Different forms of the element iron
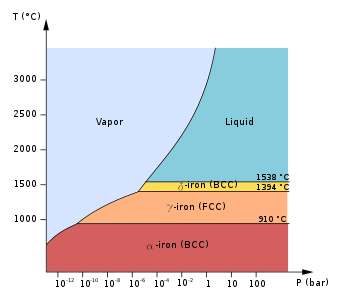 Low-pressure
phase diagram
of pure iron. BCC is
body centered cubic
and FCC is
face-centered cubic
.
Low-pressure
phase diagram
of pure iron. BCC is
body centered cubic
and FCC is
face-centered cubic
.
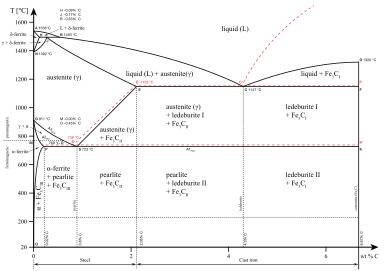 Iron-carbon
eutectic
phase diagram, showing various forms of Fe
x
C
y
substances.
Iron-carbon
eutectic
phase diagram, showing various forms of Fe
x
C
y
substances.
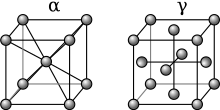 Iron allotropes, showing the differences in structure. The alpha iron (α-Fe) is a body-centered cubic (BCC) and the gamma iron (γ-Fe) is a face-centered cubic (FCC).
Iron allotropes, showing the differences in structure. The alpha iron (α-Fe) is a body-centered cubic (BCC) and the gamma iron (γ-Fe) is a face-centered cubic (FCC).
At
atmospheric pressure
, three
allotropic
forms of
iron
exist, depending on temperature:
alpha iron (α-Fe, ferrite)
,
gamma iron (γ-Fe, austenite)
, and
delta iron (δ-Fe)
. At very high pressure, a fourth form exists,
epsilon iron (ε-Fe, hexaferrum)
. Some controversial experimental evidence suggests the existence of a fifth high-pressure form that is stable at very high pressures and temperatures.
[1]
The phases of iron at atmospheric pressure are important because of the differences in solubility of
carbon
, forming different types of
steel
. The high-pressure phases of iron are important as models for the solid parts of planetary cores. The
inner core
of the
Earth
is generally assumed to consist essentially of a crystalline iron-nickel
alloy
with ε structure.
[2]
[3]
[4]
The outer core surrounding the solid inner core is believed to be composed of liquid iron mixed with
nickel
and trace amounts of lighter elements.
Standard pressure allotropes
[
edit
]
Alpha iron (α-Fe)
[
edit
]
Below 912 °C (1,674 °F), iron has a
body-centered cubic
(bcc)
crystal
structure and is known as α-iron or
ferrite
. It is
thermodynamically stable
and a fairly soft metal. α-Fe can be subjected to pressures up to ca. 15 GPa before transforming into a high-pressure form termed ε-Fe discussed below.
Magnetically, α-iron is
paramagnetic
at high temperatures. However, below its
Curie temperature
(
T
C
or
A
2
) of 771 °C (1044K or 1420 °F),
[5]
it becomes
ferromagnetic
. In the past, the paramagnetic form of α-iron was known as
beta iron
(β-Fe).
[6]
[7]
Even though the slight tetragonal distortion in the ferromagnetic state does constitute a true phase transition, the continuous nature of this transition results in only minor importance in steel
heat treating
. The A
2
line forms the boundary between the beta iron and alpha fields in the
phase diagram
in Figure 1.
Similarly, the A
2
boundary is of only minor importance compared to the A
1
(
eutectoid
), A
3
and A
cm
critical temperatures. The A
cm
, where
austenite
is in equilibrium with
cementite
+ γ-Fe, is beyond the right edge in Fig. 1. The α + γ phase field is, technically, the β + γ field above the A
2
. The beta designation maintains continuity of the Greek-letter progression of phases in iron and steel: α-Fe, β-Fe,
austenite
(γ-Fe), high-temperature δ-Fe, and high-pressure
hexaferrum
(ε-Fe).
 Molar volume vs. pressure for α-Fe at room temperature.
Molar volume vs. pressure for α-Fe at room temperature.
The primary
phase
of low-carbon or
mild steel
and most
cast irons
at room temperature is
ferromagnetic
α-Fe.
[8]
[9]
It has a hardness of approximately 80
Brinell
.
[10]
[11]
The maximum
solubility
of carbon is about 0.02 wt% at 727 °C (1,341 °F) and 0.001% at 0 °C (32 °F).
[12]
When it dissolves in iron, carbon atoms occupy interstitial "holes". Being about twice the diameter of the
tetrahedral hole
, the carbon introduces a strong local strain field.
