Hydrogen that is produced biologically
 Microbial hydrogen production.
Microbial hydrogen production.
Biohydrogen
is
H
2
that is produced biologically.
[1]
Interest is high in this technology because H
2
is a
clean fuel
and can be readily produced from certain kinds of
biomass
,
[2]
including biological waste.
[3]
Furthermore some
photosynthetic
microorganisms are capable to produce H
2
directly from water splitting using light as energy source.
[4]
[5]
Besides the promising possibilities of biological hydrogen production, many challenges characterize this technology. First challenges include those intrinsic to H
2
, such as storage and transportation of an
explosive
noncondensible gas. Additionally, hydrogen producing organisms are
poisoned by O
2
and yields of H
2
are often low.
Biochemical principles
[
edit
]
The main reactions driving hydrogen formation involve the oxidation of substrates to obtain electrons. Then, these electrons are transferred to free
protons
to form molecular hydrogen. This proton reduction reaction is normally performed by an enzyme family known as
hydrogenases
.
In heterotrophic organisms, electrons are produced during the
fermentation
of sugars.
Hydrogen
gas is produced in many types of fermentation as a way to regenerate NAD
+
from
NADH
.
Electrons
are transferred to
ferredoxin
, or can be directly accepted from NADH by a
hydrogenase
, producing H
2
. Because of this most of the reactions start with
glucose
, which is converted to
acetic acid
.
[6]

A related reaction gives
formate
instead of
carbon dioxide
:

These reactions are exergonic by 216 and 209 kcal/mol, respectively.
It has been estimated that 99% of all organisms utilize or produce
dihydrogen
(H
2
). Most of these species are microbes and their ability to use or produce H
2
as a metabolite arises from the expression of H
2
metalloenzymes
known as hydrogenases.
[7]
Enzymes within this widely diverse family are commonly sub-classified into three different types based on the active site metal content: [FeFe]-hydrogenases (iron-iron), [NiFe]-hydrogenases (nickel-iron) hydrogenases, and [Fe]-hydrogenases (iron-only).
[8]
Many organisms express these enzymes. Notable examples are members of the genera
Clostridium
,
Desulfovibrio
,
Ralstonia
or the pathogen
Helicobacter
, being most of them strict-anaerobes or facultative microorganisms. Other microorganisms such
green algae
also express highly active hydrogenases, as it is the case for members of the genera
Chlamydomonas
.
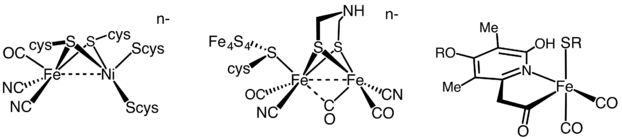 The active site structures of the three types of hydrogenase enzymes.
The active site structures of the three types of hydrogenase enzymes.
Due to the extreme diversity of hydrogenase enzymes, on-going efforts are focused on screening for novel enzymes with improved features,
[9]
[10]
[11]
as well as engineering already characterized hydrogenases to confer them more desirable characteristics.
[12]
Production by algae
[
edit
]
The
biological hydrogen production
with
algae
is a method of photobiological
water splitting
which is done in a
closed
photobioreactor
based on the
production of hydrogen
as a
solar fuel
by
algae
.
[13]
[14]
Algae
produce hydrogen under certain conditions. In 2000 it was discovered that if
C. reinhardtii
algae are deprived of
sulfur
they will switch from the production of
oxygen
, as in normal
photosynthesis
, to the production of hydrogen.
[15]
[16]
[17]
Green algae express [FeFe] hydrogenases, being some of them considered the most efficient hydrogenases with turnover rates superior to 10
4
s
?1
. This remarkable catalytic efficiency is nonetheless shadowed by its extreme sensitivity to oxygen, being irreversibly inactivated by O
2
[12]
.
When the cells are deprived from sulfur,
oxygen evolution
stops due to photo-damage of
photosystem II
, in this state the cells start consuming O
2
and provide the ideal anaerobic environment for the native [FeFe] hydrogenases to catalyze H
2
production.
