM)-neogenin signaling hub"
.
Science
.
341
(6141): 77?80.
Bibcode
:
2013Sci...341...77B
.
doi
:
10.1126/science.1232322
.
PMC
4730555
.
PMID
23744777
.
- ^
Scheu, Arne (2021).
"NeissLock provides an inducible protein anhydride for covalent targeting of endogenous proteins"
.
Nature Communications
.
12
(1): 717.
Bibcode
:
2021NatCo..12..717S
.
doi
:
10.1038/s41467-021-20963-5
.
PMC
7846742
.
PMID
33514717
.
External links
[
edit
]
|
|---|
Hydrocarbons
(only C and H)
| |
|---|
Only
carbon
,
hydrogen
,
and
oxygen
(only C, H and O)
| |
|---|
Only one
element,
not being
carbon,
hydrogen,
or oxygen
(one element,
not C, H or O)
| |
|---|
| Other
| |
|---|
|