Mild steel
(carbon steel with up to about 0.2 wt% C) consists mostly of α-Fe and increasing amounts of
cementite
(Fe
3
C, an iron carbide). The mixture adopts a lamellar structure called
pearlite
. Since
bainite
and pearlite each contain α-Fe as a component, any iron-carbon alloy will contain some amount of α-Fe if it is allowed to reach
equilibrium
at room temperature. The amount of α-Fe depends on the cooling process.
A
2
critical temperature and induction heating
[
edit
]
 Figure 1:
The beta field and A
2
critical temperature on the iron-rich side of the iron-carbon phase diagram.
[5]
Figure 1:
The beta field and A
2
critical temperature on the iron-rich side of the iron-carbon phase diagram.
[5]
β-Fe and the A
2
critical temperature are important in
induction heating
of steel, such as for surface-hardening heat treatments. Steel is typically austenitized at 900?1000 °C before it is
quenched
and
tempered
. The high-frequency alternating magnetic field of induction heating heats the steel by two mechanisms below the Curie temperature: resistance or
Joule heating
and ferromagnetic
hysteresis
losses. Above the A
2
boundary, the hysteresis mechanism disappears and the required amount of energy per degree of temperature increase is thus substantially larger than below A
2
. Load-matching circuits may be needed to vary the
impedance
in the induction power source to compensate for the change.
[13]
Gamma iron (γ-Fe)
[
edit
]
When heating iron above 912 °C (1,674 °F), its
crystal structure
changes to a
face-centered cubic
(fcc) crystalline structure. In this form it is called gamma iron (γ-Fe) or
austenite
. γ-iron can dissolve considerably more carbon (as much as 2.04% by mass at 1,146 °C). This γ form of carbon saturation is exhibited in
austenitic stainless steel
.
Delta iron (δ-Fe)
[
edit
]
Peculiarly, above 1,394 °C (2,541 °F) iron changes back into the bcc structure, known as δ-Fe.
[14]
δ-iron can dissolve as much as 0.08% of carbon by mass at 1,475 °C. It is stable up to its melting point of 1,538 °C (2,800 °F). δ-Fe cannot exist above 5.2 GPa, with austenite instead transitioning directly to a molten phase at these high pressures.
[15]
High pressure allotropes
[
edit
]
Epsilon iron / Hexaferrum (ε-Fe)
[
edit
]
At pressures above approximately 10-13 GPa and temperatures up to around 700 K, α-iron changes into a
hexagonal close-packed
(hcp) structure, which is also known as ε-iron or hexaferrum;
[16]
the higher-temperature γ-phase also changes into ε-iron, but generally requires far higher pressures as temperature increases. The
triple point
of hexaferrum, ferrite, and austenite is 10.5 GPa at 750 K.
[15]
Antiferromagnetism
in alloys of epsilon-Fe with Mn, Os and Ru has been observed.
[17]
Experimental high temperature and pressure
[
edit
]
An alternate stable form, if it exists, may appear at pressures of at least 50 GPa and temperatures of at least 1,500 K; it has been thought to have an
orthorhombic
or a double hcp structure.
[1]
As of December 2011
[update]
, recent and ongoing experiments are being conducted on high-pressure and
superdense carbon allotropes
.
Phase transitions
[
edit
]
Melting and boiling points
[
edit
]
The melting point of iron is experimentally well defined for pressures less than 50 GPa.
For greater pressures, published data (as of 2007) put the γ-ε-liquid
triple point
at pressures that differ by tens of gigapascals and 1000 K in the melting point. Generally speaking,
molecular dynamics
computer simulations of iron melting and shock wave experiments suggest higher melting points and a much steeper slope of the melting curve than static experiments carried out in
diamond anvil cells
.
[18]
The melting and boiling points of iron, along with its
enthalpy of atomization
, are lower than those of the earlier group 3d elements from
scandium
to
chromium
, showing the lessened contribution of the
3d electrons
to metallic bonding as they are attracted more and more into the inert core by the
nucleus
;
[19]
however, they are higher than the values for the previous element
manganese
because that element has a half-filled 3d subshell and consequently its d-electrons are not easily
delocalized
. This same trend appears for
ruthenium
but not
osmium
.
[20]
Structural phase transitions
[
edit
]
The exact temperatures at which iron will transition from one crystal structure to another depends on how much and what type of other elements are dissolved in the iron. The phase boundary between the different solid phases is drawn on a
binary phase diagram
, usually plotted as temperature versus percent iron. Adding some elements, such as
Chromium
, narrows the temperature range for the gamma phase, while others increase the temperature range of the gamma phase. In elements that reduce the gamma phase range, the alpha-gamma phase boundary connects with the gamma-delta phase boundary, forming what is usually called the
Gamma loop
. Adding Gamma loop additives keeps the iron in a body-centered cubic structure and prevents the steel from suffering
phase transition
to other solid states.
[21]
See also
[
edit
]
References
[
edit
]
- ^
a
b
Boehler, Reinhard (2000).
"High-pressure experiments and the phase diagram of lower mantle and core materials"
.