Photosynthesis
[
edit
]
Photosynthesis
in
cyanobacteria
and
green algae
splits water into hydrogen ions and electrons. The electrons are transported over
ferredoxins
.
[19]
Fe-Fe-hydrogenases
(enzymes) combine them into hydrogen gas. In
Chlamydomonas reinhardtii
Photosystem II
produces in direct conversion of sunlight 80% of the electrons that end up in the hydrogen gas.
[20]
In 2020 scientists reported the development of algal-cell based micro-emulsion for multicellular
spheroid
microbial reactors
capable of producing
hydrogen
alongside either oxygen or CO
2
via photosynthesis in daylight under air. Enclosing the microreactors with synergistic bacteria was shown to increase levels of hydrogen production via reduction of O
2
concentrations.
[21]
[18]
Improving production by light harvesting antenna reduction
[
edit
]
The
chlorophyll
(Chl) antenna size in green algae is minimized, or truncated, to maximize photobiological solar conversion efficiency and H
2
production. It has been shown that
Light-harvesting complex
photosystem II light-harvesting protein
LHCBM9 promotes efficient light energy dissipation.
[22]
The truncated Chl antenna size minimizes absorption and wasteful dissipation of sunlight by individual cells, resulting in better light utilization efficiency and greater photosynthetic efficiency when the green alga are grown as a mass culture in bioreactors.
[23]
Economics
[
edit
]
With current reports for algae-based biohydrogen, it would take about 25,000 square kilometre algal farming to produce biohydrogen equivalent to the energy provided by gasoline in the US alone. This area represents approximately 10% of the area devoted to growing soya in the US.
[24]
Bioreactor design issues
[
edit
]
- Restriction of photosynthetic hydrogen production by accumulation of a
proton gradient
.
- Competitive inhibition of photosynthetic hydrogen production by carbon dioxide.
- Requirement for bicarbonate binding at
photosystem II
(PSII) for
efficient photosynthetic activity
.
- Competitive drainage of electrons by oxygen in algal hydrogen production.
- Economics must reach competitive price to other sources of energy and the economics are dependent on several parameters.
- A major technical obstacle is the efficiency in converting solar energy into chemical energy stored in molecular hydrogen.
Attempts are in progress to solve these problems via
bioengineering
.
Production by cyanobacteria
[
edit
]
Biological hydrogen production
is also observed in
nitrogen-fixing
cyanobacteria
. This microorganisms can grow forming filaments. Under nitrogen-limited conditions some cells can specialize and form
heterocysts
, which ensures an anaerobic intracellular space to ease N
2
fixation by the
nitrogenase
enzyme expressed also inside.
Under nitrogen-fixation conditions, the nitrogenase enzyme accepts electrons and consume ATP to break the triple dinitrogen bond and reduce it to ammonia.
[25]
During the catalytic cycle of the nitrogenase enzyme, molecular hydrogen is also produced.

Nevertheless, since the production of H
2
is an important loss of energy for the cells, most of nitrogen fixing cyanobacteria also feature at least one uptake hydrogenase.
[26]
Uptake hydrogenases exhibit a catalytic bias towards oxygen oxidation, thus can assimilate the produced H
2
as a way to recover part of the energy invested during the nitrogen fixation process.
History
[
edit
]
In 1933,
Marjory Stephenson
and her student Stickland reported that cell suspensions catalysed the reduction of
methylene blue
with H
2
. Six years later,
Hans Gaffron
observed that the green photosynthetic alga
Chlamydomonas reinhardtii
, would sometimes produce hydrogen.
[27]
In the late 1990s
Anastasios Melis
discovered that deprivation of sulfur induces the alga to switch from the production of oxygen (normal photosynthesis) to the production of hydrogen. He found that the
enzyme
responsible for this reaction is
hydrogenase
, but that the hydrogenase lost this function in the presence of oxygen. Melis also discovered that depleting the amount of sulfur available to the algae interrupted their internal oxygen flow, allowing the hydrogenase an environment in which it can react, causing the algae to produce hydrogen.
