From Wikipedia, the free encyclopedia
Organic compound with one carboxyl and two hydroxyl groups
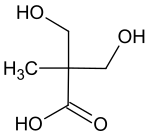
Dimethylolpropionic acid
|
| Names
|
Preferred IUPAC name
3-Hydroxy-2-(hydroxymethyl)-2-methylpropanoic acid
|
| Other names
2,2-Bis(hydroxymethyl)propionic acid; Hhmp
|
| Identifiers
|
|
|
|
|
|
|
| ChEMBL
|
|
| ChemSpider
|
|
| ECHA InfoCard
|
100.023.006

|
| EC Number
|
|
|
|
|
| UNII
|
|
|
|
|
InChI=1S/C5H10O4/c1-5(2-6,3-7)4(8)9/h6-7H,2-3H2,1H3,(H,8,9)
Key: PTBDIHRZYDMNKB-UHFFFAOYSA-N
|
|
|
| Properties
|
|
|
C
5
H
10
O
4
|
| Molar mass
|
134.131
g·mol
?1
|
| Melting point
|
190 °C (374 °F; 463 K)
|
| Hazards
|
| GHS
labelling
:
|
|
|

|
|
|
Warning
|
|
|
H319
,
H335
|
|
|
P261
,
P264
,
P271
,
P280
,
P304+P340
,
P305+P351+P338
,
P312
,
P337+P313
,
P403+P233
,
P405
,
P501
|
| Related compounds
|
Related compounds
|
Isobutyric acid
|
Except where otherwise noted, data are given for materials in their
standard state
(at 25 °C [77 °F], 100 kPa).
|
Chemical compound
Dimethylolpropionic acid (DMPA)
is a chemical compound that has the full
IUPAC
name of
2,2-bis(hydroxymethyl)propionic acid
and is an
organic compound
with one carboxyl and two hydroxy groups.
[1]
It has the
CAS Registry Number
of 4767-03-7.
Properties
[
edit
]
DMPA is an odorless free flowing white crystalline solid and essentially non-toxic. DMPA has two different functional groups hydroxyl and carboxylic acid so the molecule can be used for a wide variety of syntheses. In addition to reaction with other chemicals, DMPA can also react with itself to produce
esters
via esterification, as one example.
Uses
[
edit
]
One key use of DMPA is in the field of coatings and adhesives. It is used as a modifier in the production of anionic
Polyurethane dispersions
.
[2]
Solvent soluble binders/resins for coatings can be converted into an aqueous binder with the use of this material. In this case it is reacted with a suitable diisocyanate such as
isophorone diisocyanate
or
TMXDI
usually along with other
polyols
to make a
prepolymer
.
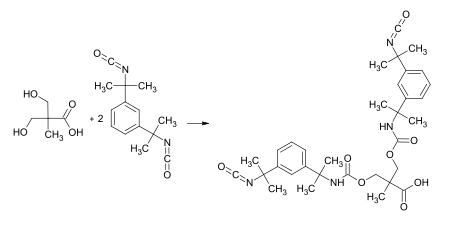 Reaction of DMPA with TMXDI
Reaction of DMPA with TMXDI
There is also the possibility of using 2,2-bis(hydroxymethyl)propionic acid for the synthesis of dendrimeric molecules, also known as hyperbranched molecules.
[3]
When each hydroxyl group is reacted with 2,2-bis (hydroxymethyl) propionic acid, the number of hydroxyl groups present in the molecule doubles. Repeating this reaction step, produces one more shell each time and thus the molecule grows. If at the end the hydroxyl groups are reacted with a bifunctional component, dendrimeric UV binders can be produced, for example. Dendrimeric molecules have low solution viscosities and improved properties.
It has a wide variety of other uses including production of
[4]
hyperbranched polyesters, waterborne polyesters, waterbased alkyd resins, and aqueous epoxy resins.
[5]
It has even found use in
polyethylene terephthalate
fiber production.
[6]
[7]
Another use is in the medical field for drug release purposes. In the business world it has been cited as an outstanding growth opportunity
[8]
See also
[
edit
]
References
[
edit
]
- ^
PubChem.
"2,2-Bis(hydroxymethyl)propionic acid"
.
pubchem.ncbi.nlm.nih.gov
. Retrieved
2019-07-17
.
- ^
Howarth, GA (2003-06-01). "Polyurethanes, polyurethane dispersions and polyureas: Past, present and future".
Surface Coatings International Part B: Coatings Transactions
.
86
(2): 111?118.
doi
:
10.1007/BF02699621
.
ISSN
1476-4865
.
- ^
Karakaya, Ceylan; Gunduz, Gungor; Aras, Leyla; Mecido?lu, ?dris A. (2007-07-02). "Synthesis of oil based hyperbranched resins and their modification with melamine-formaldehyde resin".
Progress in Organic Coatings
.
59
(4): 265?273.
doi
:
10.1016/j.porgcoat.2007.03.004
.
ISSN
0300-9440
.
- ^
Zhang, Xinli (2011). "Modifications and applications of hyperbranched aliphatic polyesters based on dimethylolpropionic acid".
Polymer International
.
60
(2): 153?166.
doi
:
10.1002/pi.2930
.
ISSN
1097-0126
.
- ^
CN application 105152907
, Yang, Kunwu, "Synthetic method of 2,2-dimethylolpropionic acid", issued 2015-08-18, assigned to Huzhou Changsheng Chemical Co. Ltd.
- ^
Huang, Zhaosong; Bi, Long; Zhang, Zhenyu; Han, Yisheng (2012-10-01).
"Effects of dimethylolpropionic acid modification on the characteristics of polyethylene terephthalate fibers"
.
Molecular Medicine Reports
.
6
(4): 709?715.
doi
:
10.3892/mmr.2012.1012
.
ISSN
1791-2997
.
PMID
22858692
.
- ^
Kaitwade, Nikhil.
"How Dimethylolpropionic Acid is used in Drug Release | FMI | Industry news and insights"
. Retrieved
2020-03-03
.
- ^
"Dimethylolpropionic Acid Market (Edition:2020) Outstanding Growth ? Henan Tianfu Chemical Co Ltd, Jiangxi Nancheng Hongdu Chemical Technology Development Co Ltd, Shenzhen Vtolo Chemicals Co Ltd"
.
AP NEWS
. 2020-02-18
. Retrieved
2020-03-03
.
External links
[
edit
]