Reviews of Geophysics
.
38
(2). American Geophysical Union: 221?245.
Bibcode
:
2000RvGeo..38..221B
.
doi
:
10.1029/1998RG000053
.
S2CID
33458168
.
- ^
Cohen, Ronald E.; Stixrude, Lars.
"Crystal at the Center of the Earth"
. Archived from
the original
on 5 February 2007
. Retrieved
2007-02-05
.
- ^
Stixrude, Lars; Cohen, Ronald E. (March 1995). "High-Pressure Elasticity of Iron and Anisotropy of Earth's Inner Core".
Science
.
267
(5206): 1972?5.
Bibcode
:
1995Sci...267.1972S
.
doi
:
10.1126/science.267.5206.1972
.
PMID
17770110
.
S2CID
39711239
.
- ^
"What is at the centre of the Earth?"
.
BBC News
. 31 August 2011.
- ^
a
b
Alloy Phase Diagrams
. ASM Handbook. Vol. 3. ASM International. 1992. pp. 2.210, 4.9.
ISBN
978-0-87170-381-1
.
- ^
Bullens, Denison Kingsley,
Steel and its Heat Treatment
, Vol. I, Fourth Ed., J. Wiley & Sons Inc., 1938, p. 86
- ^
Avner, Sydney H. (1974).
Introduction to physical metallurgy
(2nd ed.). McGraw-Hill. p. 225.
ISBN
978-0-07-002499-1
.
- ^
Maranian, Peter (2009),
Reducing Brittle and Fatigue Failures in Steel Structures
, New York: American Society of Civil Engineers,
ISBN
978-0-7844-1067-7
.
- ^
Greenwood, Norman N.
; Earnshaw, Alan (1997).
Chemistry of the Elements
(2nd ed.).
Butterworth-Heinemann
.
ISBN
978-0-08-037941-8
.
- ^
Structure of plain steel
, retrieved
2008-10-21
.
- ^
Alvarenga, Henrique Duarte; Van de Putte, Tom; Van Steenberge, Nele; Setsma, Jilt; Terryn, Herman (January 2015). "Influence of Carbide Morphology and Microstructure on the Kinetics of Superficial Decarburization of C-Mn Steels".
Metall Mater Trans A
.
46
(1): 123?133.
Bibcode
:
2015MMTA...46..123A
.
doi
:
10.1007/s11661-014-2600-y
.
S2CID
136871961
.
- ^
Smith, William F.; Hashemi, Javad (2006).
Foundations of Materials Science and Engineering
(4th ed.). McGraw-Hill. p. 363.
ISBN
0-07-295358-6
.
- ^
Semiatin, S. Lee; Stutz, David E. (1986).
Induction Heat Treatment of Steel
. ASM International. pp. 95?98.
ISBN
978-0-87170-211-1
.
- ^
Lyman, Taylor, ed. (1973).
Metallography, Structures and Phase Diagrams
. Metals Handbook. Vol. 8 (8th ed.). Metals Park, Ohio: ASM International.
OCLC
490375371
.
- ^
a
b
Anzellini, Simone; Errandonea, Daniel (2021-09-29).
"Properties of Transition Metals and Their Compounds at Extreme Conditions"
.
Crystals
.
11
(10): 1185.
doi
:
10.3390/cryst11101185
.
ISSN
2073-4352
.
- ^
Mathon, Olivier; Baudelet, Francois; Itie, J. Paul; Polian, Alain; d'Astuto, Matteo; Chervin, Jean-Claude; Pascarelli, Sakura (14 December 2004). "Dynamics of the magnetic and structural alpha-epsilon phase transition in iron".
Physical Review Letters
.
93
(25): 255503.
arXiv
:
cond-mat/0405439
.
Bibcode
:
2004PhRvL..93y5503M
.
doi
:
10.1103/PhysRevLett.93.255503
.
PMID
15697906
.
S2CID
19228886
.
- ^
Fletcher, Geoffrey C.; Addis, Robert P. (November 1974).
"The magnetic state of the phase of iron"
(PDF)
.
Journal of Physics F: Metal Physics
.
4
(11): 1954.
Bibcode
:
1974JPhF....4.1951F
.
doi
:
10.1088/0305-4608/4/11/020
. Retrieved
December 30,
2011
.
- ^
Boehler, Reinhard; Ross, M. (2007). "Properties of Rocks and Minerals_High-Pressure Melting".
Mineral Physics
. Treatise on Geophysics. Vol. 2. Elsevier. pp. 527?41.
doi
:
10.1016/B978-044452748-6.00047-X
.
ISBN
978-0-444-52748-6
.
- ^
Greenwood and Earnshaw, p. 1116
- ^
Greenwood and Earnshaw, pp. 1074?75
- ^
Kutz, Myer, ed. (2002-07-22).
Handbook of Materials Selection
. p. 44.
ISBN
978-0-471-35924-1
. Retrieved
December 19,
2013
.