[28]
Chlamydomonas moewusii
is also a promising strain for the production of hydrogen.
[29]
[30]
Industrial hydrogen
[
edit
]
Competing for biohydrogen, at least for commercial applications, are many mature industrial processes.
Steam reforming
of
natural gas
- sometimes referred to as steam methane reforming (SMR) - is the most common method of producing bulk hydrogen at about 95% of the world production.
[31]
[32]
[33]

See also
[
edit
]
References
[
edit
]
- ^
M. Rogner, ed. (2015).
Biohydrogen
. De Gruyter.
ISBN
978-3-11-033673-3
.
- ^
Y.-H. Percival Zhang "Hydrogen Production from Carbohydrates: A Mini-Review" in "Sustainable Production of Fuels, Chemicals, and Fibers from Forest Biomass" ACS Symposium Series, 2011, Volume 1067, pages=203-216.
- ^
Wijayasekera, Sachindra Chamode; Hewage, Kasun; Siddiqui, Osamah; Hettiaratchi, Patrick; Sadiq, Rehan (2022-01-29).
"Waste-to-hydrogen technologies: A critical review of techno-economic and socio-environmental sustainability"
.
International Journal of Hydrogen Energy
.
47
(9): 5842?5870.
doi
:
10.1016/j.ijhydene.2021.11.226
.
ISSN
0360-3199
.
S2CID
245348607
.
- ^
Bolatkhan, Kenzhegul; Kossalbayev, Bekzhan D.; Zayadan, Bolatkhan K.; Tomo, Tatsuya; Veziroglu, T. Nejat; Allakhverdiev, Suleyman I. (2019-03-01).
"Hydrogen production from phototrophic microorganisms: Reality and perspectives"
.
International Journal of Hydrogen Energy
.
44
(12): 5799?5811.
doi
:
10.1016/j.ijhydene.2019.01.092
.
ISSN
0360-3199
.
S2CID
104465557
.
- ^
Vasiliadou, Ioanna A.; Berna, Antonio; Manchon, Carlos; Melero, Juan A.; Martinez, Fernando; Esteve-Nunez, Abraham; Puyol, Daniel (2018).
"Biological and Bioelectrochemical Systems for Hydrogen Production and Carbon Fixation Using Purple Phototrophic Bacteria"
.
Frontiers in Energy Research
.
6
.
doi
:
10.3389/fenrg.2018.00107
.
ISSN
2296-598X
.
- ^
Thauer, R. K. (1998).
"Biochemistry of Methanogenesis: a Tribute to Marjory Stephenson"
.
Microbiology
.
144
: 2377?2406.
doi
:
10.1099/00221287-144-9-2377
.
PMID
9782487
.
- ^
Lubitz, Wolfgang
; Ogata, Hideaki; Rudiger, Olaf; Reijerse, Edward (2014). "Hydrogenases".
Chemical Reviews
.
114
(8): 4081?148.
doi
:
10.1021/cr4005814
.
PMID
24655035
.
- ^
Vignais, Paulette M.; Billoud, Bernard (2007-10-01).
"Occurrence, Classification, and Biological Function of Hydrogenases: An Overview"
.
Chemical Reviews
.
107
(10): 4206?4272.
doi
:
10.1021/cr050196r
.
ISSN
0009-2665
.
PMID
17927159
.
- ^
Land, Henrik; Ceccaldi, Pierre; Meszaros, Livia S.; Lorenzi, Marco; Redman, Holly J.; Senger, Moritz; Stripp, Sven T.; Berggren, Gustav (2019-11-06).
"Discovery of novel [FeFe]-hydrogenases for biocatalytic H2-production"
.
Chemical Science
.
10
(43): 9941?9948.
doi
:
10.1039/C9SC03717A
.
ISSN
2041-6539
.
PMC
6984386
.
PMID
32055351
.
- ^
Grinter, Rhys; Kropp, Ashleigh; Venugopal, Hari; Senger, Moritz; Badley, Jack; Cabotaje, Princess R.; Jia, Ruyu; Duan, Zehui; Huang, Ping; Stripp, Sven T.; Barlow, Christopher K.; Belousoff, Matthew; Shafaat, Hannah S.; Cook, Gregory M.; Schittenhelm, Ralf B. (March 2023).
"Structural basis for bacterial energy extraction from atmospheric hydrogen"
.
Nature
.
615
(7952): 541?547.
Bibcode
:
2023Natur.615..541G
.
doi
:
10.1038/s41586-023-05781-7
.
ISSN
1476-4687
.
PMC
10017518
.
PMID
36890228
.
- ^
Morra, Simone (2022).
"Fantastic [FeFe]-Hydrogenases and Where to Find Them"
.
Frontiers in Microbiology
.
13
: 853626.
doi
:
10.3389/fmicb.2022.853626
.
ISSN
1664-302X
.
PMC
8924675
.
PMID
35308355
.
- ^
a
b
Lu, Yuan; Koo, Jamin (November 2019).
"O2 sensitivity and H2 production activity of hydrogenases-A review"
.
Biotechnology and Bioengineering
.
116
(11): 3124?3135.
doi
:
10.1002/bit.27136
.
ISSN
1097-0290
.
PMID
31403182
.
S2CID
199539477
.
- ^
2013 - Gimpel JA, et al Advances in microalgae engineering and synthetic biology applications for biofuel production
- ^
Hemschemeier, Anja; Melis, Anastasios; Happe, Thomas (2009).
"Analytical approaches to photobiological hydrogen production in unicellular green algae"
.
Photosynthesis Research
.
102
(2?3): 523?540.
Bibcode
:
2009PhoRe.102..523H
.
doi
:
10.1007/s11120-009-9415-5
.
ISSN
0166-8595
.
PMC
2777220
.
PMID
19291418
.
- ^
Wired-Mutant Algae Is Hydrogen Factory
Archived
August 27, 2006, at the
Wayback Machine
- ^
"Further reading - New Scientist"
. Archived from
the original
on 2008-10-31
. Retrieved
2009-03-11
.
- ^
Melis, Anastasios; Zhang, Liping; Forestier, Marc; Ghirardi, Maria L.; Seibert, Michael (2000-01-01).
"Sustained Photobiological Hydrogen Gas Production upon Reversible Inactivation of Oxygen Evolution in the Green AlgaChlamydomonas reinhardtii"
.
Plant Physiology
.
122
(1): 127?136.
doi
:
10.1104/pp.122.1.127
.
ISSN
1532-2548
.
PMC
58851
.
PMID
10631256
.
- ^
a
b
Xu, Zhijun; Wang, Shengliang; Zhao, Chunyu; Li, Shangsong; Liu, Xiaoman; Wang, Lei; Li, Mei; Huang, Xin; Mann, Stephen (25 November 2020).
"Photosynthetic hydrogen production by droplet-based microbial micro-reactors under aerobic conditions"
.
Nature Communications
.
11
(1): 5985.
Bibcode
:
2020NatCo..11.5985X
.
doi
:
10.1038/s41467-020-19823-5
.
ISSN
2041-1723
.
PMC
7689460
.
PMID
33239636
.
 Available under
CC BY 4.0
.
Available under
CC BY 4.0
.
- ^
Peden, E. A.; Boehm, M.; Mulder, D. W.; Davis, R.; Old, W. M.; King, P. W.; Ghirardi, M. L.; Dubini, A. (2013).
"Identification of Global Ferredoxin Interaction Networks in Chlamydomonas reinhardtii"
.
Journal of Biological Chemistry
.
288
(49): 35192?35209.
doi
:
10.1074/jbc.M113.483727
.
ISSN
0021-9258
.
PMC
3853270
.
PMID
24100040
.
- ^
Volgusheva, A.; Styring, S.; Mamedov, F. (2013).
"Increased photosystem II stability promotes H2 production in sulfur-deprived Chlamydomonas reinhardtii"
.
Proceedings of the National Academy of Sciences
.
110
(18): 7223?7228.
Bibcode
:
2013PNAS..110.7223V
.
doi
:
10.1073/pnas.1220645110
.
ISSN
0027-8424
.
PMC
3645517
.
PMID
23589846
.
- ^
"Research creates hydrogen-producing living droplets, paving way for alternative future energy source"
.
phys.org
. Retrieved
9 December
2020
.
- ^
Grewe, S.; Ballottari, M.; Alcocer, M.; D'Andrea, C.; Blifernez-Klassen, O.; Hankamer, B.; Mussgnug, J. H.; Bassi, R.; Kruse, O. (2014).
"Light-Harvesting Complex Protein LHCBM9 Is Critical for Photosystem II Activity and Hydrogen Production in Chlamydomonas reinhardtii"
.
The Plant Cell
.
26
(4): 1598?1611.
doi
:
10.1105/tpc.114.124198
.
ISSN
1040-4651
.
PMC
4036574
.
PMID
24706511
.
- ^
Kirst, H.; Garcia-Cerdan, J. G.; Zurbriggen, A.; Ruehle, T.; Melis, A. (2012).
"Truncated Photosystem Chlorophyll Antenna Size in the Green Microalga Chlamydomonas reinhardtii upon Deletion of the TLA3-CpSRP43 Gene"
.
Plant Physiology
.
160
(4): 2251?2260.
doi
:
10.1104/pp.112.206672
.
ISSN
0032-0889
.
PMC
3510145
.
PMID
23043081
.
- ^
Growing hydrogen for the cars of tomorrow
- ^
"5.15C: Nitrogen Fixation Mechanism"
.
Biology LibreTexts
. 2017-05-11
. Retrieved
2023-04-07
.
- ^
Tamagnini, Paula; Axelsson, Rikard; Lindberg, Pia; Oxelfelt, Fredrik; Wunschiers, Robbe; Lindblad, Peter (March 2002).
"Hydrogenases and Hydrogen Metabolism of Cyanobacteria"
.
Microbiology and Molecular Biology Reviews
.
66
(1): 1?20.
doi
:
10.1128/MMBR.66.1.1-20.2002
.
ISSN
1092-2172
.
PMC
120778
.
PMID
11875125
.
- ^
Algae: Power Plant of the Future?
- ^
"Multiplatform Pinup Girl"
.
Wired
. 2002-04-01.
Archived
from the original on 2012-10-20.
- ^
Melis A, Happe T (2001).
"Hydrogen Production. Green Algae as a Source of Energy"
.
Plant Physiol
.
127
(3): 740?748.
doi
:
10.1104/pp.010498
.
PMC
1540156
.
PMID
11706159
.
- ^
Yang, Shihui; Guarnieri, Michael T; Smolinski, Sharon; Ghirardi, Maria; Pienkos, Philip T (2013).
"De novo transcriptomic analysis of hydrogen production in the green alga Chlamydomonas moewusii through RNA-Seq"
.
Biotechnology for Biofuels
.
6
(1): 118.
doi
:
10.1186/1754-6834-6-118
.
ISSN
1754-6834
.
PMC
3846465
.
PMID
23971877
.
- ^
P. Haussinger, R. Lohmuller, A. M. Watson, "Hydrogen, 2. Production" in Ullmann's Encyclopedia of Industrial Chemistry, 2012, Wiley-VCH, Weinheim.
doi
:
10.1002/14356007.o13_o03
- ^
Ogden, J.M. (1999). "Prospects for building a hydrogen energy infrastructure".
Annual Review of Energy and the Environment
.
24
: 227?279.
doi
:
10.1146/annurev.energy.24.1.227
.
- ^
"Hydrogen Production: Natural Gas Reforming"
. Department of Energy
. Retrieved
6 April
2017
.
External links
[
edit
]